 Back
BackWater and Life: Properties, Acids, and Bases
Study Guide - Smart Notes

Water and Life
Importance of Water in Biology
Water is fundamental to life on Earth, making up the majority of living organisms and dominating the environment. Its unique physical and chemical properties enable life to exist and thrive.
Abundance: Water covers about three-quarters of Earth's surface and is the only common substance that exists naturally as a solid, liquid, and gas.
Cellular Composition: Most cells are 70–95% water, and life originated in aquatic environments.
Biochemical Role: Water acts as a reactant in many metabolic reactions and surrounds cells, providing a medium for biological processes.
Structure and Polarity of Water
Polar Covalent Bonds and Hydrogen Bonding
The structure of water molecules and their polarity are central to water's unique properties.
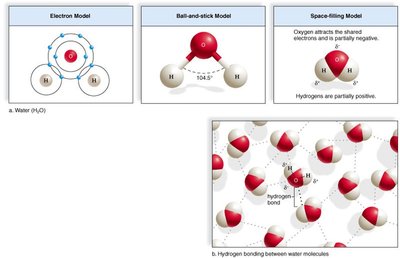
Molecular Shape: Water (H2O) has a bent, V-shaped structure with two hydrogen atoms covalently bonded to an oxygen atom.
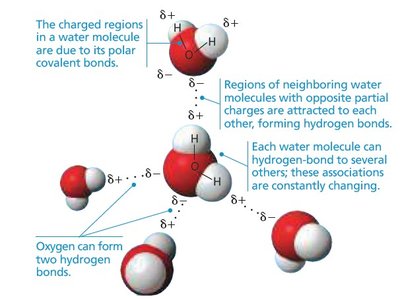
Polarity: Oxygen is more electronegative than hydrogen, resulting in a polar molecule with partial negative (δ-) charge near oxygen and partial positive (δ+) charges near hydrogens.
Hydrogen Bonding: The polarity allows water molecules to form hydrogen bonds with each other, leading to many of water's special properties.

Properties of Water
Key Physical and Chemical Properties
Water's polarity and hydrogen bonding result in several important properties that are essential for life.
Polarity: Water is a polar molecule, making it an excellent solvent for ionic and polar substances.
Universal Solvent: Water dissolves more substances than any other liquid, facilitating biochemical reactions.
Cohesion: Water molecules stick together due to hydrogen bonding, aiding in the transport of water in plants.
Adhesion: Water molecules can also stick to other substances, helping water move against gravity in plant vessels.
High Heat Capacity: Water absorbs and releases large amounts of heat with little temperature change, stabilizing environments and organisms.
High Surface Tension: Water's surface resists external force, allowing small organisms to move on its surface.
Density of Ice: Solid water (ice) is less dense than liquid water, causing ice to float and insulate aquatic life.

Water as a Solvent
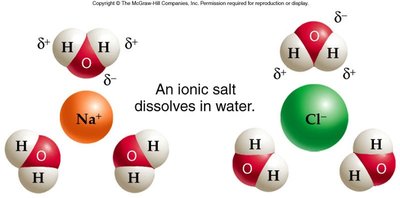
Water's polarity enables it to dissolve many substances, making it the medium for most biological chemistry.
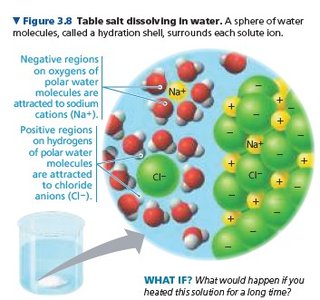
Solution: A mixture of a solute (substance dissolved) and a solvent (dissolving agent, usually water).
Hydration Shell: Water molecules surround dissolved ions, stabilizing them in solution.
Hydrophilic and Hydrophobic Substances
Substances interact with water differently based on their chemical nature.
Hydrophilic: Water-loving substances (e.g., salts, sugars) are polar or ionic and dissolve easily in water.
Hydrophobic: Water-fearing substances (e.g., oils) are nonpolar and do not dissolve in water.
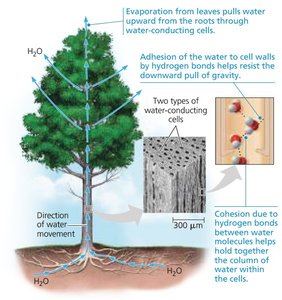
Cohesion and Adhesion in Plants
Cohesion and adhesion are critical for water transport in plants, allowing water to move upward against gravity.
Cohesion: Hydrogen bonds hold water molecules together, enabling the upward pull of water in plant vessels.
Adhesion: Water molecules adhere to the walls of plant vessels, assisting in water movement.
High Heat Capacity and Specific Heat
Water's high specific heat helps buffer temperature changes in organisms and environments.
Definition: Specific heat is the amount of heat required to raise the temperature of 1 g of a substance by 1°C.
Biological Importance: Water's high specific heat stabilizes ocean and body temperatures, protecting organisms from rapid temperature changes.
Heat of Vaporization and Evaporative Cooling
Water requires significant energy to change from liquid to gas, which helps moderate Earth's climate and cool organisms through evaporation.
Heat of Vaporization: The energy needed to convert 1 g of liquid water to gas (about 580 cal at room temperature).
Evaporative Cooling: As water evaporates, it removes heat, cooling surfaces (e.g., sweating in mammals).
Surface Tension
Water's surface tension is a result of cohesive forces, allowing certain organisms to move on its surface.
Definition: Surface tension is the force required to break the surface of a liquid.
Biological Example: Some insects, like the raft spider, can walk on water due to high surface tension.

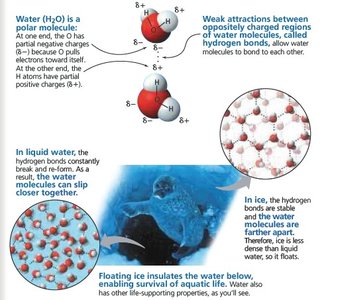
Density of Ice and Its Ecological Importance
Ice is less dense than liquid water, causing it to float and insulate aquatic environments.
Maximum Density: Water is densest at 4°C; below this temperature, it expands as it freezes.
Ecological Impact: Floating ice insulates water below, allowing aquatic life to survive in cold climates. Ice also provides habitat for Arctic animals.


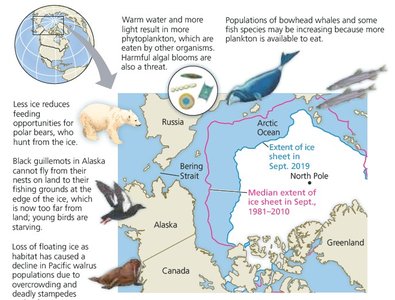
Effect of Climate Change on the Arctic
Climate change is reducing Arctic ice, threatening habitats and altering ecosystems.
Loss of Ice: Reduces habitat for species like polar bears and seals, and affects food webs.
Wider Impacts: Changes in ice cover influence global climate and marine biodiversity.
Acids, Bases, and pH
Dissociation of Water and pH Scale
Water can dissociate into hydrogen ions (H+) and hydroxide ions (OH−), affecting the acidity or basicity of solutions.
Dissociation Reaction:
pH Definition: pH is the negative logarithm of the hydrogen ion concentration:
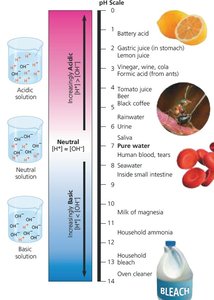
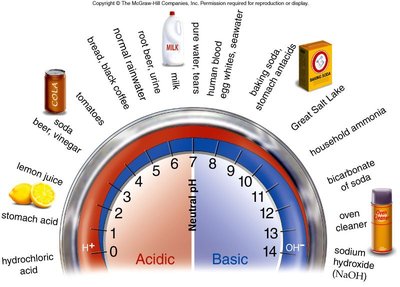
pH Scale: Ranges from 1 (most acidic) to 14 (most basic), with 7 being neutral.
Acids and Bases
Acids and bases alter the concentration of H+ and OH− in solutions, impacting biological systems.
Acids: Substances that increase H+ concentration (e.g., HCl dissociates to H+ and Cl−).
Bases: Substances that decrease H+ concentration, often by releasing OH− (e.g., NaOH dissociates to Na+ and OH−).
Biological Importance of pH
Organisms are highly sensitive to pH changes, which can disrupt cellular processes.
Homeostasis: Human blood pH is tightly regulated around 7.4; deviations can be fatal.
Buffers: Biological fluids contain buffers that minimize pH changes by accepting or donating H+ ions.
Buffers in Biological Systems
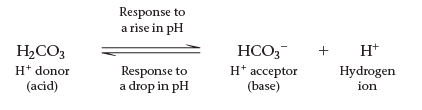
Buffers are mixtures of weak acids and their conjugate bases that stabilize pH in biological fluids.
Mechanism: Buffers accept H+ when in excess and donate H+ when depleted.
Example: The carbonic acid-bicarbonate buffer system in blood:
Environmental Acidification
Human activities, such as burning fossil fuels, increase CO2 and other pollutants, leading to acidification of oceans and precipitation.
Ocean Acidification: CO2 dissolves in water to form carbonic acid, lowering ocean pH and threatening marine life.
Acid Precipitation: Sulfur and nitrogen oxides form strong acids in the atmosphere, harming ecosystems.
Regulation: Environmental policies, such as the Clean Air Act, aim to reduce acidifying emissions.
