 Back
BackCell and Molecular Biology: Foundations, Biochemistry, and Bioenergetics
Study Guide - Smart Notes

Introduction to Cell Biology
The Discovery of Cells
Cell biology began with the discovery of cells by Robert Hooke in 1665, who observed chambers in cork and named them 'cells.' Later, Anton van Leeuwenhoek was the first to describe living single cells, observing 'animalcules' in pond water. By the 1830s, the importance of cells as the fundamental units of life was widely recognized.

Basic Properties of Cells
Cells are the smallest units of life and exhibit several key properties:
Growth and Reproduction: Cells can grow and reproduce, even outside the organism in laboratory culture (e.g., HeLa cells).
Complexity and Organization: Cells have highly organized structures, with organelles arranged in predictable patterns.
Genetic Program: Cells possess genetic material (DNA) that encodes instructions for cellular activities and reproduction.
Self-Replication: Cells reproduce by division (mitosis and meiosis), ensuring equal distribution of genetic material.
Energy Acquisition and Use: Cells acquire energy (photosynthesis, respiration) and use it to maintain complexity.
Metabolism: Cells carry out chemical reactions, collectively known as metabolism, often catalyzed by enzymes.
Mechanical Activities: Cells move materials, assemble/disassemble structures, and can move themselves.
Response to Stimuli: Cells sense and respond to environmental changes via receptors.
Self-Regulation: Cells regulate their internal environment; breakdowns can lead to diseases like cancer.
Evolution: Cells evolve over time, forming the basis of biological diversity.
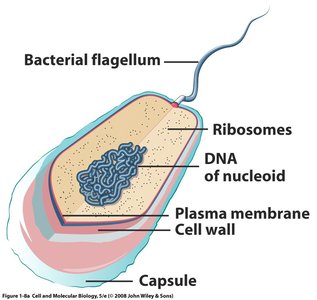
Prokaryotic and Eukaryotic Cells
Fundamental Differences
Cells are classified into two major types: Prokaryotes and Eukaryotes. Prokaryotes (bacteria, cyanobacteria) are structurally simpler and lack a nucleus, while eukaryotes (protists, fungi, plants, animals) are more complex and possess a nucleus and membrane-bound organelles.
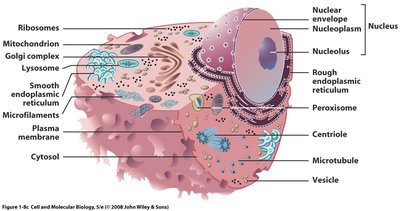
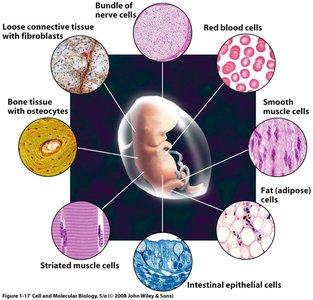
Multicellularity and Differentiation
Multicellular organisms exhibit cell differentiation, where specialized cells perform distinct functions, providing advantages through division of labor.
Comparison of Prokaryotic and Eukaryotic Cells
Both cell types share several features, but eukaryotes possess additional complexity:
Feature | Prokaryotes | Eukaryotes |
|---|---|---|
Plasma membrane | Yes | Yes |
Genetic material | DNA, no nucleus | DNA in nucleus |
Organelles | Absent | Present |
Cell division | Binary fission | Mitosis/meiosis |
Metabolic pathways | Shared | Shared |
Photosynthesis | Cyanobacteria | Plants, algae |


Viruses, Viroids, and Prions
Non-Living Biological Agents
Viruses are non-living agents composed of a protein coat and nucleic acid. They are obligate intracellular parasites, requiring host machinery for replication. Viroids are simpler, consisting of a short RNA strand without a protein coat, and typically cause plant diseases. Prions are infectious proteins lacking nucleic acid, responsible for neurodegenerative diseases in animals and humans.
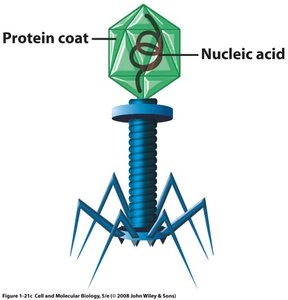
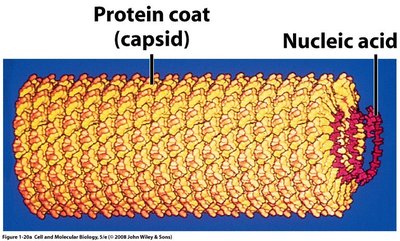
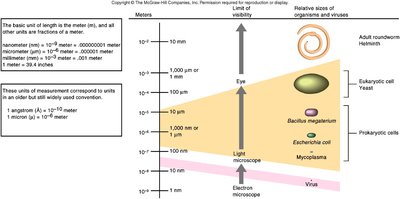
Size in the Microbial World
Microbial entities vary greatly in size, with viruses being much smaller than cells and only visible with electron microscopy.
Biochemistry: Atoms, Elements, and Chemical Bonds
Atoms and Elements
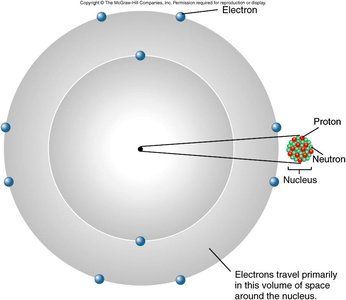
Atoms are the basic units of matter, composed of protons, neutrons, and electrons. The arrangement of electrons in orbitals determines chemical properties and bonding behavior.
Chemical Bonds
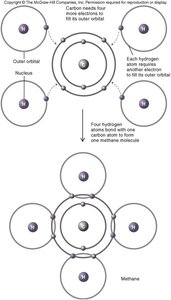
Atoms achieve stability by filling their outer electron orbitals, forming bonds through electron sharing or transfer. The main types of chemical bonds are:
Covalent Bonds: Electrons are shared between atoms, forming strong, stable bonds. Can be polar or non-polar.
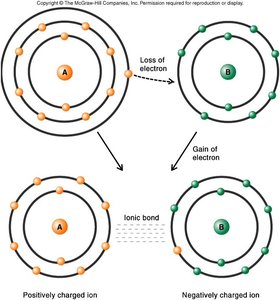
Ionic Bonds: Electrons are transferred, creating charged ions that attract each other.
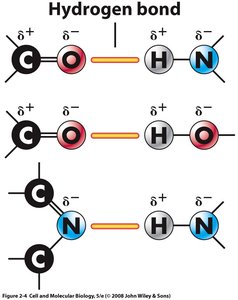
Hydrogen Bonds: Weak attractions between polar molecules, important in water and DNA structure.
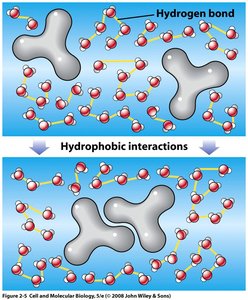
Hydrophobic Interactions: Nonpolar molecules aggregate to minimize contact with water.
van der Waals Forces: Weak, transient attractions between nonpolar molecules.
Acids, Bases, and Buffers
Definitions and Properties
Acids are molecules that donate protons (H+), while bases accept protons. The strength of acids and bases varies, and they exist in conjugate pairs. Buffers help maintain stable pH by binding or releasing H+ and OH- ions.
pH and Calculations
pH is a measure of hydrogen ion concentration:
Biological Macromolecules
Types of Biological Molecules
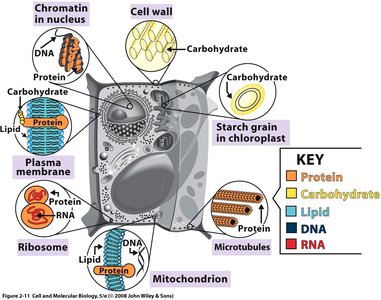
Cells contain four major classes of biological molecules:
Carbohydrates: Energy storage and structural components. Monosaccharides (simple sugars) can form disaccharides and oligosaccharides.
Lipids: Nonpolar molecules (fats, oils, phospholipids, sterols) important for membranes and energy storage.
Proteins: Polymers of amino acids, serving as enzymes, structural elements, and regulatory molecules.
Nucleic Acids: DNA and RNA, polymers of nucleotides, store and transmit genetic information.
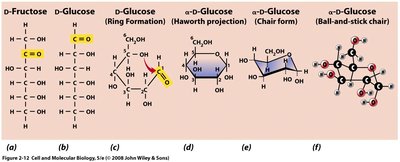
Carbohydrates
Carbohydrates have the general formula (CH2O)n and include monosaccharides (3-7 carbons), disaccharides (e.g., sucrose, lactose), and oligosaccharides. They serve as energy sources and building materials.
Lipids
Lipids are composed mainly of C, H, and O, and are insoluble in water. They include fats, oils, phospholipids, and sterols, and play roles in energy storage, membrane structure, and signaling.
Proteins
Proteins are polymers of 20 different amino acids, each with a central carbon, amino group, carboxyl group, and variable side chain (R group). Proteins have four levels of structure:
Primary: Sequence of amino acids.
Secondary: Local folding (α-helix, β-sheet).
Tertiary: Overall 3D shape (globular or fibrous).
Quaternary: Association of multiple polypeptide chains.
Nucleic Acids
Nucleic acids (DNA and RNA) are polymers of nucleotides, each consisting of a five-carbon sugar, a nitrogenous base (purine or pyrimidine), and a phosphate group. DNA is double-stranded and forms a double helix; RNA is typically single-stranded.
Bioenergetics, Enzymes, and Metabolism
Bioenergetics and Energy Transformations
Bioenergetics studies energy transformations in living organisms. Energy exists as potential (position) or kinetic (motion). The First Law of Thermodynamics states that energy cannot be created or destroyed, only transformed. The Second Law of Thermodynamics states that entropy (disorder) increases in spontaneous processes.
Free Energy and Thermodynamics
Free energy (G) is the energy available to do work. The direction and spontaneity of reactions are predicted by the Gibbs free energy equation:
Spontaneous reactions have (exergonic); non-spontaneous reactions have (endergonic).
At equilibrium:
Equilibrium constant:
Standard free energy change:
ATP Hydrolysis
ATP hydrolysis is a key exergonic reaction in cells:
kcal/mol (standard conditions)
Actual free energy change depends on concentrations of reactants and products:
Enzymes and Metabolic Pathways
Enzymes are biological catalysts that accelerate metabolic reactions. Enzyme activity is affected by pH, temperature, and inhibitors. Metabolism includes catabolism (breakdown of molecules for energy) and anabolism (synthesis of complex molecules).
Redox Reactions and Energy Carriers
During catabolism, carbon is more reduced when it carries more energy. NAD+ is an essential electron carrier, regenerated in the absence of oxygen via fermentation.
Additional info: For exam preparation, students should be able to calculate pH, pOH, [OH-], and [H+], understand the role of buffers, and solve problems involving free energy changes and equilibrium constants.
----------------------------------------
