 Back
BackChemical Components of Life: Cell Biology Study Guide
Study Guide - Smart Notes

Chemical Context of Life
Elements and Atoms
The foundation of cell biology begins with understanding the chemical elements and atoms that compose living organisms. Elements are substances that cannot be broken down into simpler substances by chemical means, and atoms are the smallest units of elements.
Elements: Key elements in biology include carbon, hydrogen, oxygen, nitrogen, and phosphorus.
Atoms: Atoms consist of protons, neutrons, and electrons. The atomic number is determined by the number of protons.
Molecules: Molecules are groups of atoms bonded together, forming the chemical basis of life.
Electronegativity and Bond Types
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. It influences the type of bond formed between atoms.
Electronegativity: Higher electronegativity means stronger attraction for electrons (e.g., O > N > C > H).
Bond Types:
Covalent Bonds: Atoms share electrons. Can be polar (unequal sharing) or non-polar (equal sharing).
Hydrogen Bonds: Weak bonds between a hydrogen atom and an electronegative atom (e.g., O or N).
Van der Waals Interactions: Weak attractions due to transient local partial charges.

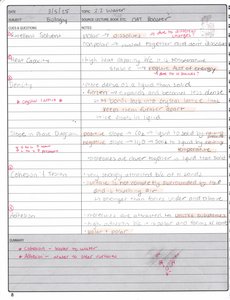
Water: Properties and Biological Importance
Water is essential for life due to its unique chemical and physical properties, which arise from its molecular structure and hydrogen bonding.
Universal Solvent: Water dissolves many substances, facilitating biochemical reactions.
High Specific Heat: Water resists temperature changes, stabilizing environments.
High Heat of Vaporization: Water absorbs large amounts of energy during evaporation.
Cohesion and Adhesion: Water molecules stick to each other (cohesion) and to other surfaces (adhesion).
Surface Tension: Water forms a "skin" at its surface due to cohesive forces.
Density: Ice is less dense than liquid water, allowing it to float.
Macromolecules
Carbohydrates
Carbohydrates are organic molecules composed of carbon, hydrogen, and oxygen. They serve as energy sources and structural components in cells.
Monosaccharides: Simple sugars (e.g., glucose, fructose).
Disaccharides: Two monosaccharides joined by glycosidic bonds (e.g., sucrose).
Polysaccharides: Long chains of monosaccharides (e.g., starch, glycogen, cellulose).
Functions: Energy storage, structural support.

Lipids
Lipids are hydrophobic molecules that include fats, oils, phospholipids, and steroids. They are important for energy storage, membrane structure, and signaling.
Fatty Acids: Long hydrocarbon chains with a carboxyl group.
Triglycerides: Glycerol + 3 fatty acids; main form of energy storage.
Phospholipids: Major component of cell membranes; amphipathic (hydrophilic head, hydrophobic tail).
Steroids: Four fused carbon rings; includes cholesterol and hormones.


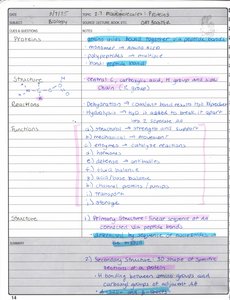
Proteins
Proteins are polymers of amino acids, performing a wide range of functions in cells including catalysis, structure, transport, and regulation.
Amino Acids: Building blocks of proteins; contain amino, carboxyl, and side chain (R group).
Peptide Bonds: Link amino acids in polypeptide chains.
Structure:
Primary: Sequence of amino acids.
Secondary: Local folding (α-helix, β-sheet) stabilized by hydrogen bonds.
Tertiary: 3D folding due to interactions among side chains.
Quaternary: Multiple polypeptide chains assembled together.
Functions: Enzymes, structural proteins, transport, signaling.

Nucleic Acids
Nucleic acids (DNA and RNA) store and transmit genetic information. They are polymers of nucleotides, each consisting of a sugar, phosphate, and nitrogenous base.
DNA: Double helix, stores genetic information.
RNA: Single-stranded, involved in protein synthesis.
Nucleotides: Composed of a pentose sugar, phosphate group, and nitrogenous base (A, T, G, C for DNA; A, U, G, C for RNA).
Base Pairing: A-T (or A-U in RNA), G-C; stabilized by hydrogen bonds.
Functions: Information storage, transmission, and expression.



Additional info:
These notes cover the chemical components of cells, including atoms, molecules, water, and macromolecules, which are foundational to cell biology.
Key equations:
