 Back
BackChapter 1 Part 2 Macromolecules of the Cell: Nucleic Acids, Polysaccharides, and Lipids
Study Guide - Smart Notes

Nucleic Acids
Structure and Function of Nucleic Acids
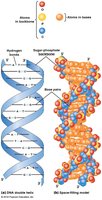
Nucleic acids are essential macromolecules in cells, responsible for the storage, transmission, and expression of genetic information. They are linear polymers composed of nucleotide monomers. The two primary types are DNA (deoxyribonucleic acid) and RNA (ribonucleic acid).
DNA: Stores genetic information and is typically double-stranded.
RNA: Transmits genetic information and is usually single-stranded.
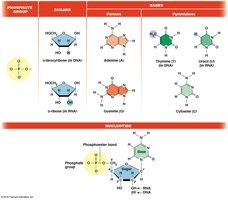
Nucleotides: Consist of a phosphate group, a 5-carbon sugar (deoxyribose in DNA, ribose in RNA), and a nitrogenous base (purines: adenine, guanine; pyrimidines: cytosine, thymine in DNA, uracil in RNA).
Acidic Component: The phosphate group is responsible for the acidic nature of nucleic acids.
Nucleic Acid Nomenclature
Nucleotides are named based on their base and the number of phosphate groups attached. For example, adenosine monophosphate (AMP) has one phosphate, adenosine diphosphate (ADP) has two, and adenosine triphosphate (ATP) has three.
Nucleoside: Base + sugar (e.g., adenosine).
Nucleotide: Nucleoside + phosphate(s) (e.g., AMP, ADP, ATP).

Bases | Nucleoside | Nucleotide | Deoxynucleoside | Deoxynucleotide |
|---|---|---|---|---|
Adenine | Adenosine | AMP | Deoxyadenosine | dAMP |
Guanine | Guanosine | GMP | Deoxyguanosine | dGMP |
Cytosine | Cytidine | CMP | Deoxycytidine | dCMP |
Uracil (RNA only) | Uridine | UMP | — | — |
Thymine (DNA only) | — | — | Deoxythymidine | dTMP |
Polymerization and Directionality
Nucleic acids are formed by linking nucleotides via a 3ʹ,5ʹ phosphodiester bridge. This creates a polynucleotide with directionality, having a 5ʹ phosphate group at one end and a 3ʹ hydroxyl group at the other. Sequences are written from 5ʹ to 3ʹ.
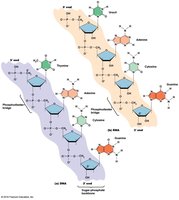
Nucleic Acid Synthesis and Base Pairing
Nucleic acid synthesis requires a template to ensure correct nucleotide addition. Complementary base pairing occurs between purines and pyrimidines:
Adenine (A) pairs with Thymine (T) (or Uracil (U) in RNA) via two hydrogen bonds.
Guanine (G) pairs with Cytosine (C) via three hydrogen bonds.
DNA strands are antiparallel.
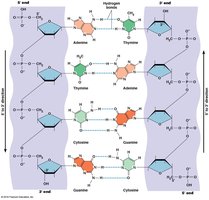
Double-Stranded Helix Structure
The DNA molecule forms a double-stranded helix stabilized by hydrogen bonds between complementary bases and base stacking interactions. RNA, while usually single-stranded, can form secondary structures through internal base pairing.
Polysaccharides
Structure and Classification of Sugars
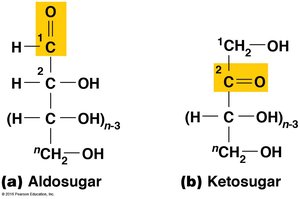
Polysaccharides are long-chain polymers of sugars, serving structural and storage functions. Sugars are classified based on their carbonyl group (aldosugars or ketosugars) and the number of carbon atoms.
Aldosugars: Terminal carbonyl group.
Ketosugars: Internal carbonyl group.
Classifications: Trioses (3C), Tetroses (4C), Pentoses (5C), Hexoses (6C), Heptoses (7C).
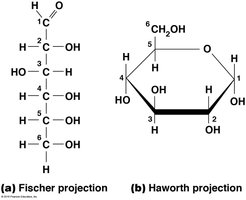
Glucose Structure and Ring Forms
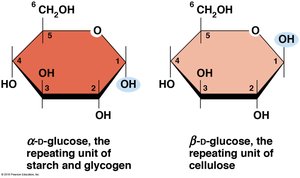
Glucose, a hexose, can be represented in Fischer (linear) or Haworth (ring) projections. It exists in two ring forms: α-d-glucose and β-d-glucose, which are repeating units in starch/glycogen and cellulose, respectively.
Glycosidic Bonds and Disaccharides
Disaccharides are formed by glycosidic bonds between two monosaccharides, with α or β linkages depending on the glucose form. Examples include maltose (α) and lactose (β).
Storage Polysaccharides
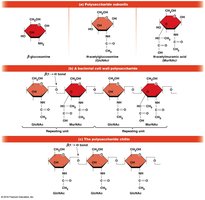
Starch (plants) and glycogen (animals, bacteria) are storage polysaccharides composed of α-d-glucose units linked by α(1→4) glycosidic bonds, with occasional α(1→6) bonds for branching.
Structural Polysaccharides
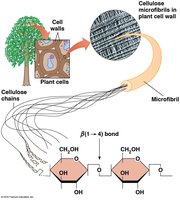
Cellulose is the primary structural polysaccharide in plant cell walls, composed of β-d-glucose units linked by β(1→4) bonds, forming rigid linear rods that aggregate into microfibrils.
Other Structural Polysaccharides
Chitin is a structural polysaccharide found in insect exoskeletons, crustacean shells, and bacterial cell walls. The PS part of LPS in gram-negative bacteria is also a polysaccharide.
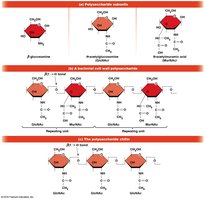
Polysaccharide Structure and Glycosidic Bonds
The type of glycosidic bond (α or β) determines the structure and function of polysaccharides. α linkages form loose helices (starch, glycogen), while β linkages form rigid rods (cellulose).
Lipids
General Properties and Functions
Lipids are hydrophobic macromolecules not formed by linear polymerization. They function in energy storage, membrane structure, and signaling. Some are amphipathic, possessing both polar and nonpolar regions.
Fatty Acids: Structure and Function
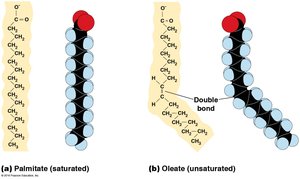
Fatty acids are long, unbranched hydrocarbon chains with a carboxyl group at one end. They are building blocks for other lipids and can be saturated (no double bonds) or unsaturated (one or more double bonds).
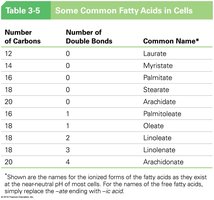
Number of Carbons | Number of Double Bonds | Common Name |
|---|---|---|
12 | 0 | Laurate |
14 | 0 | Myristate |
16 | 0 | Palmitate |
18 | 0 | Stearate |
20 | 0 | Arachidate |
16 | 1 | Palmitoleate |
18 | 1 | Oleate |
18 | 2 | Linoleate |
18 | 3 | Linolenate |
20 | 4 | Arachidonate |
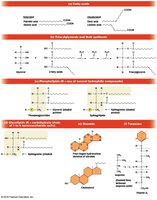
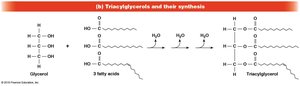
Triacylglycerols: Storage Lipids
Triacylglycerols are storage lipids formed by esterification of three fatty acids to glycerol. Saturated triacylglycerols are solid at room temperature (fats), while unsaturated ones are liquid (oils).
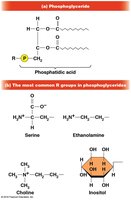
Phospholipids: Membrane Structure
Phospholipids are amphipathic molecules crucial for membrane structure, consisting of a glycerol backbone, two fatty acids, a phosphate group, and a polar head group.
Glycolipids: Specialized Membrane Components
Glycolipids contain a carbohydrate group instead of a phosphate and are often derivatives of sphingosine or glycerol. They are found mainly in the outer monolayer of the plasma membrane.
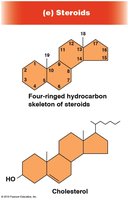
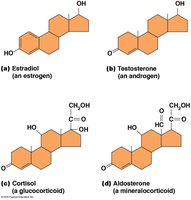
Steroids: Structure and Functions
Steroids are lipids with a four-ringed hydrocarbon skeleton. Cholesterol is the precursor for other steroids, which act as signaling molecules (hormones) and are mostly non-polar.
General Trends in Biological Polymers
The order and bonding of monomers in biological polymers determine their three-dimensional structure and function. Each class of polymer (nucleic acids, polysaccharides, lipids) exhibits unique structural and functional properties based on their monomer composition and linkage types.
