 Back
BackChapter 1 Macromolecules of the Cell: Structure and Function of Proteins and Nucleic Acids
Study Guide - Smart Notes

The Macromolecules of the Cell
Overview of Biological Macromolecules
Cells are composed of a diverse array of macromolecules, primarily synthesized from a limited set of small molecules. These macromolecules include proteins, nucleic acids, polysaccharides, and lipids, each with distinct monomeric units and biological roles.
Proteins: Polymers of amino acids, responsible for catalysis, structure, transport, signaling, and defense.
Nucleic Acids: Polymers of nucleotides, essential for storage, transmission, and expression of genetic information.
Polysaccharides: Polymers of sugars, involved in energy storage and structural support.
Lipids: Diverse group, not true polymers, but important for membrane structure and energy storage.
Kind of Molecule | Number Present | Names of Molecules | Role in Cell |
|---|---|---|---|
Amino acids | 20 | See list in Table 3-2 | Monomeric units of all proteins |
Asymmetric bases | 5 | Adenine, Guanine, Cytosine, Thymine, Uracil | Components of nucleic acids |
Sugars | varies | Ribose, Deoxyribose, Glucose | Components of RNA, DNA, energy metabolism |
Lipids | varies | Fatty acids, Cholesterol | Components of membranes, energy storage |

Proteins: Structure and Function
Diversity of Protein Function
Proteins are the most functionally diverse macromolecules in the cell, performing a wide range of tasks:
Enzymes: Catalyze biochemical reactions.
Structural proteins: Provide support and shape (e.g., collagen, keratin).
Motility proteins: Enable movement (e.g., actin, myosin).
Regulatory proteins: Control cellular processes (e.g., transcription factors).
Transport proteins: Move substances across membranes (e.g., hemoglobin, ion channels).
Signaling proteins: Mediate communication (e.g., hormones, growth factors).
Receptor proteins: Detect and respond to signals.
Defensive proteins: Protect against pathogens (e.g., antibodies).
Storage proteins: Store amino acids or ions (e.g., ferritin).
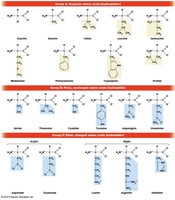
Amino Acids: The Monomers of Proteins
Proteins are linear polymers of 20 standard amino acids, each with a common structure: a central (α) carbon, an amino group, a carboxyl group, a hydrogen atom, and a variable R group (side chain).
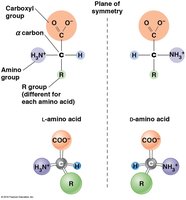
L- and D-isomers: Most amino acids exist as L- and D-isomers, but only L-amino acids are incorporated into proteins. Glycine is unique in lacking chirality and thus does not have separate L and D forms.
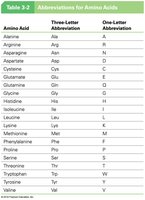
Amino Acid | Three-Letter Abbreviation | One-Letter Abbreviation |
|---|---|---|
Alanine | Ala | A |
Arginine | Arg | R |
Asparagine | Asn | N |
Aspartic acid | Asp | D |
Cysteine | Cys | C |
Glutamine | Gln | Q |
Glutamic acid | Glu | E |
Glycine | Gly | G |
Histidine | His | H |
Isoleucine | Ile | I |
Leucine | Leu | L |
Lysine | Lys | K |
Methionine | Met | M |
Phenylalanine | Phe | F |
Proline | Pro | P |
Serine | Ser | S |
Threonine | Thr | T |
Tryptophan | Trp | W |
Tyrosine | Tyr | Y |
Valine | Val | V |
Peptide Bond Formation and Protein Structure
Amino acids are joined by peptide bonds through dehydration (condensation) reactions, forming polypeptides with directionality (N-terminus to C-terminus).
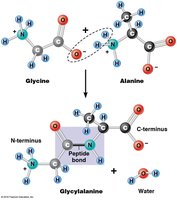
Polypeptide: Linear chain of amino acids.
Protein: A functional molecule, which may consist of one or more polypeptides.
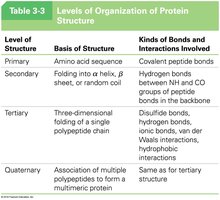
Levels of Protein Structure
Proteins exhibit hierarchical structural organization, each level stabilized by specific bonds and interactions.
Level of Structure | Basis of Structure | Kinds of Bonds and Interactions Involved |
|---|---|---|
Primary | Amino acid sequence | Covalent peptide bonds |
Secondary | Folding into α helix, β sheet, or random coil | Hydrogen bonds between NH and CO groups of peptide bonds in the backbone |
Tertiary | Three-dimensional folding of a single polypeptide chain | Disulfide bonds, hydrogen bonds, ionic bonds, van der Waals interactions, hydrophobic interactions |
Quaternary | Association of multiple polypeptides to form a multimeric protein | Same as for tertiary structure |
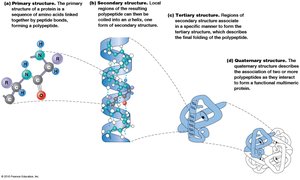
Primary Structure
The primary structure is the unique sequence of amino acids in a polypeptide, determined by the gene encoding the protein. This sequence dictates all higher levels of structure.
Secondary Structure
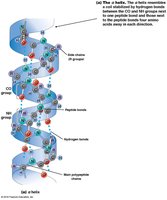
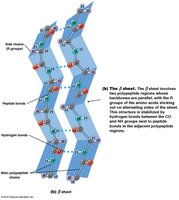
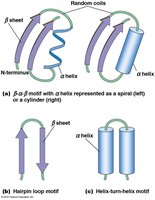
Secondary structure refers to local folding patterns stabilized by hydrogen bonds between backbone atoms. The two main types are:
α Helix: A right-handed coil with hydrogen bonds between every fourth amino acid.
β Sheet: Extended strands connected by hydrogen bonds, can be parallel or antiparallel.
Common motifs include β-α-β motifs, hairpin loops, and helix-turn-helix motifs.
Tertiary Structure
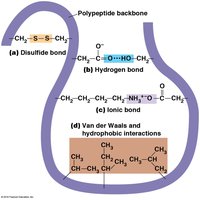
Tertiary structure is the overall three-dimensional shape of a single polypeptide, stabilized by interactions among R groups (side chains). These include hydrophobic interactions, hydrogen bonds, ionic bonds, van der Waals forces, and disulfide bridges.
Quaternary Structure
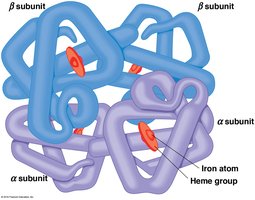
Quaternary structure arises when two or more polypeptide chains (subunits) associate to form a functional protein complex. The arrangement and interaction of these subunits are critical for protein function.
Fibrous and Globular Proteins
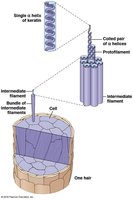
Fibrous proteins: Extended, repetitive secondary structure (e.g., silk fibroin, keratin in hair).
Globular proteins: Compact, complex tertiary structure (e.g., enzymes, hemoglobin).
Nucleic Acids: Structure and Function
Nucleic Acid Components
Nucleic acids are polymers of nucleotides, each composed of a phosphate group, a five-carbon sugar (ribose or deoxyribose), and a nitrogenous base (purine or pyrimidine).
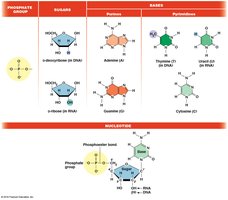
DNA: Deoxyribonucleic acid, stores genetic information.
RNA: Ribonucleic acid, involved in gene expression and regulation.
Nucleotide Nomenclature
Nucleotides are named based on their base, sugar, and number of phosphate groups (e.g., AMP, ADP, ATP).

Bases | RNA Nucleotide | DNA Nucleotide |
|---|---|---|
Adenine | Adenosine monophosphate (AMP) | Deoxyadenosine monophosphate (dAMP) |
Guanine | Guanosine monophosphate (GMP) | Deoxyguanosine monophosphate (dGMP) |
Cytosine | Cytidine monophosphate (CMP) | Deoxycytidine monophosphate (dCMP) |
Uracil | Uridine monophosphate (UMP) | — |
Thymine | — | Deoxythymidine monophosphate (dTMP) |
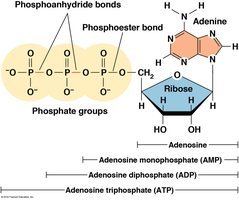
Polynucleotide Structure
Nucleotides are joined by 3',5'-phosphodiester bonds, forming a sugar-phosphate backbone with directionality (5' to 3').
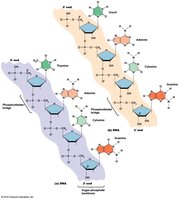
Base Pairing and Double Helix
DNA is typically double-stranded, with complementary base pairing (A-T, G-C) and antiparallel strands. RNA is usually single-stranded but can form local double-stranded regions via intramolecular base pairing.
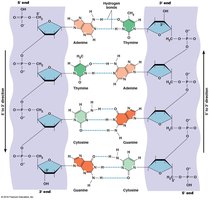
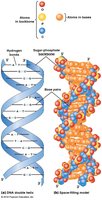
Hydrogen bonds: Two between A and T (or A and U in RNA), three between G and C.
Antiparallel orientation: The two DNA strands run in opposite directions (5' to 3' and 3' to 5').
Nucleic Acid Synthesis
Nucleic acids are synthesized using a template strand, ensuring correct base pairing and sequence fidelity. DNA replication and transcription rely on complementary relationships between purines and pyrimidines.
