8. Thermochemistry
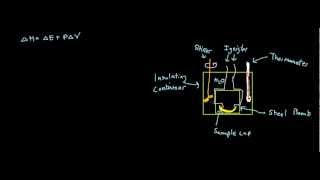
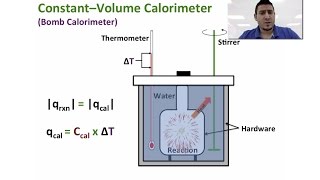
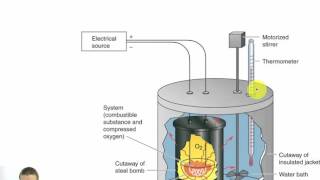
Constant-Volume Calorimetry

Get help from an AI Tutor
Ask a question to get started.
Problem 73
Textbook Question
Textbook QuestionWhen 0.514 g of biphenyl (C12H10) undergoes combustion in a bomb calorimeter, the temperature rises from 25.8 °C to 29.4 °C. Find ΔErxn for the combustion of biphenyl in kJ>mol biphenyl. The heat capacity of the bomb calorimeter, determined in a separate experiment, is 5.86 kJ/°C.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
4149
views
3
rank
1
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos