19. Chemical Thermodynamics
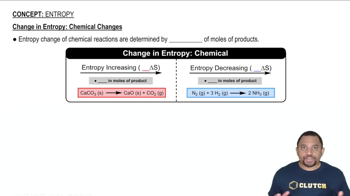
Entropy

Get help from an AI Tutor
Ask a question to get started.
Problem 42f
Textbook Question
Textbook QuestionGiven the values of ΔH rxn, ΔSrxn, and T, determine ΔSuniv and predict whether or not each reaction is spontaneous. (Assume that all reactants and products are in their standard states.) c. ΔH° rxn = +95 kJ; ΔSrxn ° = -157 J>K; T = 298 K
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
146
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos