 Back
BackAlcohols, Phenols, and Thiols: Structure, Nomenclature, and Biological Relevance
Study Guide - Smart Notes

Alcohols, Phenols, and Thiols
Introduction to Functional Groups
Alcohols, phenols, and thiols are important classes of organic compounds, each defined by a characteristic functional group. Their structures and properties are foundational in general, organic, and biological chemistry, with significant roles in health, industry, and nature.
Alcohols contain a hydroxyl group (–OH) attached to a saturated carbon atom.
Phenols have a hydroxyl group (–OH) directly bonded to a benzene ring.
Thiols possess a thiol group (–SH), analogous to alcohols but with sulfur replacing oxygen.
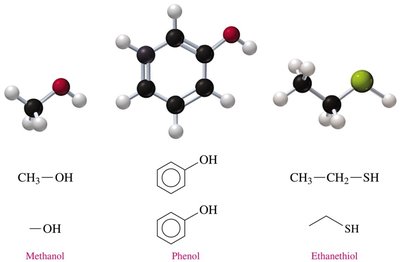
Example: Methanol (CH3OH), phenol (C6H5OH), and ethanethiol (CH3CH2SH) illustrate the basic structures of these groups.
Identifying Alcohols, Phenols, and Thiols
Recognition of these functional groups is essential for understanding their chemical behavior and nomenclature.
Alcohols: Hydroxyl group attached to an aliphatic carbon.
Phenols: Hydroxyl group attached to an aromatic ring.
Thiols: Sulfhydryl group attached to a carbon chain or ring.

Nomenclature of Alcohols, Phenols, and Thiols
Naming Alcohols
Alcohols are named using the IUPAC system by replacing the -e ending of the parent alkane with -ol. For common names, the alkyl group name is followed by 'alcohol'.
For chains with three or more carbons, number the chain to give the lowest possible number to the –OH group.
Alcohols with two or three –OH groups are named as diols or triols, respectively.
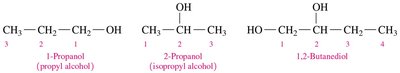
Example: 1-Propanol (CH3CH2CH2OH), 2-Propanol (isopropyl alcohol), and 1,2-Butanediol (HOCH2CH(OH)CH2CH3).
Naming Cyclic Alcohols
Cyclic alcohols are named as cycloalkanols. The ring is numbered starting from the carbon attached to the –OH group. If there are no substituents, the position number for the hydroxyl group is omitted.

Example: Cyclohexanol and 2-methylcyclopentanol.
Naming Phenols
Phenol is the IUPAC name for a benzene ring with a hydroxyl group. When additional substituents are present, the ring is numbered starting from the carbon bonded to the –OH group. The terms ortho, meta, and para are used for common names of simple phenols. Methylphenols are also called cresols.

Example: 3-chlorophenol (m-chlorophenol), 4-ethylphenol (p-ethylphenol), 3-methylphenol (m-cresol).
Naming Thiols
Thiols are named by adding 'thiol' to the alkane name of the longest carbon chain and numbering from the end nearer the –SH group. They are also known as mercaptans.
Example: Methanethiol (CH3SH), 1-propanethiol (CH3CH2CH2SH), 2-propene-1-thiol (H2C=CHCH2SH).

Applications and Biological Relevance
Alcohols in Health and Industry
Alcohols are widely used in industry and medicine. Methanol is found in solvents and paint removers but is highly toxic. Ethanol is produced by fermentation and is used in beverages, solvents, and medicines.
Methanol: Oxidized to formaldehyde in the body, causing toxicity.
Ethanol: Produced by fermentation of sugars; used in alcoholic beverages and as a solvent.

Fermentation equation:
Diols and Polyols
Compounds with multiple hydroxyl groups, such as ethylene glycol (1,2-ethanediol), are used as antifreeze and in the production of synthetic fibers. These compounds are toxic if ingested.

Phenols in Health and Nature
Phenols are found in essential oils of plants and are used in the manufacture of plastics (e.g., Bisphenol A). They also occur in spices such as nutmeg, thyme, cloves, and vanilla, contributing to flavor and aroma.

Thiols: Odor and Detection
Thiols are known for their strong odors and are found in foods such as cheese, onions, garlic, and oysters. They are also used to detect gas leaks due to their distinctive smell. Some thiols, like trans-2-butene-1-thiol, are responsible for the odor of skunk spray.


Summary Table: Key Structures and Naming
Name | Functional Group | Example Structure | IUPAC Naming Rule |
|---|---|---|---|
Alcohol | –OH (hydroxyl) | CH3CH2OH (ethanol) | Replace -e in alkane with -ol; number chain for –OH position |
Phenol | –OH on benzene ring | C6H5OH (phenol) | Number ring from –OH; use ortho/meta/para for common names |
Thiol | –SH (sulfhydryl) | CH3SH (methanethiol) | Add 'thiol' to alkane name; number chain for –SH position |
Conclusion
Alcohols, phenols, and thiols are essential functional groups in organic chemistry, with diverse roles in biological systems, industry, and daily life. Understanding their structures, nomenclature, and properties is fundamental for further study in chemistry and related fields.
