 Back
BackAlcohols, Phenols, Thiols, and Ethers: Structure, Properties, and Reactions
Study Guide - Smart Notes

Organic Compounds Containing Oxygen and Sulfur
Introduction to Alcohols, Phenols, Thiols, and Ethers
Organic compounds containing oxygen and sulfur are fundamental in both biological and industrial chemistry. This chapter focuses on alcohols, phenols, thiols, and ethers, exploring their structures, nomenclature, properties, and reactions.
Alcohols
Definition and Structure
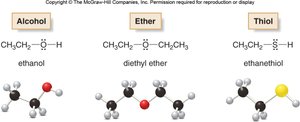
Alcohols are organic compounds in which a hydroxyl group (–OH) is bonded to a saturated, alkane-like carbon atom. The general formula is R–OH, where R is an alkyl group.
Primary (1°) alcohol: –OH group attached to a carbon bonded to one other carbon.
Secondary (2°) alcohol: –OH group attached to a carbon bonded to two other carbons.
Tertiary (3°) alcohol: –OH group attached to a carbon bonded to three other carbons.
Common Alcohols and Their Uses
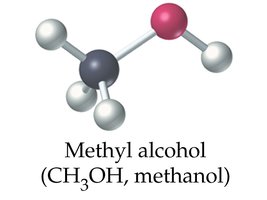
Methanol (CH3OH): Also known as wood alcohol; toxic and used as a solvent and fuel.
Ethanol (CH3CH2OH): Found in alcoholic beverages and used as a solvent and fuel.
Isopropyl alcohol ((CH3)2CHOH): Used as rubbing alcohol and antiseptic.
Ethylene glycol (HOCH2CH2OH): Used as antifreeze.
Glycerol (HOCH2CH(OH)CH2OH): Used in cosmetics and food.
Nomenclature of Alcohols
Alcohols are named by replacing the -e ending of the parent alkane with -ol. The position of the –OH group is indicated by a number. For common names, the alkyl group is named first, followed by 'alcohol'.
Number the longest carbon chain containing the –OH group from the end nearest the –OH.
Indicate the position of the –OH group and any substituents.


Physical Properties of Alcohols
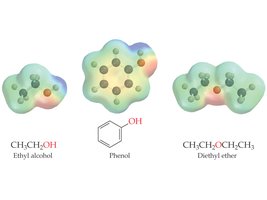
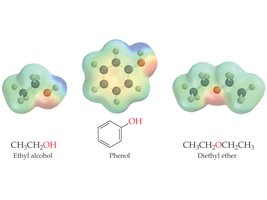
Alcohols are more polar than hydrocarbons due to the electronegative oxygen atom. They can form hydrogen bonds, resulting in higher boiling points and greater solubility in water compared to alkanes of similar molecular mass.
Hydrogen bonding increases boiling point and water solubility.
Alcohols with more –OH groups are even more soluble and have higher boiling points.
Phenols
Definition and Structure
Phenols are compounds in which a hydroxyl group is directly bonded to an aromatic benzene ring. Phenol itself (C6H5OH) is a well-known antiseptic and precursor to many important chemicals.
Phenols are more acidic than alcohols.
Substituted phenols are named by indicating the position and type of substituent.
Nomenclature of Phenols
Number the ring to give the substituents the lowest possible numbers.
Use prefixes o- (ortho), m- (meta), and p- (para) for common names.
Properties and Uses of Phenols
Phenols are used as antiseptics and disinfectants.
They are found in essential oils and flavorings (e.g., thymol, vanillin).
Ethers
Definition and Structure
Ethers are compounds in which an oxygen atom is bonded to two organic groups (R–O–R'). Ethers are less polar than alcohols and do not form hydrogen bonds with themselves.
Common ethers: diethyl ether (CH3CH2OCH2CH3), used as an anesthetic.

Nomenclature of Ethers
Common names: Name each R group alphabetically, followed by 'ether'.
IUPAC names: The larger group is the parent alkane; the smaller group plus oxygen is named as an alkoxy substituent.
Physical Properties of Ethers
Ethers have lower boiling points than alcohols of similar mass.
They are more soluble in water than alkanes but less than alcohols.
Thiols and Disulfides
Definition and Structure
Thiols (mercaptans) are sulfur analogs of alcohols, containing an –SH group. Disulfides contain an S–S bond and are formed by oxidation of thiols.
Thiols are named by adding '-thiol' to the parent hydrocarbon name.
They are known for their strong, often unpleasant odors.

Biological Importance
Disulfide bonds (S–S) are crucial for protein structure (e.g., in hair and enzymes).
Thiols are used as odorants in natural gas for leak detection.
Reactions of Alcohols
Dehydration
Alcohols can undergo dehydration (loss of water) in the presence of a strong acid and heat to form alkenes. The major product is determined by Zaitsev’s Rule: the more substituted alkene is favored.
General reaction:
Oxidation
Alcohols can be oxidized to different products depending on their classification:
Primary alcohols → Aldehydes → Carboxylic acids
Secondary alcohols → Ketones
Tertiary alcohols do not oxidize easily
Common oxidizing agents: potassium permanganate (KMnO4), dichromate (K2Cr2O7).
Acidity of Alcohols and Phenols
Alcohols are very weak acids, but phenols are much more acidic due to resonance stabilization of the phenoxide ion. Phenols can react with bases to form phenoxide salts.
Summary Table: Comparison of Alcohols, Ethers, and Thiols
Class | General Structure | Key Properties | Example |
|---|---|---|---|
Alcohol | R–OH | Hydrogen bonding, high boiling point, polar | Ethanol |
Ether | R–O–R' | Lower boiling point, less polar, no H-bonding between molecules | Diethyl ether |
Thiol | R–SH | Strong odor, less polar than alcohols | Ethanethiol |
Concept Map
This chapter connects the structure, nomenclature, properties, and reactions of alcohols, phenols, thiols, and ethers, providing a foundation for understanding their roles in chemistry and biology.
