 Back
BackAlcohols, Phenols, Thiols, and Ethers: Structure, Nomenclature, Properties, and Reactions
Study Guide - Smart Notes

Chapter 13: Alcohols, Phenols, Thiols, and Ethers
13.1 Alcohols, Phenols, and Thiols
This section introduces the structure, nomenclature, and basic properties of alcohols, phenols, and thiols, which are organic compounds containing hydroxyl or thiol functional groups.
Alcohols
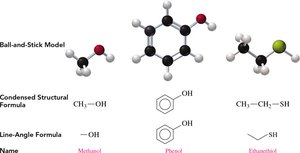
Definition: Alcohols are organic compounds containing a hydroxyl group (–OH) attached to a saturated carbon atom.
General Formula: R–OH, where R is an alkyl group.
Naming: The IUPAC name is formed by replacing the -e of the parent alkane with -ol. For chains with three or more carbons, the position of the –OH group is indicated by a number.
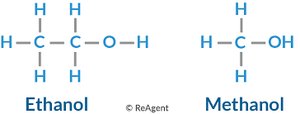
Examples: Methanol (CH3OH), Ethanol (CH3CH2OH)
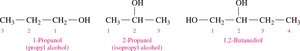
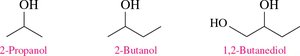
Alcohols with Multiple –OH Groups: Named as diols (two –OH) or triols (three –OH).
Cyclic Alcohols: Named as cycloalkanols. The ring is numbered from the carbon attached to the –OH group.
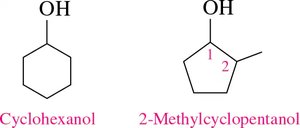
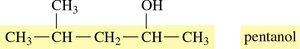
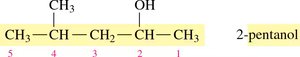
Naming Alcohols: Example
Step 1: Identify the longest carbon chain containing the –OH group and change the ending to -ol.
Step 2: Number the chain from the end nearest the –OH group.
Step 3: Name and locate substituents.
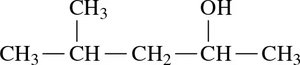
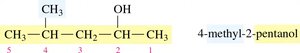
Phenols
Definition: Phenols are compounds where a hydroxyl group is directly attached to a benzene ring.
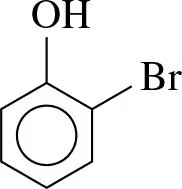
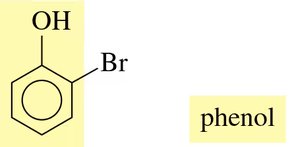
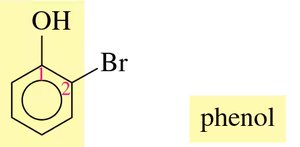
Naming: The parent compound is phenol. Substituents are numbered starting from the carbon bonded to the –OH group. The terms ortho (o-), meta (m-), and para (p-) are used for common names.
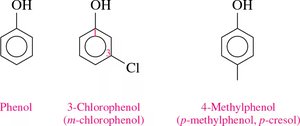
Naming Example: For a phenol with a bromine substituent, number the ring and assign the correct name (e.g., 2-bromophenol or o-bromophenol).

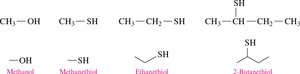
Thiols
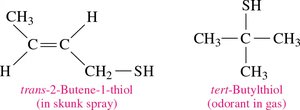
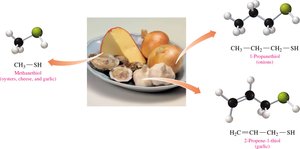
Definition: Thiols (mercaptans) are organic compounds containing a thiol group (–SH).
Properties: Often have strong, unpleasant odors; found in onions, garlic, and skunk spray.
Naming: Add thiol to the alkane name and number the chain from the end nearest the –SH group.
13.2 Ethers
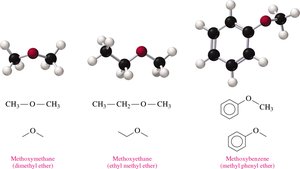
Ethers are organic compounds with an oxygen atom connected to two alkyl or aromatic groups.
General Structure: R–O–R', where R and R' are alkyl or aryl groups.
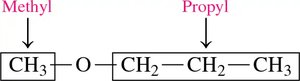
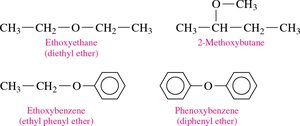
Naming (Common): List the names of the groups attached to oxygen in alphabetical order, followed by "ether" (e.g., ethyl methyl ether).
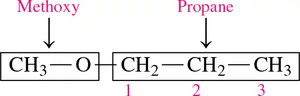
Naming (IUPAC): Name the smaller group as an alkoxy substituent on the larger alkane chain (e.g., methoxypropane).
13.3 Physical Properties of Alcohols, Phenols, and Ethers
This section discusses the classification, boiling points, and solubility of alcohols, phenols, and ethers.
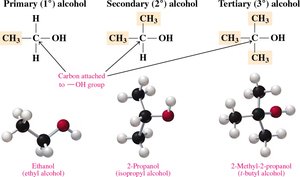
Classification of Alcohols
Primary (1°) Alcohol: The carbon with the –OH group is attached to one other carbon.
Secondary (2°) Alcohol: The carbon with the –OH group is attached to two other carbons.
Tertiary (3°) Alcohol: The carbon with the –OH group is attached to three other carbons.
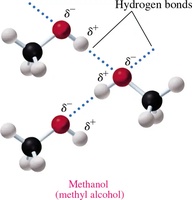
Boiling Points
Hydrogen Bonding: Alcohols can form hydrogen bonds due to the polar –OH group, resulting in higher boiling points compared to ethers and alkanes of similar molar mass.
Ethers: Cannot form hydrogen bonds with themselves, so their boiling points are similar to alkanes.
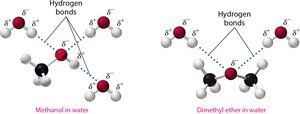
Solubility in Water
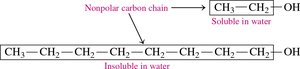
Alcohols: Small alcohols (1–3 carbons) are completely miscible in water due to hydrogen bonding. Solubility decreases as the nonpolar hydrocarbon chain increases.
Ethers: Slightly soluble in water due to the polar oxygen atom, but less so than alcohols.
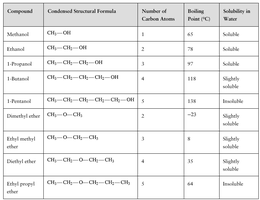
Compound | Condensed Structural Formula | Number of Carbon Atoms | Boiling Point (°C) | Solubility in Water |
|---|---|---|---|---|
Methanol | CH3OH | 1 | 65 | Soluble |
Ethanol | CH3CH2OH | 2 | 78 | Soluble |
1-Propanol | CH3CH2CH2OH | 3 | 97 | Soluble |
1-Butanol | CH3CH2CH2CH2OH | 4 | 118 | Slightly soluble |
1-Pentanol | CH3CH2CH2CH2CH2OH | 5 | 138 | Insoluble |
Dimethyl ether | CH3OCH3 | 2 | -25 | Slightly soluble |
Ethyl methyl ether | CH3OCH2CH3 | 3 | 8 | Slightly soluble |
Diethyl ether | CH3CH2OCH2CH3 | 4 | 35 | Slightly soluble |
Ethyl propyl ether | CH3CH2OCH2CH2CH3 | 5 | 64 | Insoluble |
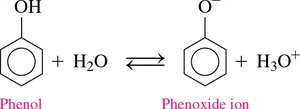
Phenol
Boiling Point: High (182°C) due to hydrogen bonding.
Solubility: Slightly soluble in water; the –OH group can form hydrogen bonds with water. Phenol is a weak acid and ionizes slightly in water.
13.4 Reactions of Alcohols and Thiols
This section covers the main chemical reactions of alcohols and thiols, including combustion, dehydration, and oxidation.
Combustion of Alcohols
Alcohols combust in oxygen to produce carbon dioxide and water.
General Equation:

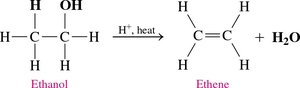
Dehydration of Alcohols
Alcohols lose a water molecule when heated with an acid catalyst (e.g., H2SO4) at high temperature (180°C), forming an alkene.
General Equation:
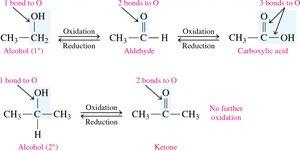
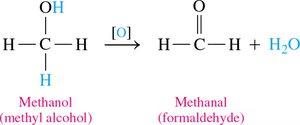
Oxidation of Alcohols
Oxidation involves the loss of hydrogen or the addition of oxygen, increasing the number of carbon–oxygen bonds.
Primary Alcohols: Oxidize to aldehydes, then to carboxylic acids.
Secondary Alcohols: Oxidize to ketones.
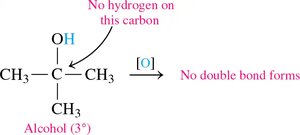
Tertiary Alcohols: Do not oxidize easily (no hydrogen on the carbon with –OH).
Additional info: The reactions of thiols often involve oxidation to disulfides, but this is not detailed in the provided material.
