 Back
BackAlcohols, Thiols, Ethers, Aldehydes, and Ketones: Structure, Naming, Properties, and Reactions
Study Guide - Smart Notes

Alcohols, Thiols, Ethers, Aldehydes, and Ketones
Functional Groups in Organic Chemistry
Organic compounds are classified based on their functional groups, which determine their chemical properties and reactivity. The main functional groups discussed here are alcohols, phenols, thiols, ethers, aldehydes, and ketones.
Alcohols: Contain a hydroxyl group (–OH) attached to a carbon atom.
Phenols: Contain a hydroxyl group bonded to a benzene ring.
Thiols: Contain a sulfhydryl group (–SH).
Ethers: Contain an oxygen atom between two carbon groups (C–O–C).
Aldehydes: Contain a carbonyl group (C=O) attached to at least one hydrogen atom.
Ketones: Contain a carbonyl group (C=O) attached to two alkyl or aromatic groups.

Naming Alcohols
Alcohols are named according to the IUPAC system by replacing the "e" in the parent alkane with "ol". Common names use the alkyl group name followed by "alcohol".
IUPAC Naming: Identify the longest carbon chain containing the –OH group, number the chain from the end nearest the –OH, and indicate the position of the –OH and any substituents.
Common Naming: Name the alkyl group attached to the –OH, followed by "alcohol".


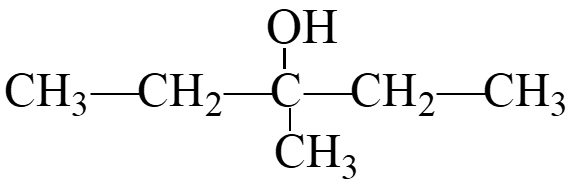
Classification of Alcohols
Alcohols are classified based on the number of alkyl groups attached to the carbon bonded to the hydroxyl group:
Primary (1°) Alcohol: The carbon with the –OH is attached to one other carbon.
Secondary (2°) Alcohol: The carbon with the –OH is attached to two other carbons.
Tertiary (3°) Alcohol: The carbon with the –OH is attached to three other carbons.
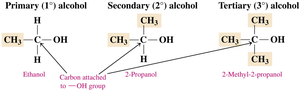
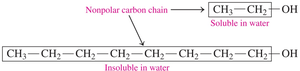
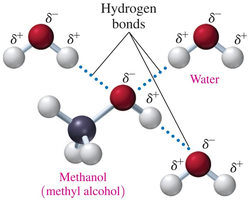
Solubility of Alcohols in Water
The solubility of alcohols in water depends on the size of the hydrocarbon chain and the presence of the polar –OH group.
Alcohols with 1–3 carbons are soluble in water due to hydrogen bonding.
Solubility decreases as the hydrocarbon chain length increases.
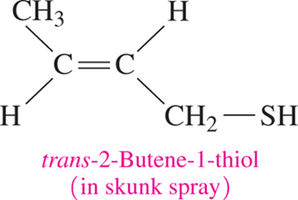
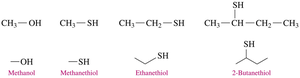
Thiols: Structure, Properties, and Naming
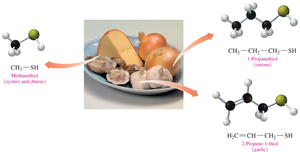
Thiols are sulfur-containing organic compounds with a sulfhydryl (–SH) group. They often have strong odors and are found in various foods and used in gas leak detection.
Naming: Add "thiol" to the alkane name of the longest chain, numbering from the end nearest the –SH group.
Properties: Strong odors, found in cheese, onions, garlic, oysters, and skunk spray.

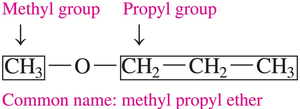
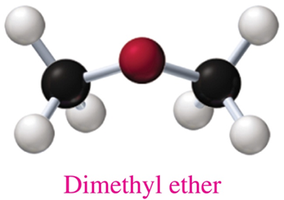
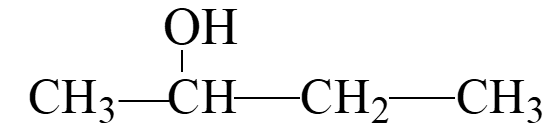
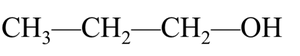
Ethers: Structure and Naming
Ethers contain an oxygen atom between two carbon groups. Most ethers are named using common names, listing the alkyl groups attached to the oxygen in alphabetical order followed by "ether".
Structure: Bent shape similar to water and alcohols.
Naming: Common names are used, e.g., "methyl propyl ether".

Solubility of Phenols
Phenols are slightly soluble in water due to their ability to form hydrogen bonds. They can react with water to produce phenoxide ions and were once used as antiseptics.
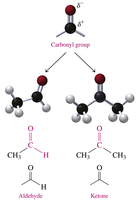
Aldehydes and Ketones: Structure and Properties
Aldehydes and ketones contain a carbonyl group (C=O), which is highly polar due to the electronegativity of oxygen. This group is responsible for their chemical reactivity and physical properties.
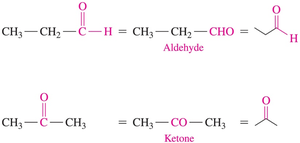
Aldehyde: Carbonyl group attached to at least one hydrogen atom.
Ketone: Carbonyl group attached to two alkyl or aromatic groups.
Naming Aldehydes
Aldehydes are named by replacing the "e" in the parent alkane with "al". The carbonyl carbon is always numbered as carbon 1. Common names are used for the first four aldehydes.

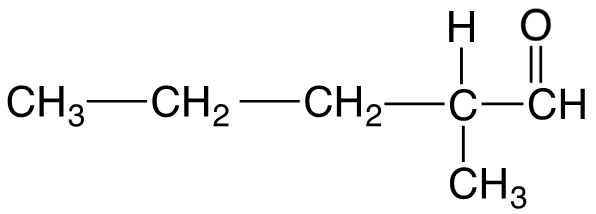
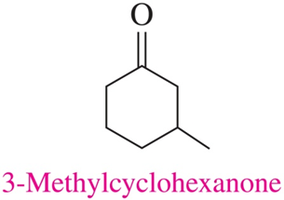
Naming Ketones
Ketones are named by replacing the "e" in the parent alkane with "one". The chain is numbered from the end nearest the carbonyl group, and substituents are named and numbered accordingly.
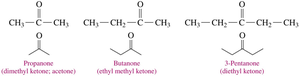
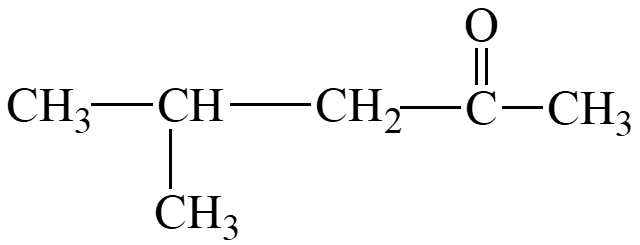
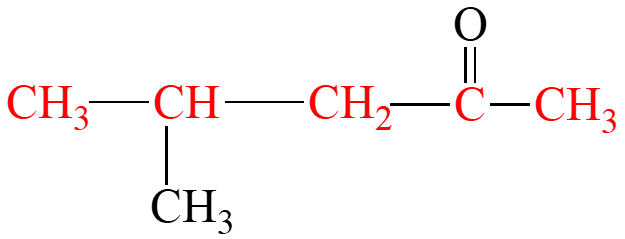
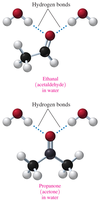
Solubility of Aldehydes and Ketones in Water
Aldehydes and ketones can form hydrogen bonds with water molecules via the carbonyl oxygen. They are very soluble in water when they have four or fewer carbons; solubility decreases with longer hydrocarbon chains.
Chemical Reactions of Alcohols
Dehydration of Alcohols
Alcohols undergo dehydration when heated with an acid catalyst, losing –H and –OH from adjacent carbon atoms to produce an alkene and water.
General equation:
Oxidation and Reduction
Oxidation increases the number of carbon–oxygen bonds, while reduction decreases them. The oxidation of alcohols depends on their classification:
Primary alcohols: Oxidized to aldehydes, which can further oxidize to carboxylic acids.
Secondary alcohols: Oxidized to ketones.
Tertiary alcohols: Do not oxidize due to lack of hydrogen on the carbon bonded to –OH.
Reduction of aldehydes and ketones (using NaBH4 or H2 with a catalyst) produces alcohols:
Aldehydes: Reduce to primary alcohols.
Ketones: Reduce to secondary alcohols.
Oxidation of thiols: Two thiol groups lose hydrogen atoms to form a disulfide.
Tests for Aldehydes and Ketones
Tollen’s Test
Tollen’s reagent (Ag+) oxidizes aldehydes but not ketones, producing metallic silver.
Benedict’s Test
Benedict’s reagent (Cu2+) reacts with aldehydes with adjacent –OH groups, forming a red precipitate of Cu2O. Simple aldehydes and ketones give a negative test.
Summary Table: Functional Groups and Their Properties
Functional Group | Structure | Naming | Solubility | Reactivity |
|---|---|---|---|---|
Alcohol | R–OH | Replace "e" with "ol" | Soluble (1–3 C) | Dehydration, oxidation |
Phenol | Ar–OH | Phenol | Slightly soluble | Forms phenoxide ions |
Thiols | R–SH | Add "thiol" | Varies | Oxidation to disulfide |
Ether | R–O–R' | Common names | Low solubility | Relatively inert |
Aldehyde | R–CHO | Replace "e" with "al" | Soluble (≤4 C) | Oxidation, reduction |
Ketone | R–CO–R' | Replace "e" with "one" | Soluble (≤4 C) | Reduction |
Additional info: This guide covers Timberlake Chapter 12, sections 12.1–12.4, focusing on the structure, naming, properties, and reactions of alcohols, thiols, ethers, aldehydes, and ketones. It includes relevant images and examples to reinforce key concepts for exam preparation.
