 Back
BackLecture 6: Alcohols
Study Guide - Smart Notes

Alcohols, Thiols, Ethers, Aldehydes, and Ketones
Functional Groups Overview
Organic compounds are classified by their functional groups, which determine their chemical properties and reactivity. The main functional groups discussed here are alcohols, phenols, thiols, ethers, aldehydes, and ketones.
Alcohols: Contain a hydroxyl group (–OH) attached to a carbon atom.
Phenols: Contain a hydroxyl group bonded to a benzene ring.
Thiols: Contain a sulfhydryl group (–SH).
Ethers: Contain an oxygen atom between two carbon groups (C–O–C).
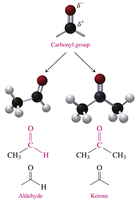
Aldehydes: Contain a carbonyl group (C=O) attached to at least one hydrogen atom.
Ketones: Contain a carbonyl group (C=O) attached to two alkyl or aromatic groups.
Naming Alcohols
Alcohols are named according to IUPAC rules by replacing the “e” in the parent alkane with “ol.” The chain is numbered from the end nearest the –OH group, and substituents are named and numbered accordingly.
IUPAC Name: Replace “e” in alkane with “ol” (e.g., hexane → hexanol).
Common Name: Name of the alkyl group followed by “alcohol” (e.g., methyl alcohol).
Example: 2-methyl-3-hexanol (hydroxyl group on carbon 3, methyl group on carbon 2).
Naming Thiols
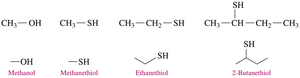
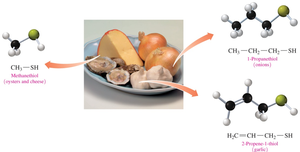
Thiols are named by adding “thiol” to the alkane name of the longest carbon chain and numbering from the end nearest the –SH group.
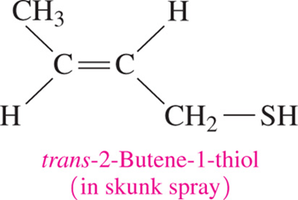
Example: Methanethiol, Ethanethiol, 2-Butanethiol.

Naming Ethers
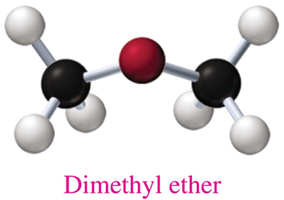
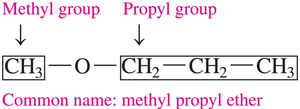
Ethers are commonly named by listing the alkyl or aromatic groups attached to the oxygen atom in alphabetical order, followed by “ether.”
Example: Dimethyl ether, methyl propyl ether.
Classification of Alcohols
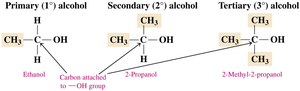
Alcohols are classified based on the number of alkyl groups attached to the carbon bonded to the hydroxyl group:
Primary (1°): One alkyl group attached.
Secondary (2°): Two alkyl groups attached.
Tertiary (3°): Three alkyl groups attached.
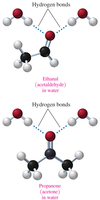
Solubility of Alcohols in Water
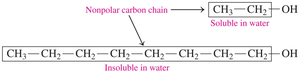
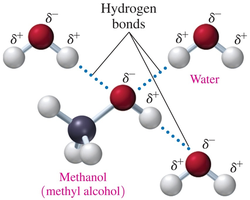
Alcohols are soluble in water due to their polar –OH group, which forms hydrogen bonds with water. Solubility decreases as the hydrocarbon chain length increases.
Alcohols with 1–3 carbons: Soluble in water.
Alcohols with longer chains: Decreased solubility.
Solubility of Phenols
Phenols are slightly soluble in water due to their ability to form hydrogen bonds. They can react with water to produce phenoxide ions and were once used as antiseptics.
Aldehydes and Ketones: The Carbonyl Group
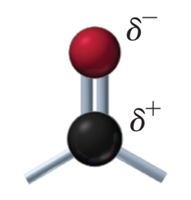
The carbonyl group (C=O) is a polar functional group with a partial positive charge on carbon and a partial negative charge on oxygen. It is found in both aldehydes and ketones.
Naming Aldehydes
Aldehydes are named by replacing the “e” in the parent alkane with “al.” The carbonyl carbon is always numbered as carbon 1.
Example: Pentanal, 2-methylpentanal.
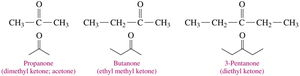
Naming Ketones
Ketones are named by replacing the “e” in the parent alkane with “one.” The chain is numbered from the end nearest the carbonyl group, and substituents are named and numbered accordingly.
Example: 2-pentanone, 4-methyl-2-pentanone.
Solubility of Aldehydes and Ketones in Water
Aldehydes and ketones form hydrogen bonds with water molecules via the carbonyl oxygen. They are very soluble when they have four or fewer carbons; solubility decreases with longer hydrocarbon chains.
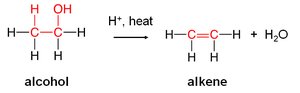
Dehydration of Alcohols
Alcohols undergo dehydration when heated with an acid catalyst, resulting in the loss of –H and –OH from adjacent carbon atoms to produce an alkene and water.
Equation:
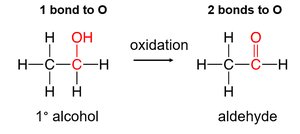
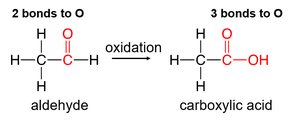
Oxidation and Reduction of Alcohols, Aldehydes, and Ketones
Oxidation increases the number of carbon–oxygen bonds, while reduction decreases them. The outcome depends on the type of alcohol:
Primary alcohols: Oxidized to aldehydes, then to carboxylic acids.
Secondary alcohols: Oxidized to ketones.
Tertiary alcohols: Do not oxidize (no hydrogen atom on the carbon bonded to –OH).
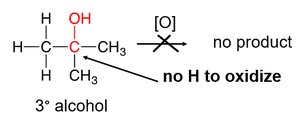
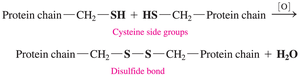
Oxidation of Thiols
Thiols undergo oxidation to form disulfide bonds, which are important in protein structure.
Equation:
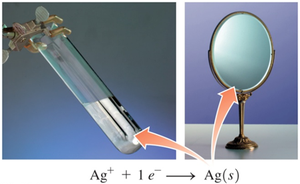
Tollen’s Test for Aldehydes
Tollen’s reagent (Ag+) oxidizes aldehydes but not ketones. The silver ion is reduced to metallic silver, forming a mirror-like deposit.
Equation:
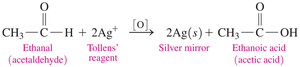
Benedict’s Test for Aldehydes
Benedict’s reagent (Cu2+) reacts with aldehydes that have an adjacent –OH group. A positive test forms a red solid of Cu2O, indicating the presence of certain sugars and aldehydes.
Reduction of Aldehydes and Ketones
Aldehydes and ketones are reduced by sodium borohydride (NaBH4) or hydrogen (H2) in the presence of a catalyst. Aldehydes reduce to primary alcohols, ketones to secondary alcohols.
Equation for aldehyde reduction:
Equation for ketone reduction:
Summary Table: Classification and Reactions of Alcohols, Thiols, Ethers, Aldehydes, and Ketones
Functional Group | General Formula | Naming Suffix | Key Reaction |
|---|---|---|---|
Alcohol | R–OH | -ol | Oxidation to aldehyde/ketone |
Thiols | R–SH | -thiol | Oxidation to disulfide |
Ether | R–O–R' | ether | Relatively inert |
Aldehyde | R–CHO | -al | Oxidation to carboxylic acid |
Ketone | R2C=O | -one | Reduction to alcohol |
Additional info: These notes expand on the brief lecture points to provide a comprehensive overview of the structure, naming, classification, solubility, and reactions of alcohols, thiols, ethers, aldehydes, and ketones, suitable for exam preparation in a general, organic, and biological chemistry course.
