 Back
BackAldehydes and Ketones: Structure, Properties, and Reactions
Study Guide - Smart Notes

Chapter 16: Aldehydes and Ketones
Carbonyl Group Structure
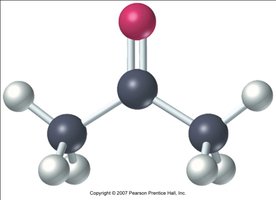
The carbonyl group is a functional group consisting of a carbon atom double-bonded to an oxygen atom (C=O). This group is central to the chemistry of aldehydes and ketones, imparting unique structural and chemical properties.
Bond Angles: The carbonyl group forms approximately 120° bond angles, resulting in a planar triangular geometry.
Polarity: The oxygen atom is more electronegative than carbon, creating a partial negative charge (δ-) on oxygen and a partial positive charge (δ+) on the carbonyl carbon.


Types of Carbonyl Compounds
Carbonyl groups are found in several important organic families. The structure and bonding of the carbonyl group determine the reactivity and properties of these compounds.
Aldehydes: Carbonyl group bonded to at least one hydrogen atom (R-CHO).
Ketones: Carbonyl group bonded to two carbon atoms (R-CO-R').
Carboxylic Acids, Esters, Amides: Other carbonyl-containing families with additional functional groups.
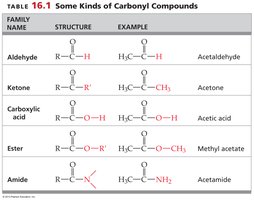
Family Name | Structure | Example |
|---|---|---|
Aldehyde | R-CHO | Acetaldehyde |
Ketone | R-CO-R' | Acetone |
Carboxylic acid | R-COOH | Acetic acid |
Ester | R-COO-R' | Methyl acetate |
Amide | R-CONH2 | Acetamide |
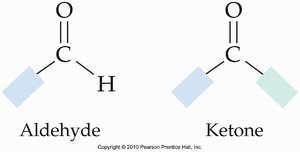
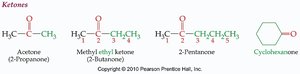
Aldehydes vs. Ketones
Aldehydes and ketones are distinguished by the groups attached to the carbonyl carbon.
Aldehyde: Carbonyl carbon is bonded to a hydrogen and an R group.
Ketone: Carbonyl carbon is bonded to two R groups (which may be the same or different).

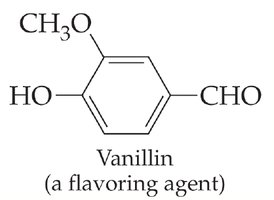
Common Examples in Biology
Aldehydes and ketones are prevalent in biological systems and industrial applications. For example, vanillin is an aldehyde used as a flavoring agent.
Vanillin: Contains an aldehyde group attached to a benzene ring.
Naming Aldehydes and Ketones
The nomenclature of aldehydes and ketones follows IUPAC rules, with specific suffixes for each class.
Aldehydes: Suffix "-al" (e.g., ethanal, propanal).
Ketones: Suffix "-one" (e.g., propanone, butanone).
Some simple compounds are known by common names (e.g., formaldehyde, acetone).
jbg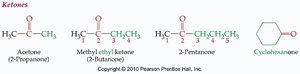
Physical Properties of Aldehydes and Ketones
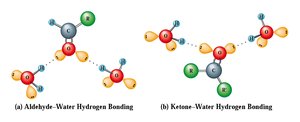
Aldehydes and ketones exhibit moderate polarity due to the carbonyl group, affecting their boiling points and solubility.
Boiling Points: Higher than alkanes of similar molecular weight due to dipole-dipole interactions, but lower than alcohols.
Water Solubility: Limited, but small aldehydes and ketones are somewhat soluble due to hydrogen bonding with water.
Odor: Many have distinctive odors, some pleasant (e.g., vanillin), others unpleasant.


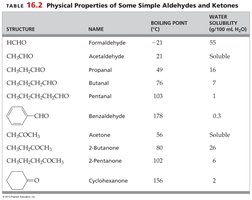
Structure | Name | Boiling Point (°C) | Water Solubility (g/100 mL H2O) |
|---|---|---|---|
HCHO | Formaldehyde | -21 | 55 |
CH3CHO | Acetaldehyde | 21 | Soluble |
CH3CH2CHO | Propanal | 49 | 16 |
CH3CH2CH2CHO | Butanal | 75 | 7 |
CH3CH2CH2CH2CHO | Pentanal | 103 | 1 |
C6H5CHO | Benzaldehyde | 178 | 0.3 |
CH3COCH3 | Acetone | 56 | Soluble |
CH3COCH2CH3 | 2-Butanone | 80 | 26 |
CH3COCH2CH2CH3 | 2-Pentanone | 102 | 6 |
C6H10O | Cyclohexanone | 156 | 2 |
Important Common Aldehydes and Ketones
Several aldehydes and ketones are widely used in industry and biology.
Formaldehyde (HCHO): Used as a preservative and in plastics; toxic due to protein crosslinking.
Acetone (CH3COCH3): Industrial solvent, nail polish remover, produced in human metabolism.
Chemical Reactions of Aldehydes and Ketones
Oxidation
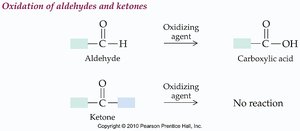
Aldehydes are easily oxidized to carboxylic acids, while ketones are generally resistant to oxidation.
Oxidation of Aldehydes:
Oxidation of Ketones: No reaction under mild conditions.
Reduction
Reduction is the reverse of oxidation. Aldehydes and ketones can be reduced to alcohols.
Aldehyde: Reduced to a primary alcohol.
Ketone: Reduced to a secondary alcohol.
General Reaction:


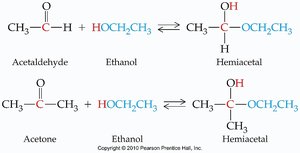
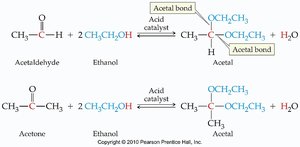
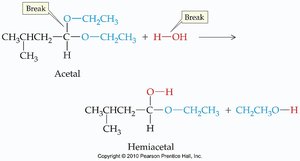
Addition of Alcohols: Hemiacetals and Acetals
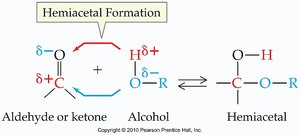
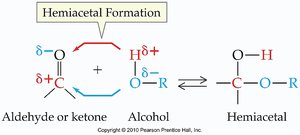
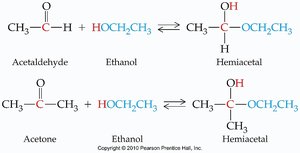
Aldehydes and ketones react with alcohols to form hemiacetals and acetals. These reactions are important in carbohydrate chemistry and organic synthesis.
Hemiacetal: Contains both an -OH and an -OR group on the same carbon.
Acetal: Contains two -OR groups on the same carbon; more stable than hemiacetals.
Formation: Aldehyde or ketone + alcohol → hemiacetal; hemiacetal + alcohol (acid catalysis) → acetal.


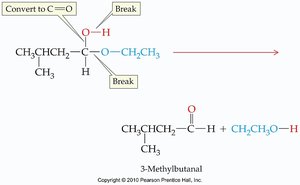
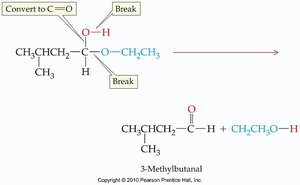
Acetal Hydrolysis
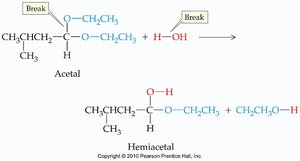
Acetals can be hydrolyzed (broken apart) by water, especially under acidic conditions. This reverses the acetal formation reaction.
Hydrolysis: Acetal + water → aldehyde/ketone + alcohols
Equilibrium: Acetal formation and hydrolysis are equilibrium reactions; conditions can shift the balance.

Summary Table: Aldehydes vs. Ketones
Property | Aldehyde | Ketone |
|---|---|---|
Structure | R-CHO | R-CO-R' |
Oxidation | To carboxylic acid | No reaction |
Reduction | To primary alcohol | To secondary alcohol |
Common Example | Formaldehyde | Acetone |
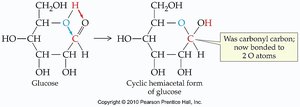
Additional info: Cyclic hemiacetals are especially important in carbohydrate chemistry, as seen in glucose. Acetals are used as protecting groups in organic synthesis due to their stability under neutral and basic conditions.
