 Back
BackAlkanes: Structure, Nomenclature, Isomerism, and Properties
Study Guide - Smart Notes

Alkanes: Structure and Nomenclature
Definition and General Formula
Alkanes are hydrocarbons containing only single bonds between carbon atoms. Their general formula is , where n is the number of carbon atoms. Alkanes are the simplest family of organic compounds and serve as the foundation for understanding more complex molecules.
Hydrocarbon: A molecule composed only of carbon and hydrogen atoms.
Alkane: A hydrocarbon with only single (sigma) bonds.
General formula:
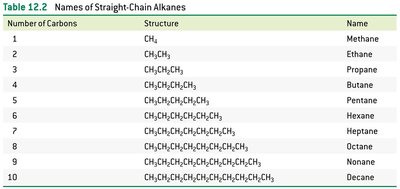
Naming Alkanes (IUPAC System)
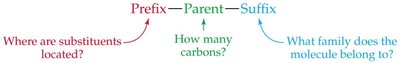
The International Union of Pure and Applied Chemistry (IUPAC) has established rules for naming alkanes. Each alkane name consists of three parts: prefix (location of substituents), parent (number of carbons in the longest chain), and suffix (family, e.g., -ane for alkanes).
Prefix: Indicates the position and identity of substituents.
Parent: Specifies the number of carbons in the longest continuous chain.
Suffix: Identifies the family (e.g., -ane for alkanes).
Straight-Chain and Branched Alkanes
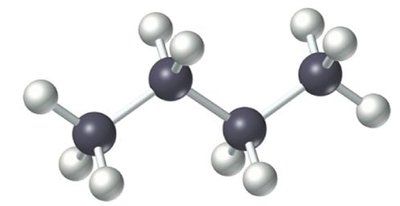
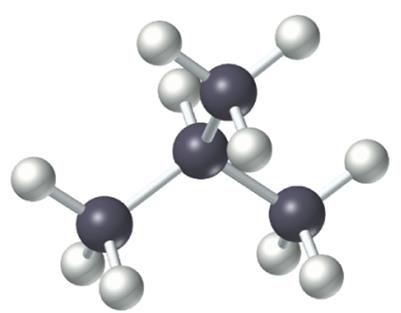
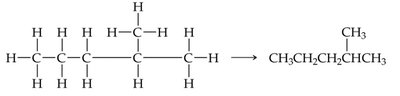
Straight-chain alkanes have all carbon atoms connected in a single, unbranched row. Branched-chain alkanes contain one or more branches off the main chain. Both types share the same molecular formula but differ in structure and properties.
Straight-chain: All carbons in a row.
Branched-chain: At least one carbon is connected to more than two other carbons.

Isomerism in Alkanes
Isomers are molecules with the same molecular formula but different structures. Structural (constitutional) isomers differ in the connectivity of their atoms, while conformers differ only in the spatial arrangement due to rotation around single bonds.
Structural isomers: Different connections among atoms; different names and properties.
Conformers: Same connectivity; differ by rotation around single bonds; same name.


Alkyl Groups and Substituents
An alkyl group is formed by removing a hydrogen atom from an alkane. Common alkyl groups include methyl (–CH3), ethyl (–CH2CH3), propyl, isopropyl, butyl, and others. Substituents are named and numbered according to their position on the main chain.
Methyl group: –CH3
Ethyl group: –CH2CH3
Isopropyl group: –CH(CH3)2
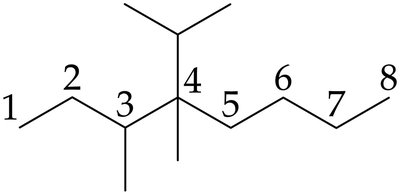
Rules for Naming Branched Alkanes
When naming branched alkanes, follow these steps:
Identify the longest continuous carbon chain (parent).
Number the chain from the end nearest the first branch.
Identify and number each substituent according to its attachment point.
Write the name as a single word, using hyphens and commas as needed.
List substituents alphabetically; use prefixes (di-, tri-, etc.) for identical groups, but do not use them for alphabetizing.




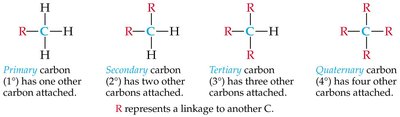
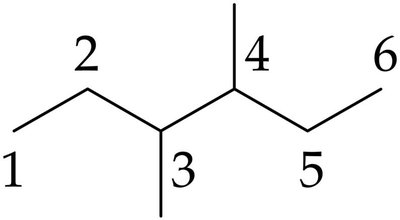
Primary, Secondary, Tertiary, and Quaternary Carbons
Carbons in alkanes are classified by the number of other carbons attached:
Primary (1°): Attached to one other carbon.
Secondary (2°): Attached to two other carbons.
Tertiary (3°): Attached to three other carbons.
Quaternary (4°): Attached to four other carbons.
Drawing Organic Structures
Types of Structural Representations
Organic molecules can be represented in several ways:
Lewis structure: Shows all atoms and bonds.
Complete structural formula: Shows all bonds explicitly.
Condensed formula: Groups atoms together, omitting some bonds.
Line structure (line-angle): Uses lines for bonds; carbon and hydrogen atoms are implied.
Ball-and-stick model: 3D representation of atoms and bonds.
Space-filling model: Shows the relative sizes of atoms.

Guidelines for Line Structures
Each line represents a carbon–carbon bond.
Ends and vertices represent carbon atoms.
Non-carbon/non-hydrogen atoms must be shown explicitly.
Neutral carbon forms four bonds; missing bonds are assumed to be C–H.

Isomerism: Structural and Conformational
Constitutional (Structural) Isomers
Constitutional isomers have the same molecular formula but different connectivity. They have different names, structures, and physical properties.

Conformers
Conformers are different spatial arrangements of the same molecule due to rotation around single bonds. No bonds are broken or reformed, and the molecules have the same name.

Cycloalkanes: Structure and Nomenclature
Definition and General Formula
Cycloalkanes are alkanes in which the carbon atoms form a ring. The general formula is , reflecting the loss of two hydrogens to form the ring.
Cyclopropane: Three-membered ring
Cyclobutane: Four-membered ring
Cyclopentane: Five-membered ring
Cyclohexane: Six-membered ring

Naming Cycloalkanes
Use "cyclo" as a prefix to the parent alkane name. Number substituents to give the lowest possible numbers, starting with the group of alphabetical priority.



Physical Properties of Alkanes
Intermolecular Forces
Alkanes are nonpolar and interact via weak London dispersion forces (a type of van der Waals force). These forces increase with molecular size and surface area.
Nonpolar: Insoluble in water, soluble in nonpolar solvents.
Low density: Less dense than water.
Flammable: Combust readily in oxygen.
Boiling point: Increases with chain length and molecular weight.
Chemical Reactions of Alkanes
Combustion
Alkanes react with oxygen in a combustion reaction, producing carbon dioxide, water, and heat.
General equation:
Application: Fuels such as methane, propane, and octane.
Halogenation
Alkanes react with halogens (Cl2, Br2) in the presence of heat or light to form alkyl halides. This is a free radical reaction.
General formula:
Alkyl halide: Compound where a halogen replaces a hydrogen atom.
Summary Table: Common Abbreviations in Organic Chemistry
Symbol | Meaning |
|---|---|
R | Residue or rest of molecule; generic carbon group |
X | Halogen atom (F, Cl, Br, I) |
Ar | Aromatic group |
Ph | Phenyl group (–C6H5) |

Additional info:
Alkanes are foundational for understanding organic chemistry and biochemistry.
Knowledge of isomerism, nomenclature, and physical properties is essential for further study of organic compounds.
