 Back
BackAlkenes, Alkynes, and Aromatic Compounds: Structure, Nomenclature, and Reactions
Study Guide - Smart Notes

Alkenes, Alkynes, and Aromatic Compounds
Introduction to Alkenes, Alkynes, and Aromatic Compounds
Alkenes, alkynes, and aromatic compounds are classes of hydrocarbons distinguished by the presence of double, triple, or delocalized bonds, respectively. Their unique bonding leads to distinct chemical and physical properties, as well as specific rules for nomenclature and reactivity.
Alkenes & Alkynes
Definitions and Classification
Saturated hydrocarbons: Molecules whose carbon atoms bond to the maximum number of hydrogen atoms (alkanes, only C–C single bonds).
Unsaturated hydrocarbons: Molecules containing carbon–carbon multiple bonds (alkenes with C=C double bonds, alkynes with C≡C triple bonds).
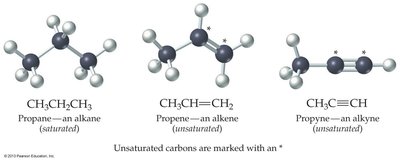
Bonding and Molecular Geometry
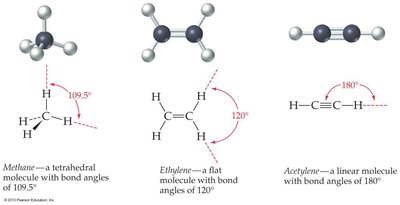
Alkanes: Tetrahedral geometry, bond angles of 109.5°.
Alkenes: Planar geometry, bond angles of 120° due to sp2 hybridization.
Alkynes: Linear geometry, bond angles of 180° due to sp hybridization.
Naming Alkenes and Alkynes
Nomenclature for alkenes and alkynes follows similar rules to alkanes, with modifications to indicate the presence and position of multiple bonds.
Change the suffix: -ane (alkane), -ene (alkene), -yne (alkyne).
Identify the longest chain containing the multiple bond as the parent chain.
Number the chain from the end nearest the multiple bond; if a branch point is present, assign the lowest possible numbers to both the multiple bond and the branch.
For cycloalkenes, the ring must contain the double bond.
Indicate the position of the multiple bond by the number of the first carbon involved.
List substituents alphabetically, using commas between numbers and hyphens between numbers and words.
Shapes and Isomerism in Alkenes and Alkynes
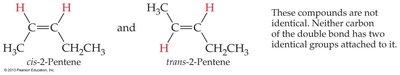
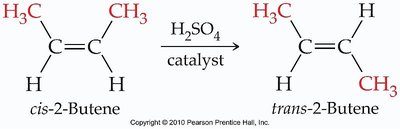
Double and triple bonds restrict rotation, leading to the possibility of geometric (cis-trans) isomerism in alkenes.
Cis-trans isomers: Occur when each carbon of the double bond has two different groups attached.
Cis isomer: Similar groups are on the same side of the double bond.
Trans isomer: Similar groups are on opposite sides of the double bond.
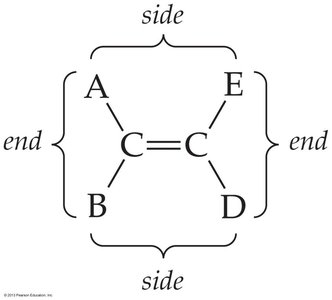
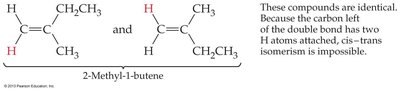
Not all alkenes can form cis-trans isomers; if either carbon of the double bond has two identical groups, isomerism is not possible.
Properties of Alkenes and Alkynes
Physical properties are similar to alkanes (nonpolar, low solubility in water).
Chemically, alkenes and alkynes are more reactive due to the presence of π bonds, which are more easily broken than σ bonds.
They readily undergo addition reactions, converting unsaturated compounds to saturated ones.
General Types of Organic Reactions
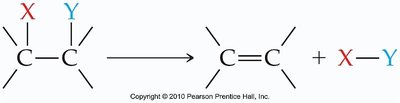
Addition Reactions
In an addition reaction, atoms or groups are added to the carbons of a multiple bond, resulting in a saturated product.
General form: (with X and Y added to the carbons)
Elimination Reactions
Elimination reactions are the reverse of addition reactions, where atoms or groups are removed from adjacent carbons, forming a multiple bond.
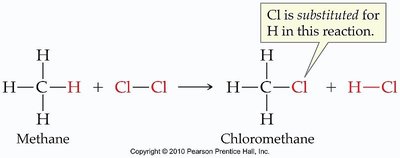
Substitution Reactions
In substitution reactions, one atom or group in a molecule is replaced by another atom or group.
Rearrangement Reactions
Rearrangement reactions involve the reorganization of bonds within a molecule to form an isomer.
Reactions of Alkenes and Alkynes
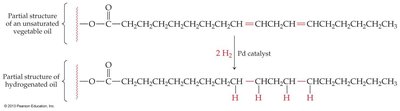
Hydrogenation
Hydrogenation is the addition of hydrogen (H2) across a double or triple bond, converting unsaturated compounds to saturated ones. A metal catalyst such as palladium (Pd) is required.
Example: Conversion of vegetable oil (unsaturated fat) to margarine (saturated fat).
Halogenation
Halogenation is the addition of halogens (Cl2 or Br2) to a double or triple bond, forming dihaloalkanes.
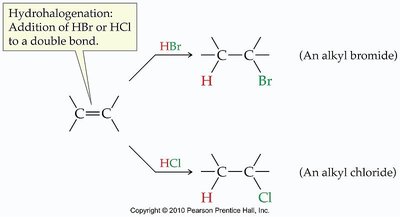
Hydrohalogenation
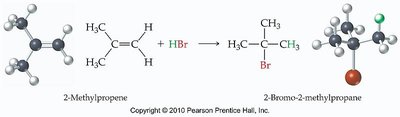
Hydrohalogenation is the addition of hydrogen halides (HCl, HBr) to alkenes or alkynes. The regioselectivity of this reaction is described by Markovnikov's Rule:
Markovnikov's Rule: The hydrogen atom attaches to the carbon with more hydrogens already attached; the halogen attaches to the carbon with fewer hydrogens.
If both carbons have equal hydrogens, two products are formed.
Hydration
Hydration is the addition of water (H2O) to an alkene, usually in the presence of a strong acid catalyst. It also follows Markovnikov's Rule, with H attaching to the carbon with more hydrogens and OH to the carbon with fewer hydrogens.
Alkene Polymers
Polymerization
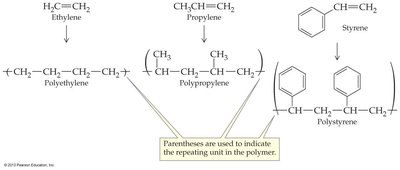
Polymerization is the process by which small molecules (monomers) join to form large molecules (polymers). Many alkenes undergo addition polymerization in the presence of a catalyst.
Monomer: The small repeating unit (e.g., ethylene, propylene).
Polymer: The large molecule formed (e.g., polyethylene, polypropylene).
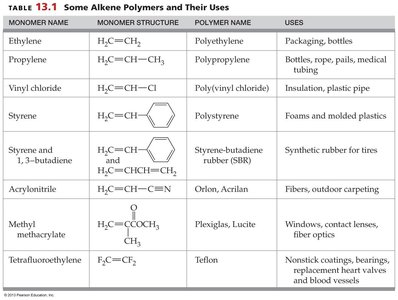
Common Alkene Polymers and Their Uses
Monomer Name | Monomer Structure | Polymer Name | Uses |
|---|---|---|---|
Ethylene | H2C=CH2 | Polyethylene | Packaging, bottles |
Propylene | H2C=CH–CH3 | Polypropylene | Bottles, rope, pails, medical equipment |
Vinyl chloride | H2C=CH–Cl | Poly(vinyl chloride) | Insulation, plastic pipe |
Styrene |
| Polystyrene | Foams and molded plastics |
Styrene and 1,3-butadiene | H2C=CH–CH=CH2 | Styrene-butadiene rubber (SBR) | Synthetic rubber for tires |
Acrylonitrile | H2C=CH–CN | Orlon, Acrilan | Fibers, outdoor carpeting |
Methyl methacrylate | H2C=C(COCH3)OCH3 | Plexiglas, Lucite | Windows, contact lenses, fiber optics |
Tetrafluoroethylene | F2C=CF2 | Teflon | Nonstick coatings, bearings, replacement heart valves and blood vessels |
Aromatic Compounds
Structure and Resonance
Aromatic compounds contain benzene-like rings with delocalized electrons. The actual structure is a resonance hybrid, with electrons shared equally among the six carbon atoms, resulting in bond order of 1.5 between each pair of carbons.
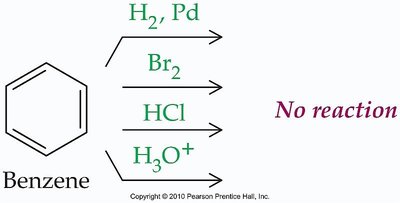
Benzene does not undergo typical addition reactions; it is unusually stable due to resonance.
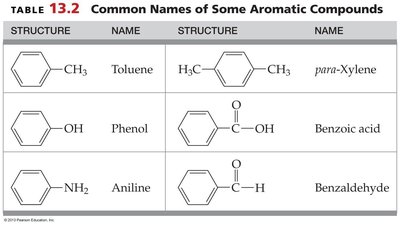
Naming Aromatic Compounds
Single substitutions on benzene are named as derivatives of benzene (e.g., chlorobenzene, nitrobenzene).
Multiple substituents are indicated by numbering or by using ortho-, meta-, para- prefixes for relative positions.
Some aromatic compounds have common names (e.g., toluene, phenol, aniline, benzoic acid, benzaldehyde, para-xylene).
Structure | Name | Structure | Name |
|---|---|---|---|
Benzene ring with CH3 | Toluene | Benzene ring with two CH3 (para) | para-Xylene |
Benzene ring with OH | Phenol | Benzene ring with COOH | Benzoic acid |
Benzene ring with NH2 | Aniline | Benzene ring with CHO | Benzaldehyde |
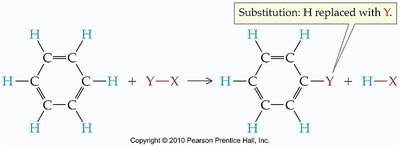
Reactions of Aromatic Compounds
Aromatic compounds typically undergo substitution reactions rather than addition reactions, preserving the aromatic ring.
Nitration: Substitution of a nitro group (–NO2).
Halogenation: Substitution of a halogen atom.
Additional info: The resonance stabilization of benzene is a key reason for its unique reactivity, and the rules for naming aromatic compounds are essential for clear communication in organic chemistry.
