 Back
BackAlkenes and Alkynes: Structure, Nomenclature, Properties, and Reactions
Study Guide - Smart Notes

Families of Organic Molecules: Hydrocarbons
Classification of Hydrocarbons
Hydrocarbons are organic compounds composed exclusively of carbon and hydrogen atoms. They are divided into several families based on the types of bonds between carbon atoms:
Alkanes: Only single bonds (saturated hydrocarbons)
Alkenes: At least one carbon–carbon double bond
Alkynes: At least one carbon–carbon triple bond
Aromatic Compounds: Six-membered rings with alternating double bonds
Alkanes are saturated, while alkenes and alkynes are unsaturated due to the presence of multiple bonds.
Nomenclature of Alkenes and Alkynes
General Principles of Naming
The IUPAC system for naming alkenes and alkynes follows a logical structure:
Prefix: Indicates the location of substituents
Parent: Specifies the number of carbon atoms in the longest chain containing the multiple bond
Suffix: Identifies the family (-ene for alkenes, -yne for alkynes)

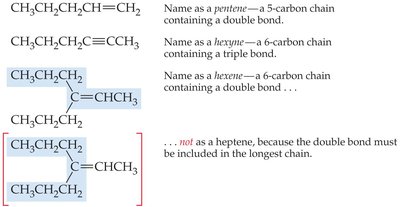
Step 1: Identify the Parent Chain
Find the longest continuous chain that includes the double or triple bond. The parent name is based on this chain, and the suffix is added accordingly.
If there are multiple double or triple bonds, use prefixes such as di-, tri-, etc.
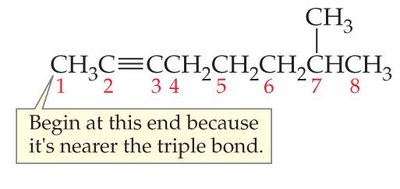
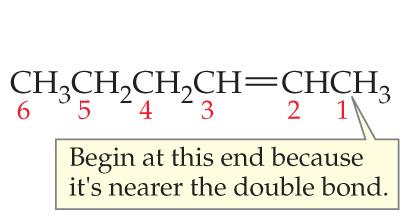
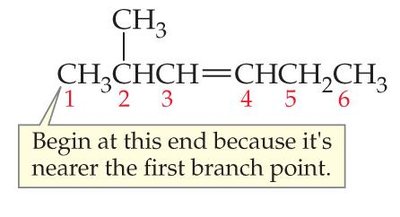
Step 2: Number the Carbon Chain
Number the chain so that the multiple bond gets the lowest possible index. If the multiple bond is equidistant from both ends, start numbering from the end nearest the first branch point.
Step 3: Naming Cycloalkenes
For cycloalkenes, assign the double-bonded carbons as positions 1 and 2, and number the ring to give substituents the lowest possible numbers.
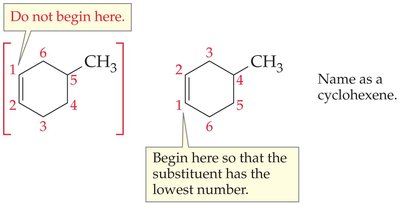
Step 4: Write the Full Name
Assign numbers to substituents and list them alphabetically.
Indicate the position of the multiple bond by the number of the first carbon involved.
Use commas to separate numbers and hyphens to separate words from numbers.
Structure and Bonding in Alkenes and Alkynes
Hybridization and Geometry
The type of bonding in hydrocarbons affects their geometry and bond angles:
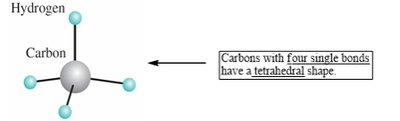
sp3 hybridization: Four single bonds, tetrahedral geometry, bond angle of 109.5°
sp2 hybridization: Double bond, trigonal planar geometry, bond angle of 120°
sp hybridization: Triple bond, linear geometry, bond angle of 180°



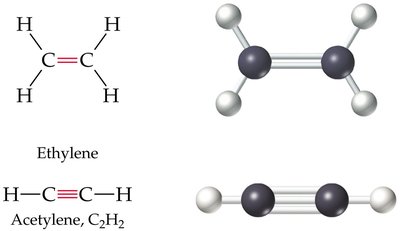
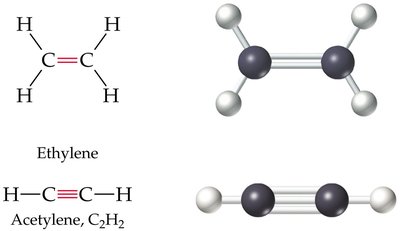
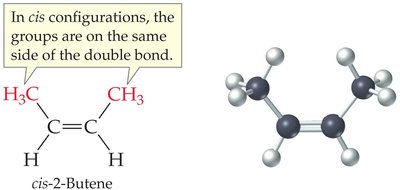
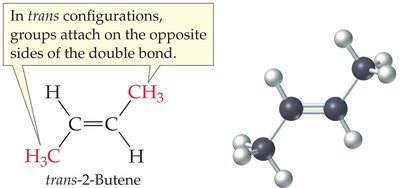
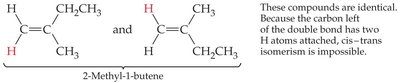
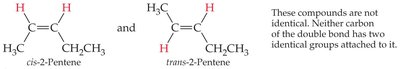
Cis–Trans (Geometric) Isomerism in Alkenes
Definition and Requirements
Alkenes can exhibit cis–trans isomerism (also called geometric isomerism) due to restricted rotation around the double bond. This occurs when each carbon of the double bond has two different substituents.
Cis isomer: Substituents are on the same side of the double bond
Trans isomer: Substituents are on opposite sides of the double bond
Physical Properties of Alkenes and Alkynes
General Properties
Alkenes and alkynes share many physical properties with alkanes, but their multiple bonds impart some differences:
Nonpolar molecules; physical properties dominated by London dispersion forces
Low boiling and melting points, which increase with molecular size
Alkenes and alkynes with 2–4 carbons are gases at room temperature
Insoluble in water, soluble in nonpolar solvents
Less dense than water
Flammable and generally nontoxic
Chemical Reactions of Alkenes and Alkynes
Types of Organic Reactions
Organic reactions are classified into four main types:
Addition reactions: Atoms are added to the multiple bond, converting it to single bonds
Elimination reactions: Atoms are removed, forming a multiple bond
Substitution reactions: One atom or group is replaced by another
Rearrangement reactions: The structure of the molecule is reorganized to form an isomer
Addition Reactions
Most reactions of alkenes and alkynes are addition reactions. A typical example is the addition of hydrogen (hydrogenation):
Alkenes/alkynes + H2 (with metal catalyst) → Alkane
General equation:
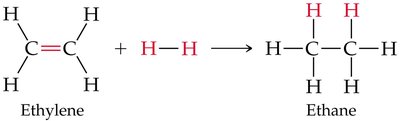
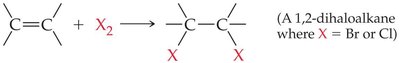
Halogenation
Alkenes and alkynes react with halogens (Cl2, Br2) to form dihaloalkanes:
Hydrohalogenation and Markovnikov's Rule
When alkenes react with hydrogen halides (HCl, HBr), the addition follows Markovnikov's rule:
The hydrogen atom attaches to the carbon with more hydrogen atoms already attached.
The halide attaches to the carbon with fewer hydrogen atoms.
Example equation:
(major product)
Summary Table: Comparison of Hydrocarbon Families
Family | General Formula | Bond Type | Geometry | Hybridization |
|---|---|---|---|---|
Alkane | CnH2n+2 | Single | Tetrahedral (109.5°) | sp3 |
Alkene | CnH2n | Double | Trigonal planar (120°) | sp2 |
Alkyne | CnH2n-2 | Triple | Linear (180°) | sp |
Additional info: The notes also briefly mention functional groups containing oxygen and nitrogen, which are covered in later chapters. The focus here is on hydrocarbons and their reactions.
