 Back
BackAmino Acids, Proteins, and Enzymes: Structure, Function, and Biological Importance
Study Guide - Smart Notes

Proteins and Amino Acids
Introduction to Proteins
Proteins are large, complex molecules essential for the structure, function, and regulation of the body's tissues and organs. They are polymers made from 20 different amino acids, and their characteristics and functions depend on the specific sequence of these amino acids.
Structural components: Cartilage, muscles, hair, and nails are made of proteins.
Enzymatic function: Proteins act as enzymes to regulate biological reactions such as digestion and cellular metabolism.
Transport: Hemoglobin and myoglobin transport oxygen in the blood and muscles.
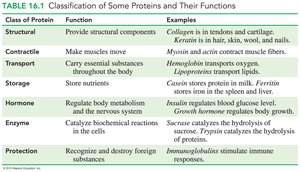
Functions of Proteins
Structural: Provide support and shape to cells and tissues.
Contractile: Enable muscle movement.
Transport: Carry essential substances throughout the body.
Storage: Store nutrients.
Hormonal: Regulate body metabolism and the nervous system.
Enzymatic: Catalyze biochemical reactions.
Protection: Recognize and destroy foreign substances.
Amino Acids: Structure and Classification
Amino acids are the building blocks of proteins. Each amino acid contains a central carbon atom (the α-carbon) bonded to an ammonium group (—NH3+), a carboxylate group (—COO−), a hydrogen atom, and a unique side chain (R group).
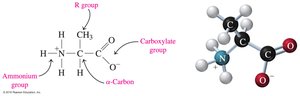
Nonpolar (hydrophobic): R group is H, alkyl, or aromatic.
Polar (hydrophilic): R group is an alcohol, thiol, or amide.
Acidic: R group is a carboxylic acid.
Basic: R group is an amine.
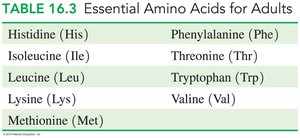
Essential Amino Acids
Of the 20 amino acids, 9 are essential and must be obtained from the diet, while the other 11 can be synthesized by the body. Complete proteins (e.g., eggs, milk, meat, fish) contain all essential amino acids, while incomplete proteins (e.g., grains, beans, nuts) may lack one or more.

Protein Structure
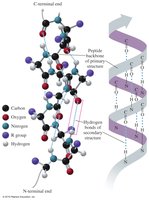
Primary Structure
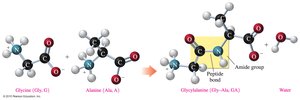
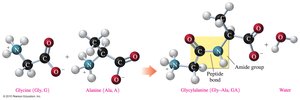
The primary structure of a protein is the specific sequence of amino acids in a polypeptide chain, held together by peptide bonds. The sequence determines the protein's properties and function.
Peptide bond: An amide bond formed between the carboxylate group of one amino acid and the ammonium group of the next.
Residue: An amino acid that is part of a polymer chain.
Secondary Structure
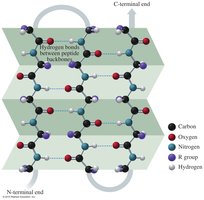
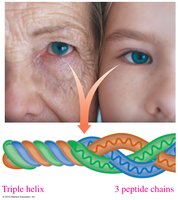
The secondary structure refers to the regular folding patterns of the polypeptide backbone, stabilized by hydrogen bonds. The main types are the alpha helix, beta-pleated sheet, and triple helix.
Alpha helix (α-helix): A coiled structure stabilized by hydrogen bonds between the C=O and N—H groups.
Beta-pleated sheet (β-sheet): Polypeptide chains lie side by side, forming sheets stabilized by hydrogen bonds.
Triple helix: Three polypeptide chains woven together, typical of collagen.
Tertiary Structure
The tertiary structure is the overall three-dimensional shape of a protein, determined by interactions between R groups of amino acids. These include hydrophobic interactions, hydrophilic interactions, salt bridges, hydrogen bonds, and disulfide bonds.
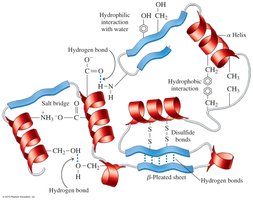
Hydrophobic interactions: Between nonpolar side chains.
Hydrophilic interactions: Between polar side chains and water.
Salt bridges: Ionic bonds between acidic and basic side chains.
Hydrogen bonds: Between polar side chains.
Disulfide bonds: Covalent bonds between cysteine residues.
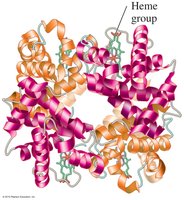
Quaternary Structure
The quaternary structure is the association of two or more polypeptide chains (subunits) to form a functional protein. Hemoglobin is a classic example, consisting of four subunits.
Summary Table: Structural Levels in Proteins
Structural Level | Characteristics |
|---|---|
Primary | Peptide bonds join amino acids in a specific sequence in a polypeptide. |
Secondary | Hydrogen bonding forms α-helix, β-pleated sheet, or triple helix along the chain. |
Tertiary | Polypeptide folds into a compact, three-dimensional shape stabilized by R group interactions. |
Quaternary | Two or more protein subunits combine to form a biologically active protein. |

Protein Denaturation and Hydrolysis
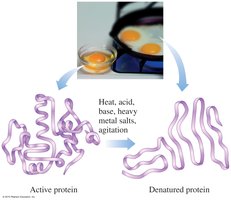
Denaturation of Proteins
Denaturation is the process by which a protein loses its native structure due to the disruption of non-covalent interactions and disulfide bonds. This can be caused by heat, acids, bases, heavy metals, or agitation, leading to loss of biological function.
Denaturing Agent | Bonds Disrupted | Examples |
|---|---|---|
Heat Above 50°C | Hydrogen bonds, hydrophobic interactions | Cooking food, autoclaving surgical items |
Acids and Bases | Hydrogen bonds, salt bridges | Yogurt, cheese production |
Organic Compounds | Hydrophobic interactions | Disinfectants |
Heavy Metal Ions | Disulfide bonds | Mercury, lead poisoning |
Agitation | Hydrogen bonds, hydrophobic interactions | Whipped cream, meringue |

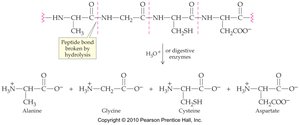
Protein Hydrolysis
Protein hydrolysis is the reverse of protein formation, where peptide bonds are broken by water, yielding individual amino acids. This process is catalyzed by enzymes during digestion.
Enzymes: Structure and Function
Enzymes as Biological Catalysts
Enzymes are proteins that act as biological catalysts, increasing the rate of chemical reactions by lowering the activation energy. Each enzyme has a specific region called the active site, where substrates bind and reactions are catalyzed.
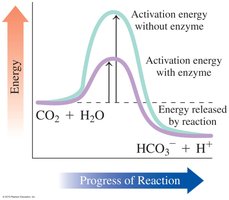
Specificity: Enzymes are highly specific for their substrates and the reactions they catalyze.
Naming: Enzyme names often end in -ase and indicate the substrate or reaction type (e.g., sucrase, oxidase).
Classification of Enzymes
Class | Type of Reaction Catalyzed |
|---|---|
Oxidoreductases | Oxidation–reduction |
Transferases | Transfer groups of atoms |
Hydrolases | Hydrolysis |
Lyases | Add or remove atoms to or from a double bond |
Isomerases | Rearrange atoms |
Ligases | Use ATP to combine small molecules |

Enzyme-Substrate Interaction
The active site of an enzyme binds the substrate, forming an enzyme-substrate (ES) complex. The reaction occurs, products are released, and the enzyme is free to catalyze another reaction.
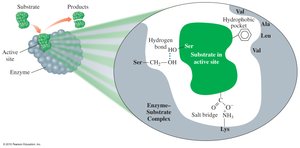
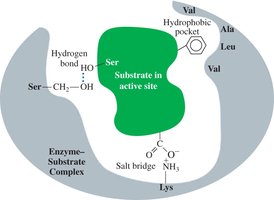
Lock-and-key model: The active site is rigid and fits only specific substrates.
Induced-fit model: The active site is flexible and adjusts to fit the substrate, enhancing catalysis.
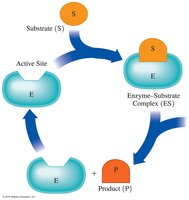
Factors Affecting Enzyme Activity
Temperature
Enzymes have an optimum temperature (usually 37°C in humans). Activity decreases at low temperatures and is lost at high temperatures due to denaturation.

pH
Each enzyme has an optimum pH, typically around 7.4 in the body. Activity decreases at pH values outside the optimum range due to disruption of the enzyme's structure.
Substrate and Enzyme Concentration
Substrate concentration: Increasing substrate increases reaction rate until the enzyme is saturated (Vmax).
Enzyme concentration: Increasing enzyme increases reaction rate proportionally.
Enzyme Inhibition
Enzyme inhibitors are molecules that decrease or stop enzyme activity. Inhibition can be reversible or irreversible.
Competitive inhibition: Inhibitor resembles the substrate and competes for the active site; can be overcome by increasing substrate concentration.
Noncompetitive inhibition: Inhibitor binds elsewhere on the enzyme, altering the active site; cannot be overcome by increasing substrate.
Irreversible inhibition: Inhibitor covalently binds to the enzyme, permanently inactivating it.
Regulation of Enzyme Activity
Allosteric Enzymes and Feedback Control
Allosteric enzymes have regulatory sites where molecules can bind to increase (positive regulator) or decrease (negative regulator) enzyme activity. Feedback control occurs when the end product of a pathway regulates an enzyme involved in its synthesis.
Covalent Modification and Genetic Control
Some enzymes are regulated by covalent modification (e.g., phosphorylation) or by genetic control, where enzyme synthesis is regulated at the DNA level.
Enzyme Cofactors and Vitamins
Cofactors and Coenzymes
Cofactors are nonprotein components essential for enzyme activity. They can be metal ions or organic molecules (coenzymes). Many vitamins function as coenzymes or are required for their synthesis.
Water-soluble vitamins: Function as coenzymes, not stored in the body, excess excreted in urine.
Fat-soluble vitamins (A, D, E, K): Stored in body fat, important for vision, bone formation, antioxidants, and blood clotting.
Antioxidants
Antioxidants such as vitamin C, vitamin E, β-carotene, and selenium protect the body from oxidative damage by neutralizing free radicals.
