 Back
BackAnalyzing Physical and Chemical Changes Using Molecular Sketches
Study Guide - Smart Notes

Q2. For each of the three changes shown in the sketches below, decide whether the change is possible. If it is possible, determine whether it is a physical change or a chemical change.
Background
Topic: Physical and Chemical Changes at the Molecular Level
This question tests your ability to interpret molecular-level diagrams and distinguish between physical and chemical changes. You are asked to analyze how the arrangement and identity of molecules change in each scenario.
Key Terms and Concepts:
Physical Change: A change in the state or appearance of a substance that does not alter its chemical composition (e.g., melting, freezing, dissolving).
Chemical Change: A process in which one or more substances are converted into new substances with different chemical properties (e.g., combustion, rusting).
Molecular Sketch: A visual representation of molecules, showing how atoms are connected and arranged.
Step-by-Step Guidance
Examine the original sample (top sketch) and note the type and arrangement of molecules present. Are all molecules identical? What do the colors and connections represent?
For Change 1 (
 ), compare the arrangement of molecules to the original. Are the molecules still the same type, just arranged differently? Or have the molecules themselves changed?
), compare the arrangement of molecules to the original. Are the molecules still the same type, just arranged differently? Or have the molecules themselves changed?For Change 2 (
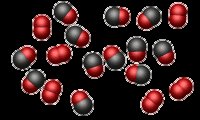 ), look for evidence of new types of molecules or atoms bonded in new ways. Does this indicate a chemical reaction?
), look for evidence of new types of molecules or atoms bonded in new ways. Does this indicate a chemical reaction?For Change 3 (
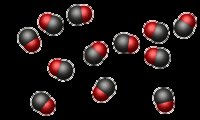 ), check if the number and type of molecules are conserved, or if new substances have formed. Is this change possible given the constraints (sealed box, no matter enters or leaves)?
), check if the number and type of molecules are conserved, or if new substances have formed. Is this change possible given the constraints (sealed box, no matter enters or leaves)?For each change, decide if it is impossible, a physical change, or a chemical change. Use the definitions above to justify your reasoning.
Try solving on your own before revealing the answer!
