 Back
BackAtoms and Atomic Theory: Laws, Mole Concept, and the Periodic Table
Study Guide - Smart Notes

Atoms and Atomic Theory
Scientific Laws: Conservation of Mass and Definite Proportions
The foundation of modern chemistry rests on several scientific laws that describe how matter behaves during chemical reactions. These laws are essential for understanding the nature of atoms and compounds.
Law of Conservation of Mass: Matter is neither created nor destroyed during a chemical reaction. The total mass of the products equals the total mass of the reactants. This principle is fundamental to all chemical equations and reactions.
Example: When 100.00 grams of mercuric oxide decomposes, it produces 92.61 grams of mercury and 7.39 grams of oxygen, demonstrating mass conservation.
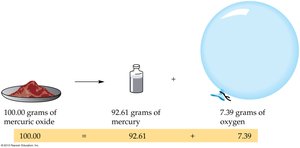
Law of Definite Proportions: A compound always contains the same elements in the same ratio by mass, regardless of the sample size or source.
Example: Lead sulfide always forms from lead and sulfur in a fixed mass ratio, with any excess of either element remaining as leftovers.

Law of Multiple Proportions
This law states that elements can combine in more than one ratio to form different compounds. Each distinct ratio corresponds to a unique compound.
Example: The formation of lead sulfide from varying amounts of lead and sulfur illustrates how different ratios yield different compounds or leftovers.
John Dalton and the Atomic Theory of Matter
Dalton's Atomic Theory
John Dalton proposed a theory that explained the behavior of atoms and the formation of compounds. His ideas laid the groundwork for modern chemistry.
Key Points:
All matter is composed of extremely small particles called atoms.
Atoms of a given element are identical, but differ from atoms of other elements.
Compounds are formed when atoms of different elements combine in fixed proportions.
Chemical reactions involve the rearrangement of atoms; atoms are not created or destroyed.
Modern Modifications: Dalton's theory has been refined to account for atomic divisibility, isotopes, and the existence of subatomic particles.
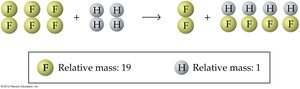
Atomic Theory in Practice
Atomic theory explains how elements combine to form compounds and how mass ratios are determined.
Example: 1 atom of hydrogen (H) combines with 1 atom of fluorine (F), so 1 g of H combines with 19 g of F. If 4 g of H interacts with 114 g of F, the result is 80 g of HF and 38 g of F left over.
Molecule: A group of chemically bonded atoms in an exact proportion.
The Mole and Avogadro's Number
Avogadro’s Number and the Mole Concept
The mole is a fundamental unit in chemistry, representing a specific number of particles (atoms, molecules, etc.). Avogadro’s number defines the quantity in one mole.
Avogadro’s Number: particles per mole.
Definition: A mole is the amount of substance containing as many elementary entities as there are atoms in 12 grams of carbon-12.
Examples:
1 mole of sodium contains atoms of Na.
1 mole of carbon dioxide contains molecules of CO2.
1 mole of sodium chloride contains formula units of NaCl.
Visual Representation: Avogadro’s number is so large that, for example, baseballs would roughly equal the mass of the earth.

Molar Mass (MM)
Molar mass is the mass (in grams) of one mole of a substance. For elements, it is numerically equal to the atomic mass, but expressed in grams per mole.
For Elements:
Sodium (Na): 22.9898 g/mol
Aluminum (Al): 26.9815 g/mol
Gold (Au): 196.9665 g/mol
For Compounds: The molar mass is the sum of the atomic masses of all atoms in the formula.
MM(CO2) = g/mol
MM(NaCl) = g/mol
Interpretation: 1 mole of a substance weighs its molar mass in grams.
Dimensional Analysis and Relationships
Dimensional analysis is used to convert between mass, moles, and number of particles. The relationships are central to solving chemical problems.
Key Relationships:
Number of atoms or molecules → Avogadro’s Number → Moles → Molar Mass → Mass of element or compound

Example: How many atoms of Ar are contained in 0.345 mole of Ar? Solution:
Example: What is the mass of 0.250 mol of Na? Solution:
Mendeleev and the Periodic Table
Atomic Mass Units and Isotopes
Atomic mass units (amu) represent the average atomic weight of an element, accounting for all naturally occurring isotopes. Not all atoms of an element are identical; isotopes are different forms of the same element.
The Birth of the Periodic Table
Dmitri Mendeleev organized elements based on their properties and increasing atomic mass, creating the first periodic table. He left intentional gaps, predicting the existence of undiscovered elements.
Example: Mendeleev’s original periodic table arranged elements in a two-dimensional matrix, with gaps for predicted elements.
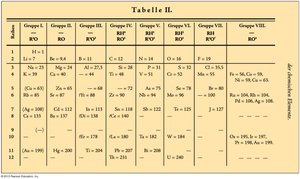
Predictive Power: The periodic table allowed chemists to predict properties of elements yet to be discovered.
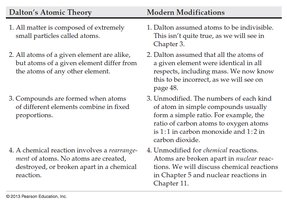
Summary Table: Dalton's Atomic Theory vs. Modern Modifications
This table compares Dalton's original atomic theory with modern scientific modifications.
Dalton's Atomic Theory | Modern Modifications |
|---|---|
All matter is composed of extremely small particles called atoms. | Dalton assumed atoms to be indivisible. This is not true, as we will see in Chapter 3. |
All atoms of a given element are alike, but atoms of a given element differ from the atoms of any other element. | Dalton assumed all atoms of a given element were identical in all respects, including mass. We now know this is incorrect, as we will see in Chapter 3. |
Compounds are formed when atoms of different elements combine in fixed proportions. | Unmodified. The numbers of each kind of atom in simple compounds usually form a simple ratio. For example, atoms combine in ratios such as 1:1 in carbon monoxide and 1:2 in carbon dioxide. |
A chemical reaction involves a rearrangement of atoms. No atoms are created, destroyed, or broken apart in a chemical reaction. | Unmodified for chemical reactions. Atoms are created or destroyed in nuclear reactions. We will discuss chemical reactions in Chapter 5 and nuclear reactions in Chapter 11. |
Key Terms and Definitions
Atom: The smallest unit of an element that retains its chemical properties.
Molecule: A group of atoms bonded together in a fixed ratio.
Mole: The amount of substance containing Avogadro’s number of particles.
Molar Mass: The mass of one mole of a substance, expressed in grams per mole.
Isotope: Atoms of the same element with different numbers of neutrons.
Atomic Mass Unit (amu): A unit of mass used to express atomic and molecular weights.
Additional info:
Some examples and solution maps referenced in the notes are not fully detailed; students should refer to their textbook for step-by-step calculations.
The H2O2 joke image (image_5) is not included, as it is not directly relevant to the academic explanation.
Image_7 and image_9 are included to visually reinforce the concept of Avogadro's number and the relationships between moles, mass, and number of particles.
Image_10 is included to show the historical context of Mendeleev's periodic table.
