 Back
BackAtoms and Elements: Isotopes, Atomic Mass, Electron Energy Levels, and Periodic Trends
Study Guide - Smart Notes

Atoms and Elements
Introduction
Atoms are the fundamental building blocks of matter, and elements are pure substances composed of only one type of atom. Understanding the structure of atoms, isotopes, atomic mass, electron energy levels, and periodic trends is essential for studying chemistry.
Isotopes and Atomic Mass
Isotopes
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons, resulting in different mass numbers. Isotopes of an element exhibit nearly identical chemical behavior but may have different physical properties due to their mass differences.
Protons: Determine the identity of the element (atomic number).
Neutrons: Differ among isotopes, affecting the mass number.
Electrons: Equal to the number of protons in a neutral atom.
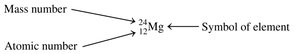
Atomic Symbol
An atomic symbol represents a specific isotope and is written as:
X: Chemical symbol of the element
A: Mass number (protons + neutrons)
Z: Atomic number (number of protons)
Examples of Isotopes
Carbon Isotopes: , ,
Each has 6 protons, but 6, 7, and 8 neutrons, respectively.
Atomic Mass
The atomic mass of an element is the weighted average of the masses of all naturally occurring isotopes, measured in atomic mass units (amu). It is not the same as the mass number of any single isotope.
Atomic mass is listed below the element symbol on the periodic table.
Calculated using the percent abundance and mass of each isotope:

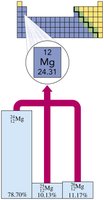
Isotopes of Magnesium
Magnesium has three naturally occurring isotopes: , , and . The atomic mass of magnesium is 24.31 amu, reflecting the weighted average of these isotopes.
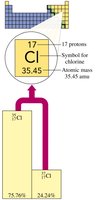
Calculating Atomic Mass: Example with Chlorine
Chlorine has two main isotopes: and . The atomic mass of chlorine (35.45 amu) is closer to the mass of $ ^{35}\mathrm{Cl} $, indicating it is more abundant.
Electron Energy Levels
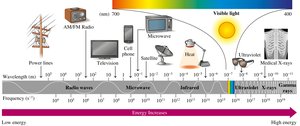
Electromagnetic Radiation and the Spectrum
Electromagnetic radiation includes all forms of energy that travel as waves, such as visible light, microwaves, and X-rays. The wavelength is the distance between wave peaks; shorter wavelengths correspond to higher energy.
Atomic Spectrum
When light from a heated element passes through a prism, it separates into distinct lines called an atomic spectrum. Each element has a unique atomic spectrum, which is related to the energy levels of its electrons.

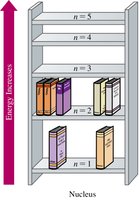
Electron Energy Levels (Principal Quantum Numbers)
Electrons in an atom occupy specific energy levels, designated by principal quantum numbers (). Energy increases as $ n $ increases and as electrons are farther from the nucleus. Electron energies are quantized, meaning electrons can only exist at certain energy levels.

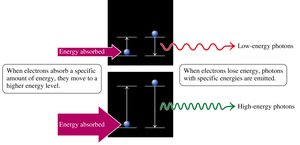
Changes in Electron Energy Levels
When an electron absorbs energy, it moves to a higher energy level (excited state).
When it falls to a lower energy level, it emits energy as light (photon).
Electron Arrangements for the First 20 Elements
Electron arrangements (configurations) describe how electrons are distributed among energy levels. For example, sodium (Na, atomic number 11) has the arrangement 2,8,1.
Period 1: H (1), He (2) fill the first energy level.
Period 2: Li (3) to Ne (10) fill the first and second energy levels.
Period 3: Na (11) to Ar (18) fill the first, second, and third energy levels.
Period 4: K (19), Ca (20) begin filling the fourth energy level.
Trends in Periodic Properties
Periodic Properties Overview
The arrangement of electrons in atoms determines periodic trends in properties such as atomic size, ionization energy, and metallic character. These trends repeat in each period of the periodic table.

Group Number and Valence Electrons
For representative elements (Groups 1A–8A), the group number indicates the number of valence electrons (electrons in the outermost energy level). Valence electrons determine chemical reactivity.
Lewis Symbols
Lewis symbols represent valence electrons as dots around the element symbol. For example, aluminum (Al) with three valence electrons is shown as:

Magnesium (Mg) with two valence electrons can be represented in several ways:
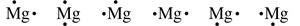
Lewis symbols for selected elements are summarized in the table below:

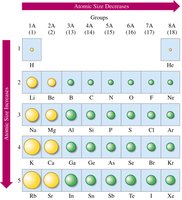
Atomic Size
Atomic size increases down a group (due to additional energy levels) and decreases across a period (due to increased nuclear charge pulling electrons closer).
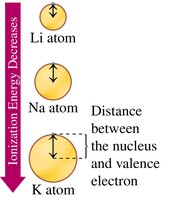
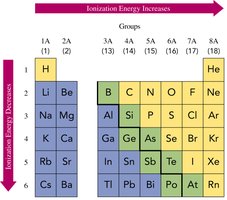
Ionization Energy
Ionization energy is the energy required to remove an outermost electron. It decreases down a group (outer electrons are farther from the nucleus) and increases across a period (nuclear charge increases).
Metallic Character
Metallic character refers to the tendency of an element to lose electrons. It increases down a group and decreases across a period.
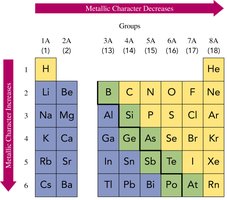
Summary of Trends in the Periodic Table
Periodic trends can be summarized as follows:
Atomic size: Increases down a group, decreases across a period.
Ionization energy: Decreases down a group, increases across a period.
Metallic character: Increases down a group, decreases across a period.
Concept Map: Atoms and Elements
Additional info: These notes cover the core concepts of isotopes, atomic mass, electron energy levels, and periodic trends, which are foundational for understanding chemical behavior and the organization of the periodic table.
