 Back
BackAtoms & Elements: Periodic Trends and Atomic Structure
Study Guide - Smart Notes

Atoms & Elements: Periodic Trends
Historical Development of Atomic Theory
The understanding of atomic structure has evolved through key scientific discoveries. Early models proposed by Dalton, Thomson, Rutherford, and Bohr laid the foundation for modern atomic theory.
Dalton's Atomic Theory (1803): All matter is composed of atoms; atoms of a given element are identical; atoms of different elements differ; compounds are combinations of atoms; chemical reactions rearrange atoms.
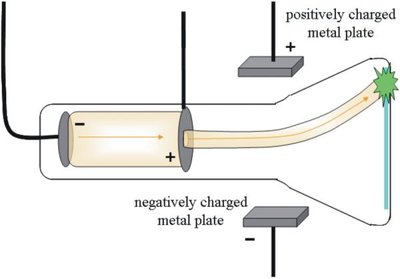
Thomson's Model (1897): Discovery of the electron as a negatively charged particle within the atom.
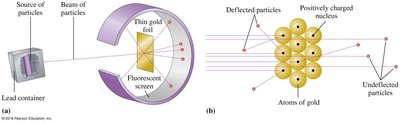
Rutherford's Model (1909): The atom has a dense, positively charged nucleus with electrons surrounding it; most of the atom is empty space.
Bohr's Model (1913): Electrons orbit the nucleus in quantized energy levels.


Atomic Structure
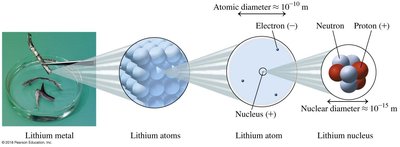
Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in defined energy levels. The nucleus is much smaller than the overall atom, but contains most of its mass.
Proton: Positively charged, located in the nucleus.
Neutron: Neutral, located in the nucleus.
Electron: Negatively charged, located outside the nucleus.
Atomic diameter: Approximately m; nuclear diameter approximately m.
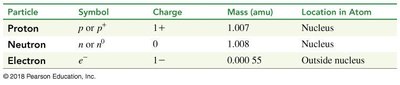
Particle | Symbol | Charge | Mass (amu) | Location in Atom |
|---|---|---|---|---|
Proton | p or p+ | 1+ | 1.007 | Nucleus |
Neutron | n or n0 | 0 | 1.008 | Nucleus |
Electron | e- | 1- | 0.00055 | Outside nucleus |
Bohr's Atomic Model
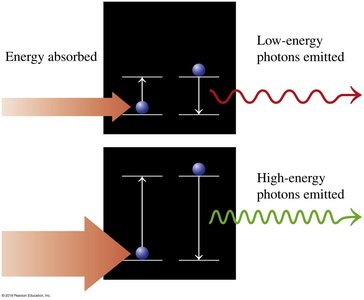
The Bohr model describes electrons orbiting the nucleus in discrete energy levels. The energy of each orbit is quantized, and electrons can absorb or emit energy to move between levels.
Lowest energy is found in the smallest orbit (closest to nucleus).
Radiation is absorbed/emitted when electrons change orbits.
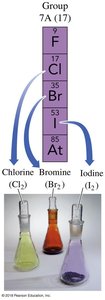
The Periodic Table
Classification of Elements
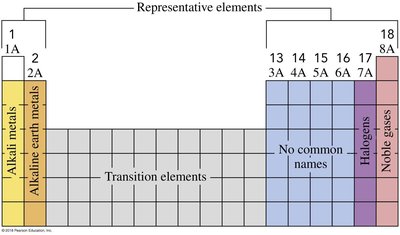
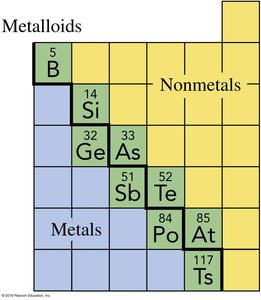
The periodic table organizes elements by increasing atomic number and groups them by similar chemical properties. Elements are classified as metals, nonmetals, and metalloids.
Groups: Vertical columns; elements in a group share similar properties.
Periods: Horizontal rows; elements in a period have increasing atomic number.
Representative elements: Groups 1A-8A.
Transition elements: Groups 3-12.
Metalloids: Elements with properties intermediate between metals and nonmetals.

Biological Relevance of Elements
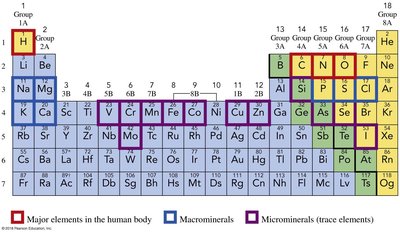
Certain elements are essential for life and are classified as major elements, macrominerals, and microminerals (trace elements).
Major elements: H, C, N, O, P, S
Macrominerals: Na, K, Mg, Ca, Cl
Microminerals: Fe, Zn, Cu, Mn, Mo, Se, I

Atomic Number, Mass Number, and Isotopes
Atomic Number and Mass Number
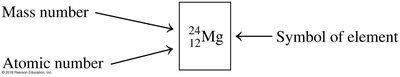
The atomic number is the number of protons in an atom and defines the element. The mass number is the sum of protons and neutrons.
Atomic Number (Z): Number of protons
Mass Number (A): Number of protons + neutrons
Element Symbol: Unique abbreviation for each element
Element | Symbol | Atomic Number | Mass Number | Number of Protons | Number of Neutrons | Number of Electrons |
|---|---|---|---|---|---|---|
Hydrogen | H | 1 | 1 | 1 | 0 | 1 |
Nitrogen | N | 7 | 14 | 7 | 7 | 7 |
Oxygen | O | 8 | 16 | 8 | 8 | 8 |
Chlorine | Cl | 17 | 37 | 17 | 20 | 17 |
Iron | Fe | 26 | 58 | 26 | 32 | 26 |
Gold | Au | 79 | 197 | 79 | 118 | 79 |
Isotopes
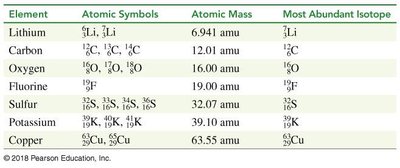
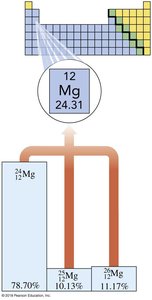
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers. Isotopes have identical chemical properties but may differ in physical properties.
Example: Magnesium has three isotopes: , ,
Atomic mass: Weighted average of all isotopes
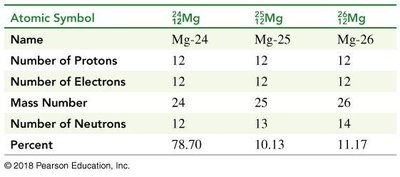
Atomic Symbol | Name | Number of Protons | Number of Electrons | Mass Number | Number of Neutrons | Percent |
|---|---|---|---|---|---|---|
Mg-24 | 12 | 12 | 24 | 12 | 78.70 | |
Mg-25 | 12 | 12 | 25 | 13 | 10.13 | |
Mg-26 | 12 | 12 | 26 | 14 | 11.17 |
Electron Energy Levels and the Electromagnetic Spectrum
Electron Energy Levels
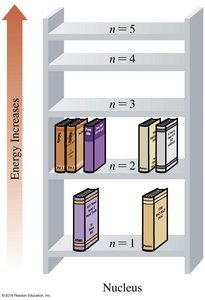
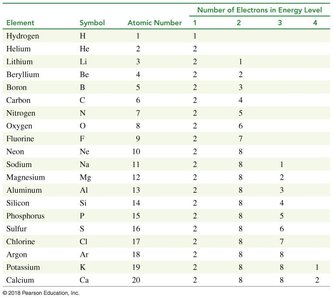
Electrons occupy quantized energy levels (shells) around the nucleus. Higher energy levels are farther from the nucleus. The arrangement of electrons determines chemical properties.
Energy levels: Denoted by principal quantum number n (n = 1, 2, 3, ...)
Valence electrons: Electrons in the outermost energy level
Electromagnetic Spectrum
The electromagnetic spectrum includes all types of electromagnetic radiation, from radio waves to gamma rays. Energy increases with frequency and decreases with wavelength.
Visible light: Only a small portion of the spectrum
Photon emission: When electrons drop to lower energy levels, photons are emitted
Periodic Trends
Atomic Size
Atomic size increases down a group (column) due to additional energy levels, and decreases across a period (row) due to increased nuclear charge pulling electrons closer.
Trend: Size increases down a group, decreases left-to-right across a period
Reason: More energy levels added down a group; increased nuclear charge contracts orbitals across a period
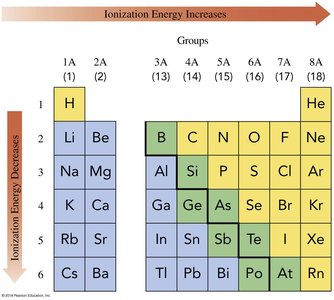
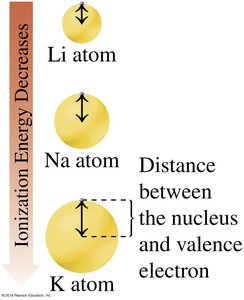
Ionization Energy
Ionization energy is the energy required to remove an electron from an atom. It decreases down a group (larger atomic radius) and increases across a period (greater nuclear charge).
Trend: Ionization energy decreases down a group, increases left-to-right across a period
Reason: Larger atomic radius makes it easier to remove electrons; increased nuclear charge makes it harder
Valence Electrons and Lewis Dot Structures
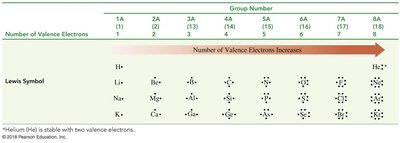
Lewis dot structures represent the valence electrons of an atom. The number of valence electrons corresponds to the group number for representative elements.
Valence electrons: Outermost electrons involved in bonding
Lewis symbol: Dots around the element symbol represent valence electrons
Group Number | Number of Valence Electrons | Lewis Symbol |
|---|---|---|
1A | 1 | H• |
2A | 2 | Be•• |
3A | 3 | B••• |
4A | 4 | C•••• |
5A | 5 | N••••• |
6A | 6 | O•••••• |
7A | 7 | F••••••• |
8A | 8 | Ne•••••••• |
Summary Table: Key Periodic Trends
Trend | Down a Group | Across a Period |
|---|---|---|
Atomic Size | Increases | Decreases |
Ionization Energy | Decreases | Increases |
Valence Electrons | Same | Increases |
