 Back
BackAtoms and Elements: Structure, Classification, and Biological Importance
Study Guide - Smart Notes

Atoms and Elements
Introduction to Atoms and Elements
Atoms and elements are foundational concepts in chemistry. Elements are pure substances that cannot be broken down into simpler substances by chemical means. Atoms are the smallest units of elements that retain their chemical properties. Understanding the structure and classification of elements is essential for studying chemical reactions and biological processes.
Elements and Symbols
Definition and Properties of Elements
Element: A pure substance from which all other substances are built. Elements cannot be broken down into simpler substances by ordinary chemical means.
Each element is represented by a unique chemical symbol, typically one or two letters (e.g., H for hydrogen, Al for aluminum).
Some symbols are derived from Latin or Greek names (e.g., Au for gold from aurum).
Example: Sulfur (S) is an element commonly found in nature.

Sources of Element Names
Element names may originate from planets, mythological figures, minerals, colors, geographic locations, or famous people.
Chemical Symbols
Symbols are one or two letters; the first is always capitalized, the second (if present) is lowercase.
Example: Co is cobalt, while CO represents a compound of carbon and oxygen.
Common Elements and Their Symbols
Hydrogen (H), Carbon (C), Nitrogen (N), Oxygen (O), Sodium (Na), Iron (Fe), Gold (Au), Silver (Ag), etc.
Some symbols are based on Latin names (e.g., Fe for ferrum).
Chemistry Link to Health: Toxicity of Mercury
Mercury (Hg) is a silvery, shiny liquid at room temperature.
It can enter the body via inhalation, skin contact, or ingestion of contaminated food/water.
Mercury exposure can damage the brain, kidneys, and impair development.

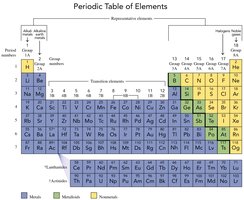
The Periodic Table
Organization and Structure
The periodic table arranges all known elements in order of increasing atomic number and groups them by similar chemical properties. It is a fundamental tool for chemists.
Vertical columns are called groups or families.
Horizontal rows are called periods.
Elements in the same group have similar chemical properties.
Group Numbers and Names
Groups are numbered 1–18 (or 1A–8A for representative elements, and 1B–8B for transition elements).
Common group names include:
Group 1A (1): Alkali metals
Group 2A (2): Alkaline earth metals
Group 7A (17): Halogens
Group 8A (18): Noble gases
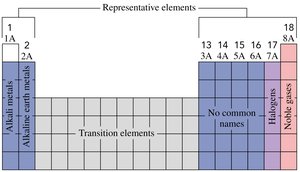
Representative Elements
Groups 1A–8A are called representative elements and display a wide range of chemical and physical properties.
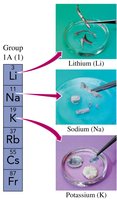
Alkali Metals and Halogens
Alkali metals (Group 1A): Highly reactive metals such as lithium (Li), sodium (Na), and potassium (K).
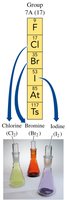
Halogens (Group 7A): Reactive nonmetals such as fluorine (F), chlorine (Cl), bromine (Br), and iodine (I).
Metals, Nonmetals, and Metalloids
Metals: Shiny, ductile, malleable, good conductors of heat and electricity, mostly solid at room temperature (except mercury).
Nonmetals: Dull, brittle, poor conductors, often gases or solids with low melting points.
Metalloids: Exhibit properties intermediate between metals and nonmetals; semiconductors.

Comparison Table: Metal, Nonmetal, Metalloid
Property | Metal | Nonmetal | Metalloid |
|---|---|---|---|
Appearance | Shiny | Dull | Intermediate |
Conductivity | Good | Poor | Moderate |
Malleability | Yes | No | Some |
State at Room Temp | Solid (except Hg) | Solid/gas | Solid |
Chemistry Link to Health: Elements Essential to Health
Major and Trace Elements in the Human Body
About 20 elements are essential for human health.
Four elements (O, C, H, N) make up 96% of body mass.
Macrominerals: Ca, P, K, Cl, S, Na, Mg—important for bones, heart, nerves, and metabolism.
Microminerals (trace elements): Present in small amounts, often as transition elements (e.g., Fe, Zn, Cu, I).
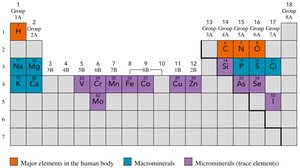
The Atom
Structure of the Atom
An atom consists of a dense nucleus containing protons and neutrons, surrounded by electrons in a large, mostly empty space. Atoms are the smallest units of elements that retain their identity in chemical reactions.

Dalton’s Atomic Theory
All matter is composed of atoms.
Atoms of the same element are identical; atoms of different elements are different.
Atoms combine in fixed ratios to form compounds.
Chemical reactions involve rearrangement of atoms; atoms are not created or destroyed.

Subatomic Particles
Proton (p+): Positive charge, located in the nucleus.
Neutron (n0): No charge, located in the nucleus.
Electron (e-): Negative charge, located outside the nucleus.
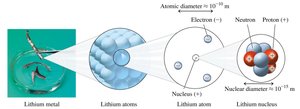
Electrical Charges in Atoms
Like charges repel; unlike charges attract.
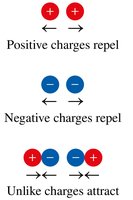
Discovery of Subatomic Particles
J. J. Thomson: Discovered the electron using cathode ray experiments; proposed the "plum-pudding" model of the atom.

Ernest Rutherford: Gold foil experiment showed that atoms have a small, dense, positively charged nucleus.
Atomic Mass Unit (amu)
1 amu is defined as one-twelfth the mass of a carbon-12 atom.
Proton mass ≈ 1 amu; neutron mass ≈ 1 amu; electron mass ≈ 0.00055 amu.
Atomic Number and Mass Number
Atomic Number (Z)
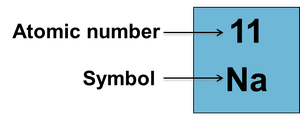
The atomic number is the number of protons in the nucleus of an atom.
It defines the identity of the element and is unique for each element.
Atomic number appears above the element symbol in the periodic table.
Mass Number (A)
The mass number is the sum of protons and neutrons in the nucleus.
Mass number = number of protons + number of neutrons
It is not shown in the periodic table because it applies to specific isotopes of an element.
Calculating Neutrons
Number of neutrons = mass number – atomic number
Example: Potassium (K) with atomic number 19 and mass number 39 has 20 neutrons.
Atoms Are Neutral
In a neutral atom, the number of protons equals the number of electrons.
This balance ensures the atom has no overall electrical charge.
Summary Table: Subatomic Particles
Particle | Symbol | Charge | Location | Relative Mass (amu) |
|---|---|---|---|---|
Proton | p+ | +1 | Nucleus | 1.007 |
Neutron | n0 | 0 | Nucleus | 1.008 |
Electron | e- | -1 | Outside nucleus | 0.00055 |
Key Equations
Number of protons = atomic number
Number of neutrons = mass number – atomic number
Number of electrons (in a neutral atom) = number of protons
Conclusion
Understanding atoms and elements, their classification in the periodic table, and their roles in biological systems is fundamental to the study of chemistry. Mastery of these concepts provides the foundation for exploring chemical reactions, compounds, and the molecular basis of life.
