 Back
BackAtoms and Elements: Structure, Properties, and the Periodic Table
Study Guide - Smart Notes

Atoms and Elements
Elements and Symbols
Elements are the fundamental substances from which all matter is composed. Each element is represented by a unique chemical symbol, typically derived from its English or Latin name. The periodic table organizes these elements based on their atomic structure and properties.
Element: A pure substance consisting of only one type of atom.
Chemical Symbol: One or two letters representing an element (e.g., Na for sodium).
Example: Aluminium foil is made entirely of aluminium atoms.
The Atom
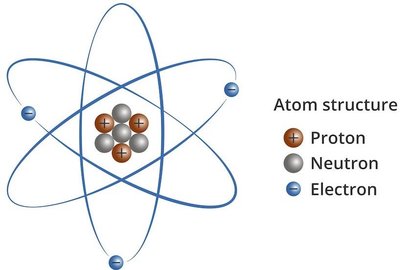
An atom is the smallest unit of an element that retains its chemical properties. Atoms are composed of three primary subatomic particles: protons, neutrons, and electrons.
Proton: Positively charged particle found in the nucleus.
Neutron: Uncharged particle found in the nucleus.
Electron: Negatively charged particle found outside the nucleus.
Nucleus: The small, dense, positively charged center of the atom containing protons and neutrons.
Electron Cloud: The region surrounding the nucleus where electrons move rapidly.
Properties of Subatomic Particles
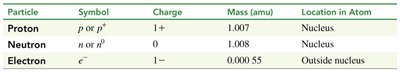
Protons, neutrons, and electrons have distinct properties, including charge, mass, and location within the atom. Their masses are measured in atomic mass units (amu), a relative unit.
Atomic Mass Unit (amu):
Proton: Charge = +1, Mass ≈ 1.007 amu, Location = Nucleus
Neutron: Charge = 0, Mass ≈ 1.008 amu, Location = Nucleus
Electron: Charge = -1, Mass ≈ 0.00055 amu, Location = Outside nucleus
Particle | Symbol | Charge | Mass (amu) | Location in Atom |
|---|---|---|---|---|
Proton | p or p+ | 1+ | 1.007 | Nucleus |
Neutron | n or n0 | 0 | 1.008 | Nucleus |
Electron | e- | 1− | 0.00055 | Outside nucleus |
Symbolic Representation of an Element
Elements are represented symbolically using their atomic number (Z), mass number (A), and chemical symbol (X):
Atomic Number (Z): Number of protons in the atom.
Mass Number (A): Total number of protons and neutrons.
Neutral Atom: Number of electrons equals number of protons.
Symbolic notation:
Example: (Sodium: 11 protons, 12 neutrons, 11 electrons)
Determining Atomic Composition
To determine the number of protons, neutrons, and electrons in an atom:
Protons: Equal to atomic number (Z).
Neutrons:
Electrons: Equal to protons in a neutral atom.
Example: Boron (): 5 protons, 6 neutrons, 5 electrons.
Example: Iron (): 26 protons, 29 neutrons, 26 electrons.
Example: Zinc (): 30 protons, 35 neutrons, 30 electrons.
Example: Gold (): 79 protons, 118 neutrons, 79 electrons.
Isotopes and Atomic Mass
Isotopes are atoms of the same element with different mass numbers due to varying numbers of neutrons. They have identical chemical properties but may differ in physical properties, such as radioactivity.
Isotope: Atoms with same atomic number (Z) but different mass number (A).
Example: Carbon isotopes: , ,
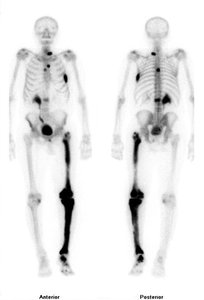
Radioactive Isotopes: Emit energy and particles; used in medical diagnostics (e.g., Tc-99 for heart and bone scans).
Stable Isotopes: Do not emit radiation; 80 elements have at least one stable isotope.
Periodic Law and the Periodic Table
The periodic law states that the physical and chemical properties of elements are periodic functions of their atomic numbers. The periodic table arranges elements in rows (periods) and columns (groups) based on increasing atomic number.
Period: Horizontal row in the periodic table.
Group: Vertical column; elements in the same group have similar properties.
Example: Group 1A (alkali metals): Lithium, sodium, potassium.
Example: Group 2A (alkaline earth metals): Beryllium, magnesium, calcium.
Example: Group 7A (halogens): Chlorine, bromine, iodine.
Example: Group 8A (noble gases): Least reactive elements.
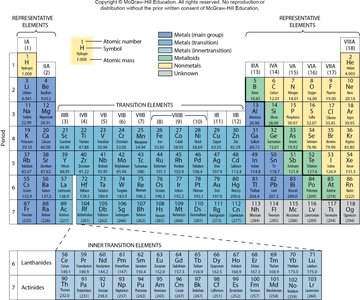
Group Numbers of Selected Elements
Identifying the group number helps predict the chemical behavior of elements:
K (Potassium): Group 1A
Si (Silicon): Group 4A
B (Boron): Group 3A
Additional info: These notes expand on the original slides by providing definitions, examples, and context for each concept, ensuring clarity and completeness for exam preparation.
