 Back
BackAtoms and Elements: Structure, Properties, and the Periodic Table
Study Guide - Smart Notes

Atoms and Elements
Introduction to Atoms and Elements
Atoms are the fundamental building blocks of matter, and elements are pure substances composed of only one type of atom. Understanding the structure and properties of atoms is essential for studying chemistry, as it forms the basis for explaining chemical behavior and the organization of the periodic table.
Elements and Symbols
Each element is represented by a unique chemical symbol, typically derived from its English or Latin name (e.g., H for hydrogen, Na for sodium). The periodic table organizes all known elements according to their atomic number and properties.
The Atom: Structure and Composition
An atom is the smallest unit of an element that retains the chemical properties of that element. Atoms are composed of three primary subatomic particles:
Protons: Positively charged particles located in the nucleus.
Neutrons: Uncharged (neutral) particles also found in the nucleus.
Electrons: Negatively charged particles that move rapidly in the space outside the nucleus.
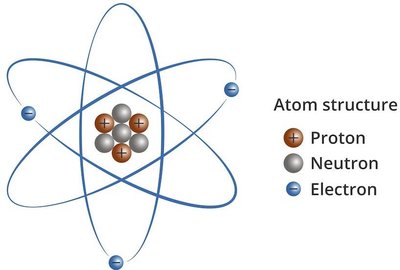
The nucleus is a small, dense, positively charged region at the center of the atom, containing protons and neutrons. Electrons occupy the space outside the nucleus.
Properties of Subatomic Particles
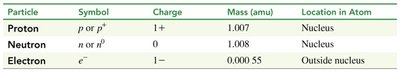
The three basic subatomic particles differ in mass, charge, and location within the atom. Their properties are summarized below:
Particle | Symbol | Charge | Mass (amu) | Location in Atom |
|---|---|---|---|---|
Proton | p or p+ | 1+ | 1.007 | Nucleus |
Neutron | n or n0 | 0 | 1.008 | Nucleus |
Electron | e- | 1− | 0.000 55 | Outside nucleus |
Atomic mass units (amu) are used to express the very small masses of these particles, where 1 amu = grams.
Symbolic Representation of Elements
Elements are represented using the notation:
A = Mass number (number of protons + number of neutrons)
Z = Atomic number (number of protons)
X = Chemical symbol of the element
For a neutral atom, the number of electrons equals the number of protons.
Example: For (sodium):
Protons: 11
Neutrons: 23 − 11 = 12
Electrons: 11
Determining the Composition of an Atom
To find the number of protons, neutrons, and electrons in an atom:
Protons = Atomic number (Z)
Neutrons = Mass number (A) − Atomic number (Z)
Electrons = Number of protons (for a neutral atom)
Example: For (boron):
Protons: 5
Neutrons: 11 − 5 = 6
Electrons: 5
Example: For iron (Fe), A = 55, Z = 26:
Neutrons: 55 − 26 = 29
Example: For zinc (Zn), A = 65, Z = 30:
Protons: 30
Neutrons: 65 − 30 = 35
Electrons: 30
Example: For gold (Au), :
Neutrons: 197 − 79 = 118
Isotopes
Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons, resulting in different mass numbers. Isotopes have identical chemical properties but may differ in nuclear stability.
Same atomic number (Z), different mass number (A)
Some isotopes are radioactive and can emit energy and particles
Radioactive isotopes are used in medicine (e.g., Tc-99 for diagnostic imaging)
Example: Isotopes of carbon:
: 6 protons, 6 neutrons
: 6 protons, 7 neutrons
: 6 protons, 8 neutrons
Isotopes of the same element have the same atomic number but different mass numbers.
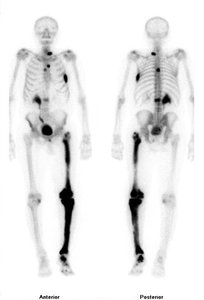
Applications: Radioactive isotopes are used to trace biochemical activity and diagnose diseases (e.g., Tc-99 scans for bone cancer).
The Periodic Table and Periodic Law
The Periodic Law states that the physical and chemical properties of elements are periodic functions of their atomic numbers. The periodic table arranges elements in order of increasing atomic number into horizontal rows (periods) and vertical columns (groups or families).
Elements in the same group have similar chemical properties.
Group 1A: Alkali metals (e.g., lithium, sodium, potassium)
Group 2A: Alkaline earth metals (e.g., beryllium, magnesium, calcium)
Group 7A: Halogens (e.g., chlorine, bromine, iodine)
Group 8A: Noble gases (least reactive elements)
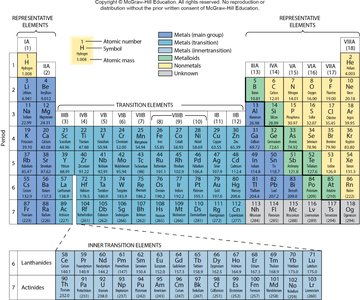
Example: Group numbers for selected elements:
K (potassium): Group 1A
Si (silicon): Group 4A
B (boron): Group 3A
Additional info: The periodic table also distinguishes between metals, nonmetals, and metalloids, and provides information about atomic mass and electron configurations.
