Back
BackAtoms and Elements: Structure, Properties, and Periodic Trends
Study Guide - Smart Notes

Atoms and Elements
Introduction
Understanding atoms and elements is fundamental to chemistry. Atoms are the basic building blocks of matter, and elements are pure substances composed of only one type of atom. The arrangement and properties of atoms determine the behavior of elements and their compounds.
Classification of Matter
Pure Substances vs. Mixtures
Pure substances consist of only one type of particle and include elements and compounds. Mixtures contain two or more substances physically combined.
Element: A pure substance that cannot be broken down into simpler substances.
Compound: A pure substance composed of two or more elements chemically combined.
Mixture: A combination of two or more substances that retain their individual properties.
Elements and Their Symbols
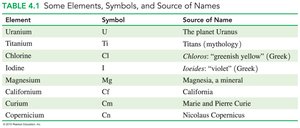
Element Names and Sources
Element names are derived from various sources, including planets, mythology, minerals, colors, geographic locations, and famous people.
Element | Symbol | Source of Name |
|---|---|---|
Uranium | U | The planet Uranus |
Titanium | Ti | Titans (mythology) |
Chlorine | Cl | Chloros: "greenish yellow" (Greek) |
Iodine | I | Iodes: "violet" (Greek) |
Magnesium | Mg | Magnesia, a mineral |
Californium | Cf | California |
Curium | Cm | Marie and Pierre Curie |
Copernicium | Cn | Nicolas Copernicus |
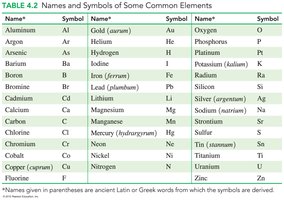
Chemical Symbols
Chemical symbols are one- or two-letter abbreviations for element names, often derived from English or Latin names.
Name | Symbol | Name | Symbol |
|---|---|---|---|
Aluminum | Al | Gold | Au |
Carbon | C | Oxygen | O |
Chlorine | Cl | Silver | Ag |
Iron | Fe | Hydrogen | H |
Mercury | Hg | Platinum | Pt |
Potassium | K | Sodium | Na |
Calcium | Ca | Zinc | Zn |
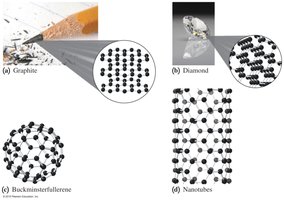
Examples of Elements
Carbon (C): Exists in several forms, including graphite, diamond, buckminsterfullerene, and nanotubes.
Mercury (Hg): A liquid metal at room temperature, toxic to humans.

The Periodic Table
Organization and Structure
The periodic table arranges elements by increasing atomic number and groups elements with similar properties into columns (groups) and rows (periods).
Groups: Vertical columns with similar properties.
Periods: Horizontal rows numbered 1–7.
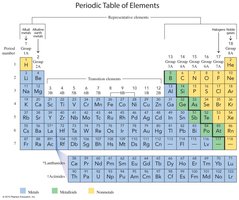
Group Names and Types
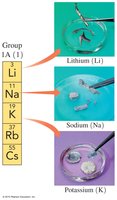
Alkali Metals (Group 1A): Highly reactive metals including Li, Na, K, Rb, Cs.
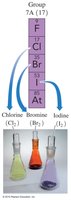
Halogens (Group 7A): Reactive nonmetals including F, Cl, Br, I.
Representative Elements: Groups 1A–8A.
Transition Elements: Groups 3B–12B.
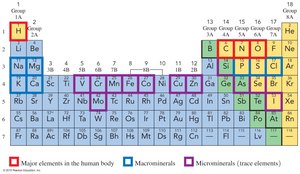
Elements Essential to Life
Twenty elements are essential for human health, with oxygen, carbon, hydrogen, and nitrogen making up 96% of body mass. Macrominerals such as Ca, P, K, Cl, S, Na, and Mg are vital for physiological functions.
Structure of the Atom
Subatomic Particles
Atoms consist of protons, neutrons, and electrons. Protons and neutrons are located in the nucleus, while electrons occupy the space around the nucleus.
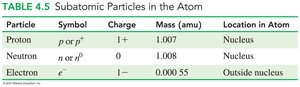
Particle | Symbol | Charge | Mass (amu) | Location |
|---|---|---|---|---|
Proton | p or p+ | 1+ | 1.007 | Nucleus |
Neutron | n or n0 | 0 | 1.008 | Nucleus |
Electron | e- | 1- | 0.00055 | Outside nucleus |
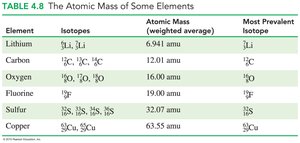
Dalton’s Atomic Theory
Atoms are tiny particles of matter.
Atoms of the same element are similar; atoms of different elements are different.
Atoms combine to form compounds.
Atoms are rearranged in chemical reactions but are not created or destroyed.
Electrical Charges in Atoms
Protons are positively charged, electrons are negatively charged, and neutrons are neutral. Like charges repel, unlike charges attract.

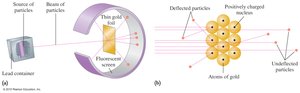
Rutherford’s Gold Foil Experiment
This experiment demonstrated the existence of a small, dense, positively charged nucleus in the atom.
Atomic Mass Unit (amu)
One atomic mass unit is defined as one-twelfth the mass of a carbon-12 atom. Protons and neutrons have masses close to 1 amu; electrons are much lighter.
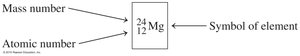
Atomic Number and Mass Number
Definitions and Calculations
Atomic Number: Number of protons in an atom; unique to each element.
Mass Number: Sum of protons and neutrons in the nucleus.
Neutral Atoms: Number of protons equals number of electrons.
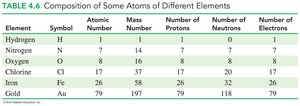
Element | Symbol | Atomic Number | Mass Number | Protons | Neutrons | Electrons |
|---|---|---|---|---|---|---|
Hydrogen | H | 1 | 1 | 1 | 0 | 1 |
Nitrogen | N | 7 | 14 | 7 | 7 | 7 |
Oxygen | O | 8 | 16 | 8 | 8 | 8 |
Chlorine | Cl | 17 | 37 | 17 | 20 | 17 |
Iron | Fe | 26 | 58 | 26 | 32 | 26 |
Gold | Au | 79 | 197 | 79 | 118 | 79 |
Isotopes and Atomic Mass
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons and thus different mass numbers. They are represented by atomic symbols showing mass and atomic numbers.
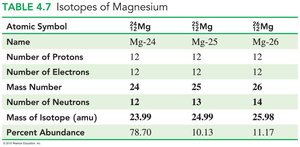
Isotopes of Magnesium
Atomic Symbol | Name | Protons | Electrons | Mass Number | Neutrons | Mass (amu) | Percent Abundance |
|---|---|---|---|---|---|---|---|
$^{24}_{12}\text{Mg}$ | Mg-24 | 12 | 12 | 24 | 12 | 23.99 | 78.70 |
$^{25}_{12}\text{Mg}$ | Mg-25 | 12 | 12 | 25 | 13 | 24.99 | 10.13 |
$^{26}_{12}\text{Mg}$ | Mg-26 | 12 | 12 | 26 | 14 | 25.98 | 11.17 |
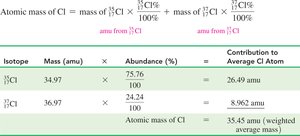
Calculating Atomic Mass
The atomic mass of an element is the weighted average of the masses of its naturally occurring isotopes, based on their percent abundance.
Formula: $\text{Atomic mass} = \sum (\text{isotope mass} \times \text{percent abundance})$
Isotope | Mass (amu) | Abundance (%) | Contribution (amu) |
|---|---|---|---|
$^{35}\text{Cl}$ | 34.97 | 75.76 | 26.49 |
$^{37}\text{Cl}$ | 36.97 | 24.24 | 8.962 |
Atomic mass of Cl (weighted average mass) | 35.45 | ||
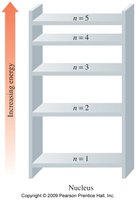
Electron Energy Levels
Energy Levels, Subshells, and Orbitals
Electrons occupy energy levels (shells) around the nucleus, which are divided into subshells and orbitals. Each energy level can hold a maximum number of electrons given by $2n^2$.
Energy Levels: n = 1, 2, 3, 4, ...
Subshells: Regions within shells with electrons of similar energy.
Orbitals: Regions within subshells where electrons are most likely found; each orbital holds up to 2 electrons.
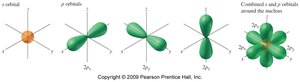
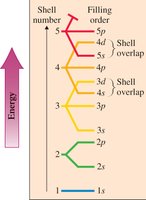
Electron Configurations and Orbital Diagrams
Electron configuration describes the distribution of electrons among subshells. Orbital diagrams show electron arrangement in orbitals.
Example: Oxygen: 1s22s22p4
Example: Sulfur: 1s22s22p63s23p4
Example: Calcium: 1s22s22p63s23p64s2
Periodic Trends
Valence Electrons and Group Number
Valence electrons are the electrons in the outermost energy level. The group number for representative elements indicates the number of valence electrons.
Group Number | Number of Valence Electrons |
|---|---|
1A (1) | 1 |
2A (2) | 2 |
3A (13) | 3 |
4A (14) | 4 |
5A (15) | 5 |
6A (16) | 6 |
7A (17) | 7 |
8A (18) | 8 |
Electron-Dot Symbols (Lewis Structures)
Electron-dot symbols represent valence electrons as dots around the element symbol.
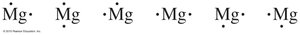

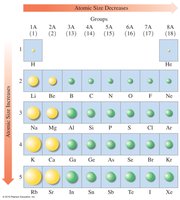
Atomic Size
Atomic size increases down a group and decreases across a period due to changes in energy levels and nuclear charge.
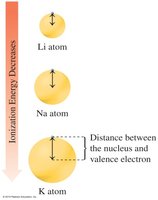
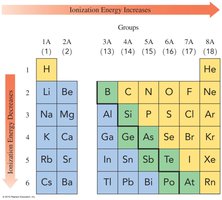
Ionization Energy
Ionization energy is the energy required to remove an electron. It decreases down a group and increases across a period.
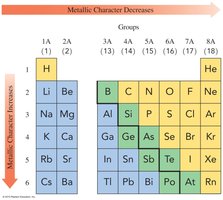
Metallic Character
Metallic character refers to the tendency of an element to lose electrons. It increases down a group and decreases across a period.
Summary of Periodic Trends
Periodic Property | Top to Bottom (Group) | Left to Right (Period) |
|---|---|---|
Valence Electrons | Remains the same | Increases |
Atomic Size | Increases | Decreases |
Ionization Energy | Decreases | Increases |
Metallic Character | Increases | Decreases |

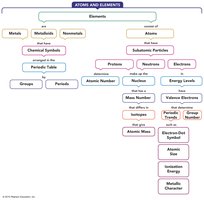
Concept Map
The concept map below summarizes the relationships between elements, atomic structure, periodic table organization, and periodic properties.