Back
BackAtoms and Elements: Structure, Properties, and Isotopes
Study Guide - Smart Notes

Atoms and Elements
Energy Content in Food
Food contains macronutrients—fat, carbohydrates, and protein—that provide energy measured in Calories (Cal). Each macronutrient has a specific caloric value per gram.
Fat: 9 Cal/g
Carbohydrate: 4 Cal/g
Protein: 4 Cal/g
Example: A cheeseburger with 19 g fat, 20 g carbohydrate, and 28 g protein contains: Fat: Cal Carbohydrate: Cal Protein: Cal Total: Cal
Types of Energy
Energy exists in various forms. The energy of motion is called kinetic energy.
Kinetic Energy: Energy due to motion.
Potential Energy: Stored energy due to position.
Chemical Energy: Energy stored in chemical bonds.
Heat Energy: Energy transferred due to temperature difference.
Wave Energy: Energy carried by waves (e.g., light, sound).
Atoms and Elements
What is an Atom?
All matter is composed of tiny particles called atoms. An atom is the smallest unit of an element that retains the chemical properties of that element.
Element: A pure substance made of only one kind of atom.
Atom: Smallest particle of an element.
Example: Aluminium foil consists of atoms of aluminium.
Composition of an Atom
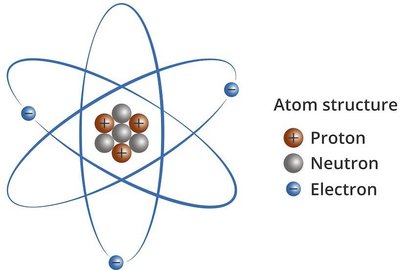
Atoms are made up of three primary subatomic particles: protons, neutrons, and electrons.
Protons: Positively charged particles (+), located in the nucleus.
Neutrons: Uncharged particles, located in the nucleus.
Electrons: Negatively charged particles (-), located outside the nucleus.
Nucleus: Small, dense, positively charged region at the center of the atom.
Electrons: Move rapidly in a large volume of space outside the nucleus.
Properties of Subatomic Particles
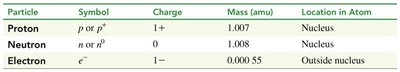
Subatomic particles have distinct properties, including charge, mass, and location within the atom. Their masses are measured in atomic mass units (amu).
Atomic Mass Unit (amu): grams
Electron Mass: amu (much smaller than protons and neutrons)
Particle | Symbol | Charge | Mass (amu) | Location in Atom |
|---|---|---|---|---|
Proton | p or p+ | 1+ | 1.007 | Nucleus |
Neutron | n or n0 | 0 | 1.008 | Nucleus |
Electron | e- | 1− | 0.00055 | Outside nucleus |
Symbolic Representation of Elements
Elements are represented symbolically using their atomic number (Z), mass number (A), and chemical symbol (X).
Mass Number (A): Number of protons + number of neutrons
Atomic Number (Z): Number of protons in the atom
Element Symbol (X): One or two-letter abbreviation for the element
Example: (Sodium): 11 protons, 23 mass number

Determining Atomic Composition
To determine the number of protons, neutrons, and electrons in an atom, use the atomic and mass numbers.
Protons: Equal to atomic number (Z)
Neutrons: Mass number (A) minus atomic number (Z)
Electrons: Equal to protons in a neutral atom
Example: (Carbon): 6 protons, 6 neutrons, 6 electrons
Example: (Argon): 18 protons, 22 neutrons, 18 electrons
Example: (Boron): 5 protons, 6 neutrons, 5 electrons
Example: (Iron): 26 protons, 29 neutrons, 26 electrons
Example: (Zinc): 30 protons, 35 neutrons, 30 electrons
Example: (Gold): 79 protons, 118 neutrons, 79 electrons
Isotopes
Isotopes are atoms of the same element with different mass numbers due to varying numbers of neutrons. Isotopes have identical chemical properties but may differ in nuclear stability.
Same atomic number (Z), different mass number (A)
Example: Carbon isotopes: , ,
Radioactive Isotopes: Emit energy and particles; used in medical diagnostics (e.g., Tc-99 for heart and bone scans)
Stable Isotopes: Do not emit radiation; 80 elements have at least one stable isotope
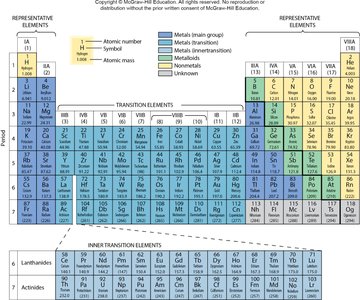
Periodic Law and the Periodic Table
The Periodic Law states that the physical and chemical properties of elements are periodic functions of their atomic numbers. The periodic table arranges elements by increasing atomic number into periods (rows) and groups (columns).
Periods: Horizontal rows
Groups: Vertical columns
Classification: Metals, nonmetals, metalloids, transition elements, lanthanides, actinides