 Back
BackAtoms and Radioactivity: Structure, Properties, and Medical Applications
Study Guide - Smart Notes

Atoms and Their Components
Subatomic Particles and Atomic Structure
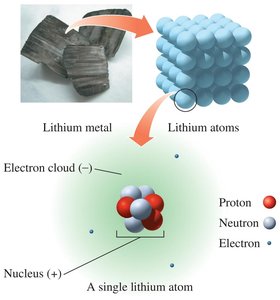
Atoms are the fundamental units of matter, composed of three main subatomic particles: protons, neutrons, and electrons. The arrangement and properties of these particles determine the characteristics of each atom.
Protons: Positively charged particles located in the nucleus.
Neutrons: Neutral particles also found in the nucleus.
Electrons: Negatively charged particles that move in the electron cloud surrounding the nucleus.
Atoms are electrically neutral because the number of protons equals the number of electrons.
Most of the atomic mass is concentrated in the nucleus; electrons contribute negligibly to mass.
Atomic mass unit (amu) is used to express the mass of subatomic particles: 1 amu for protons and neutrons, 0 amu for electrons.
"
Atomic Number and Mass Number
Identifying Elements and Calculating Subatomic Particles
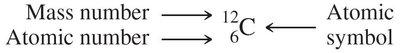
The atomic number is the number of protons in an atom and defines the element. The mass number is the sum of protons and neutrons.
Atomic number is found above each element in the periodic table.
Mass number = number of protons + number of neutrons.
Symbolic notation:
To determine subatomic particles: Find the element, use atomic number for protons/electrons, use mass number for neutrons.
⑥ .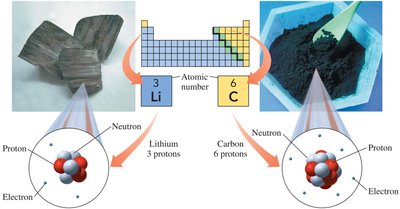
Isotopes and Atomic Mass
Isotopes and Average Atomic Mass
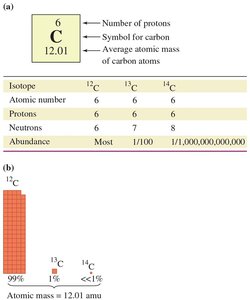
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers. The atomic mass of an element is the weighted average of all its naturally occurring isotopes.
Isotopes are represented by symbolic notation or by stating the mass number (e.g., carbon-12, carbon-13).
Atomic mass depends on the abundance and mass of each isotope.
Example: Chlorine has two stable isotopes, Cl-35 and Cl-37, with different abundances and masses.
Radioactivity and Radioisotopes
Types of Radiation and Biological Effects
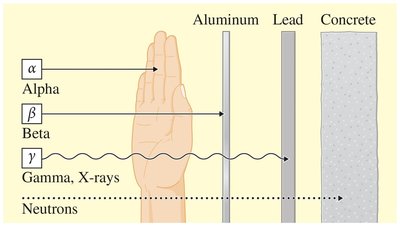
Radioactivity is the spontaneous emission of energy from the nucleus of unstable atoms. Radioisotopes are isotopes that emit radiation to become stable. There are several forms of nuclear radiation:
Alpha (α) particles: Helium nuclei, 2+ charge, low penetration.
Beta (β) particles: High-energy electrons, 1− charge, moderate penetration.
Gamma (γ) rays: High-energy electromagnetic radiation, no charge, high penetration.
Positrons: Positive charge, same mass as beta particles.
Neutrons: No charge, high penetration.
Ionizing radiation can damage living cells by ejecting electrons, potentially causing cancer. Protective measures include shielding and monitoring exposure.
Nuclear Equations and Radioactive Decay
Writing and Balancing Nuclear Equations
Nuclear decay equations show the transformation of a radioactive nucleus into a new nucleus and radiation. The atomic and mass numbers must balance on both sides of the equation.
Alpha decay:
Beta decay: A neutron becomes a proton, emitting a beta particle.
Gamma decay: Only energy is emitted; atomic and mass numbers remain unchanged.
Radioisotopes can be produced by bombarding stable isotopes with particles.
Radiation Units and Half-Lives
Measuring Radioactivity and Decay Rates
The curie (Ci) and becquerel (Bq) are units for measuring radioactivity. The half-life is the time required for half of the radioactive atoms in a sample to decay.
Physical half-life: Time for 50% decay of a radioisotope.
Biological half-life: Time for elimination from the body.
Effective half-life: Combination of physical and biological half-lives.
Equation for remaining isotope: where = number of half-lives.
Medical Applications for Radioisotopes
Diagnosis and Treatment Using Radioisotopes
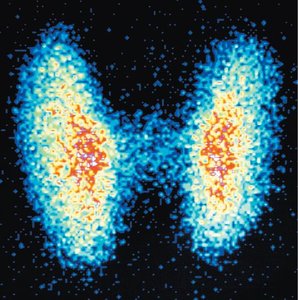
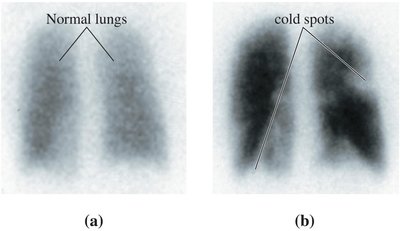
Radioisotopes are used in medicine for imaging and treatment. They concentrate in specific tissues, allowing for diagnostic imaging or targeted therapy.
Diagnostic use: Small doses of radioisotopes (tracers) with short half-lives are used to image organs (e.g., technetium-99m for lung scans).
Therapeutic use: Higher doses are used to destroy diseased tissues (e.g., iodine-131 for thyroid cancer)..
External beam radiation therapy uses gamma rays from cobalt-60.
Brachytherapy involves implanting radioactive seeds in tumors.
PET scans use positron-emitting isotopes (e.g., fluorine-18) to detect functional abnormalities.

Summary Table: Types of Radiation and Their Properties
Type | Symbol | Charge | Penetration | Shielding |
|---|---|---|---|---|
Alpha (α) | 2+ | Low (stops at skin) | Paper, clothing | |
Beta (β) | 1− | Moderate, (stops at skin layer) | Plastic, aluminum, gloves | |
Gamma (γ) | 0 | High (fully penetrates body) | Lead, concrete | |
Neutron (n) | 0 | Very high | Concrete, water |
