 Back
BackAtoms and the Periodic Table: Structured Study Notes
Study Guide - Smart Notes

Atomic Theory and Structure of Atoms
Dalton's Atomic Theory
The foundation of modern chemistry rests on Dalton's atomic theory, which describes the nature of matter and the behavior of atoms. Dalton proposed four key assumptions:
All matter is composed of atoms, the smallest identifiable unit of an element.
Atoms of a given element differ from those of other elements.
Chemical compounds consist of atoms combined in specific ratios; only whole atoms can combine.
Chemical reactions change only the way atoms are combined in compounds, not the atoms themselves.
Modern evidence for atomic theory includes the use of the Scanning Tunneling Microscope (STM), which allows visualization of atoms on surfaces.

Subatomic Particles
Atoms are composed of three fundamental subatomic particles:
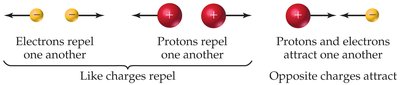
Protons: Positively charged, located in the nucleus.
Neutrons: Electrically neutral, located in the nucleus.
Electrons: Negatively charged, move rapidly around the nucleus.
Like charges repel, while opposite charges attract, which is fundamental to atomic structure.
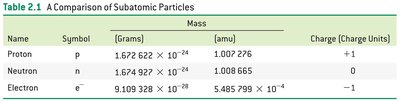
Comparison of Subatomic Particles
The properties of protons, neutrons, and electrons are summarized below:
Name | Symbol | Mass (grams) | Mass (amu) | Charge |
|---|---|---|---|---|
Proton | p | 1.672622 × 10-24 | 1.007276 | +1 |
Neutron | n | 1.674927 × 10-24 | 1.008665 | 0 |
Electron | e- | 9.109328 × 10-28 | 5.485799 × 10-4 | -1 |
Nucleus and Atomic Structure
The nucleus contains protons and neutrons, held together by the nuclear strong force. Electrons occupy the space around the nucleus.
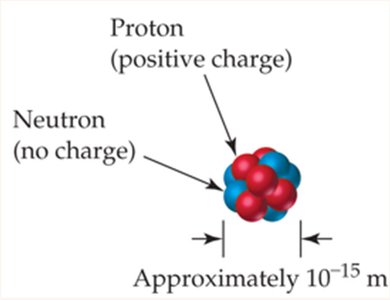
The nucleus is extremely small compared to the overall size of the atom, analogous to a pea in a stadium.


Elements and Atomic Number
Atomic Number and Mass Number
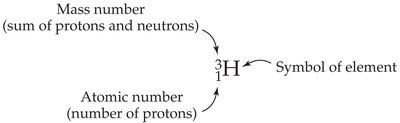
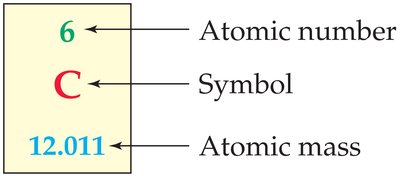
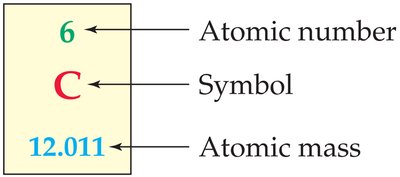
The atomic number (Z) is the number of protons in an atom and determines the element's identity. The mass number (A) is the sum of protons and neutrons.
For neutral atoms, the number of electrons equals the number of protons.
Atoms are electrically neutral when protons and electrons are equal.
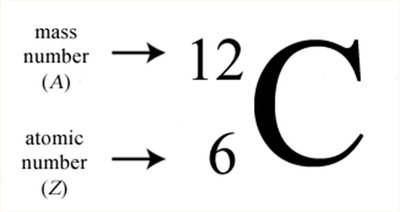
Isotopes and Atomic Mass
Definition and Representation of Isotopes
Isotopes are atoms of the same element (same atomic number) but different mass numbers due to varying numbers of neutrons.
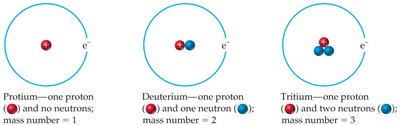
Isotopes are represented with mass number as a superscript and atomic number as a subscript before the element symbol.
Atomic Mass and Calculation
The atomic mass (or atomic weight) of an element is the weighted average of its naturally occurring isotopes, calculated as:
Example: Chlorine's atomic mass is calculated from its two main isotopes:
Chlorine-35: 75.77% at 34.969 amu
Chlorine-37: 24.23% at 36.966 amu
The Periodic Table
Structure and Classification
The periodic table organizes elements by increasing atomic number and groups elements with similar properties into columns (groups) and rows (periods).
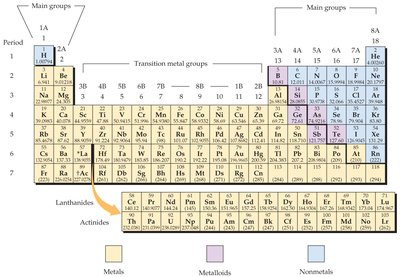
Groups and Periods
Elements in the same group (vertical column) have similar chemical properties. The main classifications are:
Main group elements (Groups 1A-8A)
Transition metals (Groups 1B-8B)
Inner transition metals (lanthanides and actinides)
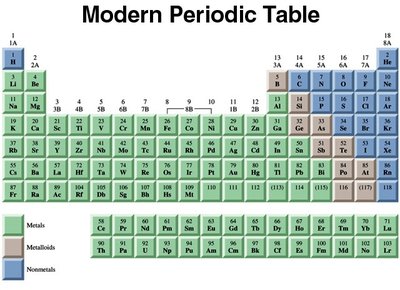
Periods (rows) contain varying numbers of elements, with lanthanides and actinides shown below the main table.


Classification by Metallic Properties
Metals: Lustrous, malleable, good conductors, mostly solids, left side of table.
Nonmetals: Poor conductors, can be gases, liquids, or solids, upper-right side.
Metalloids: Intermediate properties, zigzag band between metals and nonmetals.



Characteristics of Major Groups
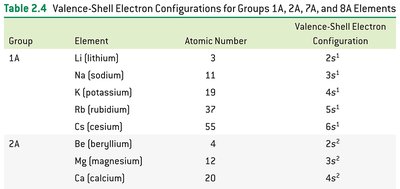
Group 1A – Alkali metals: Shiny, soft, highly reactive, never found pure in nature.
Group 2A – Alkaline earth metals: Lustrous, silvery, less reactive than alkali metals.
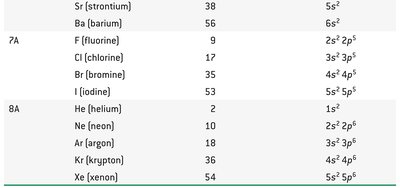
Group 7A – Halogens: Colorful, corrosive nonmetals, found only in compounds.
Group 8A – Noble gases: Colorless, chemically inert gases.
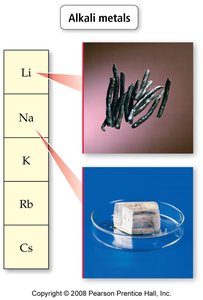
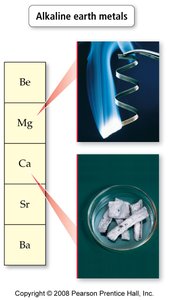

Electronic Structure of Atoms
Quantum Mechanical Model
The arrangement of electrons determines element properties. Electrons exhibit both particle and wave behavior and are restricted to quantized energy levels.

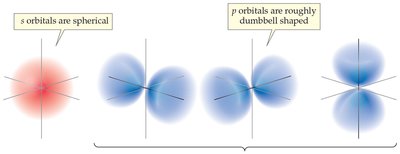
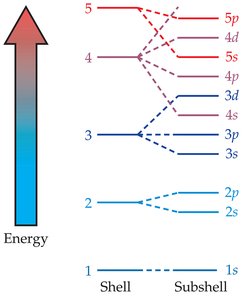
Shells, Subshells, and Orbitals
Electrons are organized into shells (energy levels), subshells (s, p, d, f), and orbitals. Each orbital can hold two electrons with opposite spins.
Shell: Distance from nucleus, determines size and energy.
Subshell: Shape of orbital (s, p, d, f).
Orbital: Spatial orientation, maximum two electrons.
Electron Distribution and Capacity
Shell Number | Subshell Designation | Number of Orbitals | Number of Electrons | Total Electron Capacity |
|---|---|---|---|---|
1 | s | 1 | 2 | 2 |
2 | s, p | 1, 3 | 2, 6 | 8 |
3 | s, p, d | 1, 3, 5 | 2, 6, 10 | 18 |
4 | s, p, d, f | 1, 3, 5, 7 | 2, 6, 10, 14 | 32 |
Electron Configurations
Rules for Electron Configuration
Aufbau Principle: Electrons fill lowest energy orbitals first.
Pauli Exclusion Principle: Each orbital holds two electrons of opposite spin.
Hund's Rule: Orbitals in the same subshell are half-filled before any is completely filled.
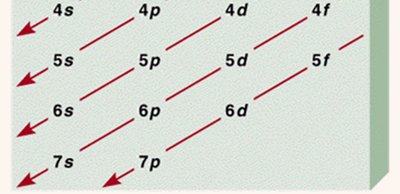
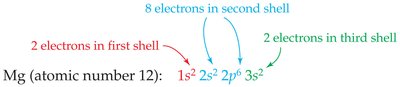
Writing Electron Configurations
Three methods:
Complete configuration: Lists all orbitals and electrons.
Abbreviated configuration: Uses noble gas core plus remaining electrons.
Orbital diagram: Boxes for orbitals, arrows for electrons.
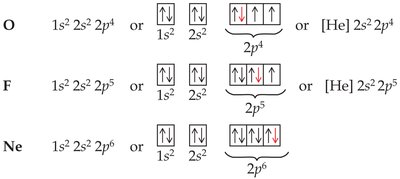
Example for oxygen:
Complete: 1s22s22p4
Abbreviated: [He] 2s22p4
Orbital diagram:
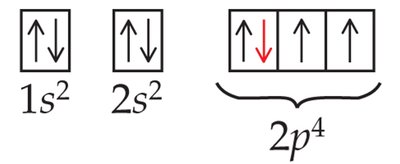
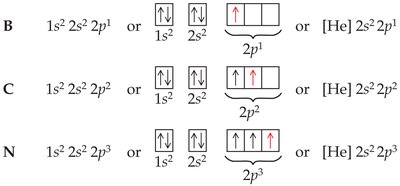
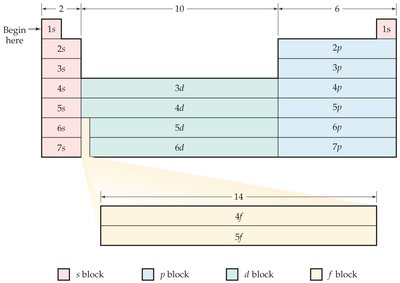
Electron Configurations and the Periodic Table
Valence Electrons and Periodic Table Blocks
Elements in the same group have similar valence electron configurations, which determine their chemical properties. The periodic table is divided into blocks (s, p, d, f) based on the subshell filled last.
Example: Group IIA elements all have two electrons in an s orbital in their valence shells (ns2).
Electron-Dot Symbols
Lewis Dot Symbols
Electron-dot (Lewis) symbols represent the valence electrons of main group elements as dots around the atomic symbol.
Summary and Study Tips
Review student learning outcomes and practice exercises.
Familiarize yourself with the periodic table and electron configurations.
Practice writing electron configurations and Lewis dot symbols for various elements.
