 Back
BackAtoms, Elements, and Chemical Bonding: Foundations of Chemistry
Study Guide - Smart Notes

Atoms and the Periodic Table
Matter and Chemical Elements
Matter is anything that takes up space and has mass, including organisms, rocks, and oceans. All matter consists of at least one chemical element, which is a pure substance made of only one type of atom. Atoms are the smallest unit of an element and therefore the smallest unit of matter. Both living and non-living matter are composed of atoms.
Matter: Physical substance occupying space and possessing mass.
Chemical Element: Pure substance consisting of only one type of atom.
Atom: Smallest unit of an element, fundamental to all matter.
Example: Atoms are the smallest units of matter.
Atomic Structure and Subatomic Particles
Atoms are composed of three subatomic particles, each with distinct charge, mass, and location within the atom:
Proton: Positively charged, located in the nucleus, mass of 1 atomic mass unit (AMU).
Neutron: No charge, located in the nucleus, mass of 1 AMU.
Electron: Negatively charged, located in electron shells orbiting the nucleus, mass nearly zero.
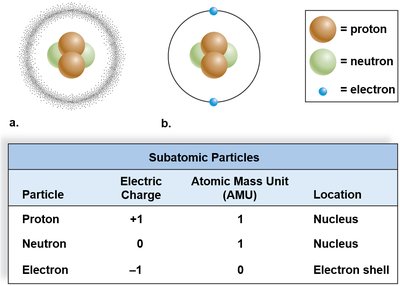
Subatomic Particle | Electric Charge | Atomic Mass Unit (AMU) | Location |
|---|---|---|---|
Proton | +1 | 1 | Nucleus |
Neutron | 0 | 1 | Nucleus |
Electron | -1 | 0 | Electron shell |
Elements of Life and the Periodic Table
Only a small subset of elements is found in living organisms. The periodic table arranges all known elements based on their chemical properties. Approximately 97% of the mass of most life forms is composed of Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, and Sulfur (CHNOPS).
Major Elements: Required for life in large amounts.
Trace Elements: Required in small amounts.
Atomic Properties
Each atom of an element has unique properties:
Atomic Number: Number of protons in the nucleus; defines the element.
Mass Number: Sum of protons and neutrons in the nucleus.
Atomic Mass: Weighted average mass of all isotopes of an element.
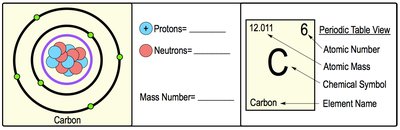
Property | Description |
|---|---|
Atomic Number | Number of protons |
Mass Number | Number of protons + neutrons |
Atomic Mass | Average mass of all isotopes |
Electron Configuration and Stability
Electron Orbitals and Energy Shells
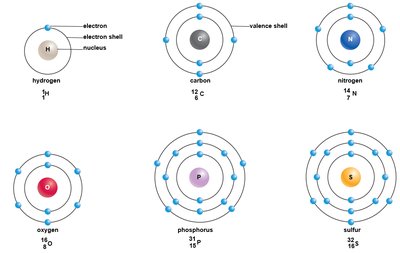
Electrons occupy three-dimensional regions called orbitals, visualized as energy shells. Shells closer to the nucleus are lower in energy, while distant shells are higher in energy. Valence electrons are found in the outermost energy shell (valence shell). The first shell holds up to 2 electrons, the second up to 8, and subsequent shells hold varying numbers.
Valence Electrons: Electrons in the outermost shell, crucial for chemical bonding.
Energy Shells: Levels where electrons reside; each shell has a maximum capacity.
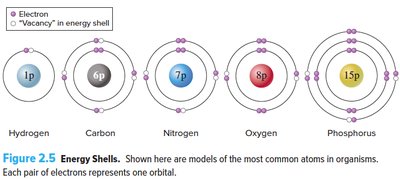
Octet Rule
The octet rule states that atoms are more stable (less reactive) when their valence shells are fully occupied, typically with 8 electrons. Atoms are more reactive when their outer shells are not full.
First shell: Holds up to 2 electrons.
Second shell: Holds up to 8 electrons.
Stability: Achieved when valence shell is full.

Isotopes and Atomic Mass
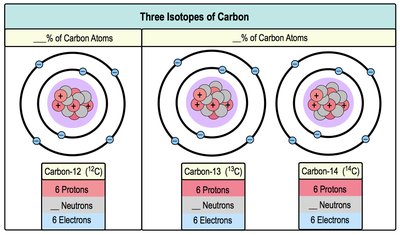
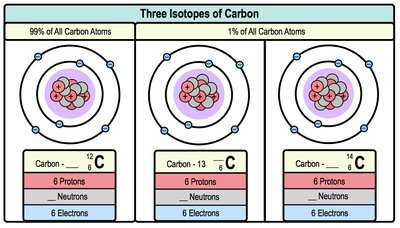
Isotopes
Isotopes are atoms of the same element that differ in the number of neutrons. They have the same atomic number (protons) but different mass numbers (protons + neutrons). Atomic mass is the average mass of all isotopes.
Example: Carbon-12, Carbon-13, and Carbon-14 are isotopes of carbon.
Isotopic Variation: Affects atomic mass but not chemical properties.

Isotope | Protons | Neutrons | Electrons |
|---|---|---|---|
Carbon-12 | 6 | 6 | 6 |
Carbon-13 | 6 | 7 | 6 |
Carbon-14 | 6 | 8 | 6 |
Radioactive Isotopes
Radioactive isotopes are unstable and break down, emitting energy as rays or particles. The half-life is the time it takes for half of the radioactive atoms in a sample to decay. Radioactive isotopes are used in medicine and radiometric dating.
Half-life: Time for half the atoms to decay.
Applications: Medical imaging, cancer treatment, fossil dating.
Introduction to Chemical Bonding
Chemical Bonds and Molecules
Chemical bonds are attractive forces between atoms, holding them together to form molecules and compounds. A molecule contains two or more chemically bound atoms, while a compound is a molecule composed of two or more different elements. Chemical formulas reveal the composition and ratio of atoms in a molecule.
Molecule: Two or more atoms chemically bonded (e.g., O2).
Compound: Molecule with two or more different elements (e.g., H2O).
Chemical Formula: Shows types and numbers of atoms (e.g., C6H12O6).
Intramolecular vs. Intermolecular Bonds
Bonds between atoms can be intramolecular (within a molecule) or intermolecular (between molecules). Intramolecular bonds are responsible for holding atoms together within a molecule, while intermolecular bonds are interactions between different molecules.
Intramolecular Bonds: Within a single molecule.
Intermolecular Bonds: Between separate molecules.
Covalent Bonds
Types of Covalent Bonds
Covalent bonds are interactions between two atoms resulting from the sharing of electrons. There are two main types:
Nonpolar Covalent Bonds: Equal sharing of electrons between atoms with similar electronegativities.
Polar Covalent Bonds: Unequal sharing of electrons due to differences in electronegativity, resulting in partial charges (δ).
Electronegativity: Measure of an atom's attraction for electrons (scale 0-4).
Noncovalent Bonds
Types of Noncovalent Bonds
Noncovalent bonds are interactions resulting from full or partial charges, without sharing of electrons. Types include ionic bonds, hydrogen bonds, van der Waals forces, and electrostatic bonds. These are generally weaker than covalent bonds but are important in biological systems.
Ionic Bonds: Attraction between oppositely charged ions.
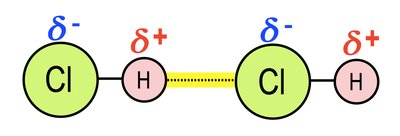
Hydrogen Bonds: Interaction between a highly electronegative atom (F, O, N) and a hydrogen atom.
Van der Waals Forces: Weak attractions due to temporary dipoles.
Ionic Bonding
Ions: Anions vs. Cations
Ions are atoms or molecules with a net electrical charge, resulting from the gain or loss of electrons. Anions are negatively charged ions (gain of electrons), while cations are positively charged ions (loss of electrons).
Anion: Negatively charged, more electrons than protons.
Cation: Positively charged, fewer electrons than protons.
Ionic Bonds
Ionic bonds are electrical attractions between oppositely charged ions. The transfer of electrons fills the valence shells of both atoms, creating charges and resulting in a stable ionic compound.
Example: Formation of NaCl from sodium (cation) and chloride (anion).
Hydrogen Bonding
Hydrogen Bonds
Hydrogen bonds are interactions between a highly electronegative atom (such as F, O, or N) and a hydrogen atom. Individually, hydrogen bonds are weak, but collectively, they can be strong and are important in biology, including the properties of water and the structure of macromolecules.
Importance: Stabilizes DNA, proteins, and contributes to water's unique properties.
