 Back
BackAtoms, Elements, and Chemical Bonding: Mini-Textbook Study Guide
Study Guide - Smart Notes

The Atom
Subatomic Particles
The atom is composed of three main subatomic particles: protons, neutrons, and electrons. Each has distinct properties and roles within the atom.
Protons (p+): Located in the nucleus, charge +1, mass 1.00727 amu. Determines the identity of the element.
Neutrons (n0): Located in the nucleus, charge 0, mass 1.00866 amu. Contributes to atomic mass and stability.
Electrons (e-): Located in the electron cloud, charge -1, mass 0.00055 amu. Responsible for chemical reactivity and bonding.

Atomic Symbols and Notation
Atomic symbols represent elements and their isotopes. The notation includes the element symbol, atomic number, and mass number.
X: Elemental symbol (e.g., Ga for Gallium)
Z: Atomic number (number of protons)
A: Mass number (protons + neutrons)
Charge: c = p - e
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons. They have identical chemical properties but different masses.
Example: Carbon-12 and Carbon-14
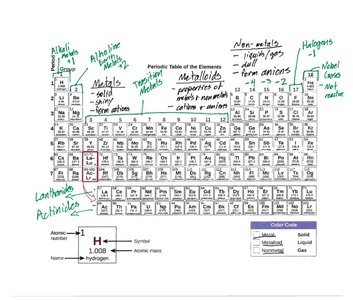
The Periodic Table
Periodic Table Structure
The periodic table organizes elements by increasing atomic number and groups elements with similar properties together.
Groups: Columns, elements share similar chemical properties.
Periods: Rows, elements have increasing atomic number.
Metals: Left and center, solid, conduct electricity.
Non-metals: Right, form anions, poor conductors.
Metalloids: Properties of both metals and non-metals.
Atomic Weight and Isotopic Mass
Atomic weight is the weighted average of the isotopic masses of an element.
Formula:
Electron Structure and Orbitals
Electron Cloud and Orbitals
Electrons occupy regions called orbitals, which are areas of probability where electrons are likely to be found. Orbitals are grouped into shells and subshells.
Principal Quantum Number (n): Describes energy level (n = 1, 2, 3, ...)
Orbital Shapes: s, p, d, f

Subshell Electron Capacity
Each subshell has a maximum electron capacity:
s: 2 electrons
p: 6 electrons
d: 10 electrons
f: 14 electrons

Electron Configuration
Electron configuration describes the arrangement of electrons in an atom. Electrons fill the lowest energy orbitals first (Aufbau principle), and Hund's rule states that electrons occupy orbitals singly before pairing.
Example: Br: [Ar] 4s2 3d10 4p5
Example: Zr: [Kr] 5s2 4d2

Chemical Bonding
Ionic Compounds
Ionic compounds are formed by the transfer of electrons from metals to non-metals, resulting in the formation of cations and anions.
Example: NaCl (Sodium chloride), MgO (Magnesium oxide)
Transition metals: Can form multiple ions (e.g., Fe2+, Fe3+)
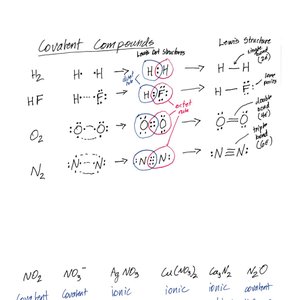
Covalent Compounds
Covalent compounds are formed by sharing electrons between two or more non-metals.
Example: CO2 (Carbon dioxide), N2O (Dinitrogen monoxide)
Naming: Use prefixes (mono-, di-, tri-, etc.) to indicate the number of atoms.
Lewis Structures
Lewis structures represent the arrangement of electrons in molecules. The octet rule states that atoms tend to have eight electrons in their valence shell.
Steps: 1) Count valence electrons, 2) Arrange atoms, 3) Use electrons to draw bonds, 4) Add lone pairs, 5) Check octet rule.
Resonance: Some molecules have multiple valid Lewis structures.
Molecular Geometry
Valence Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory predicts the shape of molecules based on the repulsion between electron pairs.
Electron Geometry: Arrangement of all electron groups
Molecular Geometry: Arrangement of bonded atoms
Example: CO2 is linear, NH3 is trigonal pyramidal, H2O is bent
Intermolecular Forces and Polarity
Polarity
Polarity arises when two covalently bonded atoms have different electronegativities, resulting in partial positive and negative charges.
Example: H2O is polar due to the difference in electronegativity between H and O.
Nuclear Chemistry
Nuclear Reactions
Nuclear reactions involve changes in the nucleus of an atom, including fission (splitting) and fusion (combining).
Types of Radiation: Alpha (α), Beta (β), Positron (β+)
Example Equations:
Half-life
Half-life is the time required for half of a radioactive sample to decay.
Formula:
Example: If 100g of a substance has a half-life of 1.3 days, after 2.6 days, 25g remains.
Acids, Bases, and Polyatomic Ions
Acids
Acids are compounds that release H+ ions in water. Naming depends on the anion present.
Examples: HCl (Hydrochloric acid), HF (Hydrofluoric acid), HNO3 (Nitric acid)
Polyatomic Ions
Polyatomic ions are charged species composed of multiple atoms.
Examples: SO42- (Sulfate), NO3- (Nitrate), NH4+ (Ammonium)
Ion | Formula |
|---|---|
Sulfate | SO42- |
Nitrate | NO3- |
Ammonium | NH4+ |
Phosphate | PO43- |
Carbonate | CO32- |
Summary Table: Key Concepts
Concept | Definition | Example |
|---|---|---|
Atom | Smallest unit of an element | H, O, C |
Isotope | Same element, different neutrons | C-12, C-14 |
Ionic Compound | Transfer of electrons | NaCl |
Covalent Compound | Sharing of electrons | CO2 |
Acid | Releases H+ in water | HCl |
Polyatomic Ion | Multiple atoms, charged | SO42- |
Additional info:
Electron configuration and periodic trends are foundational for understanding chemical reactivity.
Resonance structures and VSEPR theory are essential for predicting molecular shape and properties.
Nuclear chemistry concepts such as half-life and types of radiation are important for understanding radioactive decay.
