 Back
BackAtoms, Elements, and Ion Formation: Properties and Classification
Study Guide - Smart Notes

Atoms and Elements
Classification of Elements
Elements are classified based on their physical and chemical properties into metals, nonmetals, and metalloids. Understanding these classifications is fundamental to predicting element behavior and their role in chemical reactions.
Metals: High thermal and electrical conductivity, malleable, ductile, shiny (metallic luster), and solid at room temperature.
Nonmetals: Poor conductors of electricity, often powdery solids or gases, dull appearance.
Metalloids: Properties intermediate between metals and nonmetals; can be shiny or dull, brittle solids, conduct electricity moderately well (used as semiconductors).
Example: Silicon is a metalloid used in computer chips due to its moderate conductivity.
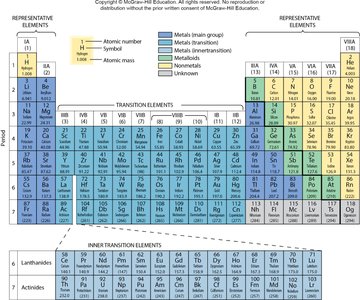
Periodic Table Organization
The periodic table arranges elements by increasing atomic number and groups them by similar properties. Groups (columns) and periods (rows) help identify element families and predict their chemical behavior.
Groups: Elements in the same group have similar valence electron configurations and chemical properties.
Periods: Elements in the same period have the same number of electron shells.
Example: Group 1A contains alkali metals, which are highly reactive.
The Octet Rule and Ion Formation
The Octet Rule
The octet rule states that atoms tend to gain, lose, or share electrons to achieve a stable configuration with eight valence electrons, similar to the nearest noble gas. This drives chemical reactivity and the formation of ions.
Elements on the right side of the periodic table move forward to the next noble gas configuration.
Elements on the left move backward to the previous noble gas configuration.
Atoms achieve stability by gaining, losing, or sharing electrons.
Ions: Cations and Anions
Ions are electrically charged particles formed when atoms gain or lose electrons. The type of ion formed depends on the element's position in the periodic table and its tendency to achieve a noble gas configuration.
Cation: Positively charged ion formed by loss of electrons (e.g., ).
Anion: Negatively charged ion formed by gain of electrons (e.g., ).
Example: Aluminum (Al) loses three electrons to form , while oxygen (O) gains two electrons to form .
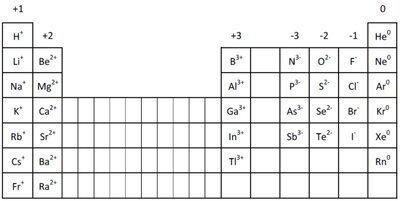
Formation of Cations and Anions
Atoms of the same group lose or gain the same number of electrons, resulting in ions with electron configurations matching the nearest noble gas.
Cation Formation: Metals lose electrons to form cations. Example:
Anion Formation: Nonmetals gain electrons to form anions. Example:
Example: Magnesium (Mg) loses two electrons to become ; chlorine (Cl) gains one electron to become .
Charges of Transition Metals
Transition metals can form ions with variable positive charges. The specific charge is indicated by Roman numerals in the ion's name (e.g., iron(II) is , copper(I) is ).
Transition metals always carry a positive charge.
Charge is specified in the compound name.
Example: Iron can form or depending on the reaction.
Calculating Subatomic Particles in Ions
Determining Protons, Neutrons, and Electrons
To fully describe an ion, it is necessary to calculate the number of protons, neutrons, and electrons. The charge of the ion affects the electron count.
Protons: Equal to the atomic number.
Neutrons: Atomic mass minus atomic number.
Electrons: Number of protons minus the ion's charge.
Example: For (potassium ion): - Protons: 19 - Neutrons: - Electrons:
Formula:
Practice Problems
Applying the above formula to various ions:
: 16 protons, 16 neutrons, electrons
: 12 protons, 12 neutrons, electrons
: 15 protons, 16 neutrons, electrons
Example: For (oxide ion): - Protons: 8 - Neutrons: - Electrons:
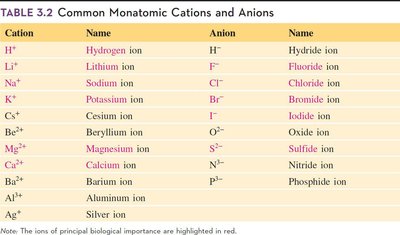
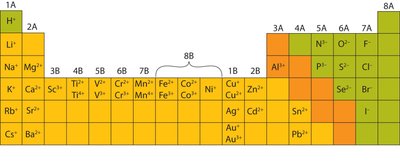
Summary Table: Common Monatomic Cations and Anions
The following table summarizes the names and charges of common monatomic ions:
Cation | Name | Anion | Name |
|---|---|---|---|
H+ | Hydrogen ion | H- | Hydride ion |
Li+ | Lithium ion | F- | Fluoride ion |
Na+ | Sodium ion | Cl- | Chloride ion |
K+ | Potassium ion | Br- | Bromide ion |
Cs+ | Cesium ion | I- | Iodide ion |
Be2+ | Beryllium ion | O2- | Oxide ion |
Mg2+ | Magnesium ion | S2- | Sulfide ion |
Ca2+ | Calcium ion | N3- | Nitride ion |
Ba2+ | Barium ion | P3- | Phosphide ion |
Al3+ | Aluminum ion | ||
Ag+ | Silver ion |

Additional info:
These notes cover the properties and classification of elements, the octet rule, ion formation, and calculation of subatomic particles in ions, which are directly relevant to Ch.4 Atoms and Elements and Ch.6 Ionic and Molecular Compounds in a general chemistry course.
