 Back
BackAtoms, Elements, and the Periodic Table: Study Notes for General Chemistry
Study Guide - Smart Notes

Atoms and Elements
Atomic Structure
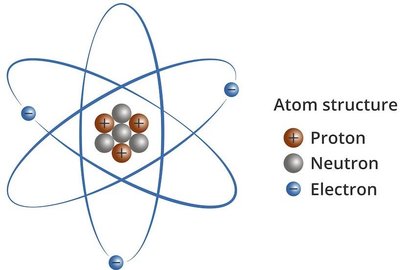
Atoms are the fundamental units of matter, composed of three primary subatomic particles: protons, neutrons, and electrons. The arrangement and number of these particles determine the identity and properties of each element.
Protons: Positively charged particles found in the nucleus. The number of protons defines the atomic number (Z) and the element.
Neutrons: Neutral particles found in the nucleus. The sum of protons and neutrons gives the mass number (A).
Electrons: Negatively charged particles that orbit the nucleus in shells or energy levels. In a neutral atom, the number of electrons equals the number of protons.
Example: Bromine (Br) with atomic number 35 and mass number 80 has 35 protons, 45 neutrons (80-35), and 35 electrons.
The Periodic Table and Periodic Law
Organization of the Periodic Table
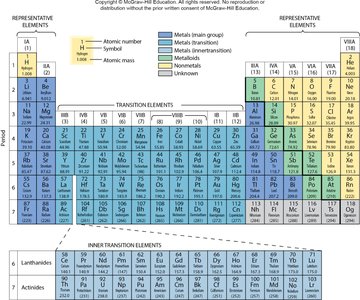
The periodic table arranges elements by increasing atomic number into horizontal rows called periods and vertical columns called groups. The periodic law states that the physical and chemical properties of elements are periodic functions of their atomic numbers.
Groups (Columns): Elements in the same group have similar chemical properties. Main groups are labeled 1A to 8A.
Periods (Rows): Elements in the same period have the same number of electron shells.
Representative Elements: Groups 1A-8A are called main group elements.
Transition Metals: Elements in the center block (groups 3-12).
Classification of Elements
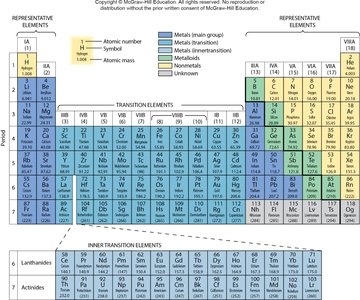
Metals: High thermal and electrical conductivity, malleable, ductile, shiny, and solid at room temperature.
Nonmetals: Poor conductors, can be gases or powdery solids, dull in appearance.
Metalloids: Properties intermediate between metals and nonmetals; often semiconductors.
Groups and Their Properties
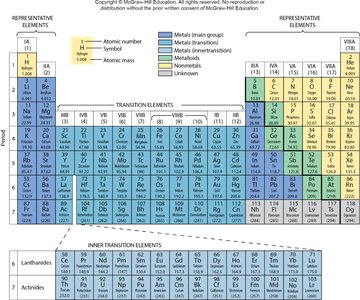
Group 1A (Alkali Metals): Highly reactive metals (e.g., Li, Na, K).
Group 2A (Alkaline Earth Metals): Reactive metals (e.g., Be, Mg, Ca).
Group 7A (Halogens): Reactive nonmetals (e.g., F, Cl, Br, I).
Group 8A (Noble Gases): Inert, very unreactive gases (e.g., He, Ne, Ar, Kr).
Electronic Structure and Valence Electrons
Electron Shells and Valence Electrons
Electrons are arranged in shells around the nucleus. The outermost shell contains the valence electrons, which determine an element's chemical reactivity. The number of valence electrons corresponds to the group number for main group elements.
Example: Aluminum (Group 3A) has 3 valence electrons; Oxygen (Group 6A) has 6 valence electrons.

The Octet Rule
The octet rule states that atoms tend to gain, lose, or share electrons to achieve a stable configuration with eight valence electrons, similar to the nearest noble gas.
Elements on the right side of the table gain electrons to reach the next noble gas configuration.
Elements on the left side lose electrons to reach the previous noble gas configuration.
Ions and Their Formation
Cations and Anions
An ion is an atom or molecule with a net electric charge due to the loss or gain of electrons.
Cation: Positively charged ion formed by the loss of electrons (e.g., Na+).
Anion: Negatively charged ion formed by the gain of electrons (e.g., Cl-).
Example: Aluminum (Al) in Group 3A loses 3 electrons to form Al3+.
Formation of Ions and the Octet Rule
Metals tend to lose electrons and form cations with a charge equal to their group number.
Nonmetals tend to gain electrons and form anions with a charge equal to 8 minus their group number.
Example Equations:
Formation of a sodium cation:
Formation of a fluoride anion:
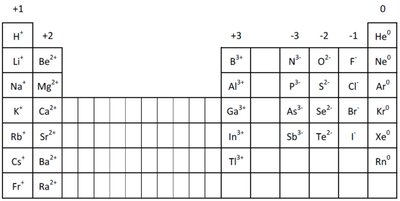
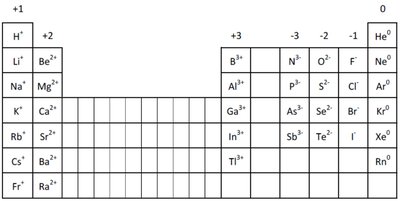
Representative Elements and Their Charges
Common Charges of Main Group Elements
The charge of ions formed by main group elements can be predicted based on their group number.
Group | Common Ion Charge | Example Elements |
|---|---|---|
1A | +1 | H+, Li+, Na+ |
2A | +2 | Be2+, Mg2+ |
3A | +3 | Al3+ |
5A | -3 | N3-, P3- |
6A | -2 | O2-, S2- |
7A | -1 | F-, Cl- |
8A | 0 | He, Ne, Ar |
Appendix: Course Schedule Overview
Lecture and Lab Schedule
The course covers foundational topics in general chemistry, including atomic structure, the periodic table, chemical bonding, and quantitative aspects of chemical reactions. Labs reinforce these concepts through hands-on experiments.
Week | Lecture Topic | Lab Activity |
|---|---|---|
1 | Chemistry in Our Lives | Check-in & Lab Safety |
2 | Chemistry and Measurements | Basic Laboratory Techniques |
3 | Matter and Energy | Separation of a Mixture |
4 | Atoms and Elements | Names and Formulas of Chemical Compounds |
5 | Ionic and Molecular Compounds | Ionic and Molecular Bonding (PHET simulation) |
6 | Chemical Quantities and Reactions | Chemical Reactions and their Classifications |
7 | Acids & Bases, Organic Chemistry | Acids & Bases (Titration), Physical Properties of Organic Compounds |
8 | Carbohydrates | Carbohydrates |
