 Back
BackAtoms, Elements, and the Periodic Table: Study Guide for General Chemistry
Study Guide - Smart Notes

Atoms and Elements
Atomic Structure
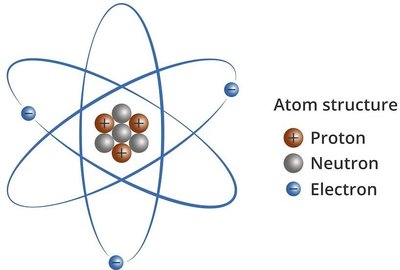
Atoms are the fundamental building blocks of matter, composed of three primary subatomic particles: protons, neutrons, and electrons. The arrangement and number of these particles determine the chemical properties of each element.
Protons: Positively charged particles found in the nucleus. The number of protons defines the atomic number (Z) of an element.
Neutrons: Neutral particles also located in the nucleus. The sum of protons and neutrons gives the mass number (A).
Electrons: Negatively charged particles that orbit the nucleus in shells.
Example: Bromine (Br) has 35 protons, 45 neutrons (80-35), and 35 electrons.
The Periodic Table and Periodic Law
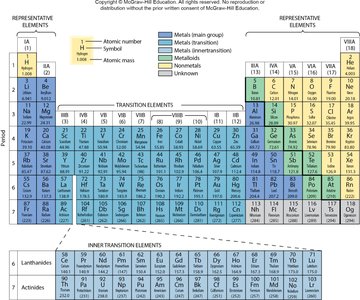
Organization of the Periodic Table
The periodic table arranges elements by increasing atomic number. Elements are grouped into horizontal rows called periods and vertical columns called groups. The periodic law states that the physical and chemical properties of elements are periodic functions of their atomic numbers.
Main Groups: Group 1A to Group 8A (representative elements).
Periods: Horizontal rows numbered 1 to 7.
Groups: Vertical columns numbered 1A to 8A.
Example: Selenium (Se) is in Group 6A, Period 4; Calcium (Ca) is in Group 2A, Period 4; Boron (B) is in Group 3A, Period 2.
Element Classification
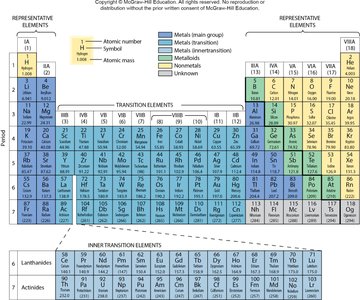
Elements are classified as metals, nonmetals, or metalloids based on their properties.
Metals: High conductivity, malleable, ductile, shiny, solid at room temperature.
Nonmetals: Poor conductivity, powdery solids or gases, dull appearance.
Metalloids: Intermediate properties, brittle, moderate conductivity, used as semiconductors.
Groups and Their Properties
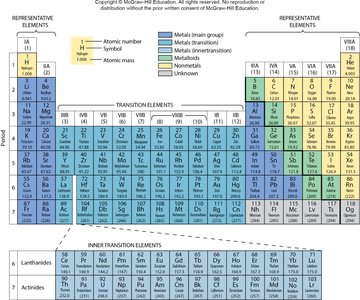
Elements in the same group have similar chemical properties due to their similar valence electron configurations.
Group 1A: Alkali metals (Li, Na, K) – highly reactive, one valence electron.
Group 2A: Alkaline earth metals (Be, Ca, Mg) – two valence electrons.
Group 7A: Halogens (Cl, Br, I) – very reactive, seven valence electrons.
Group 8A: Noble gases (He, Ne, Kr) – inert, eight valence electrons (except He).
Electronic Structure and Valence Electrons
Electron Shells and Valence Electrons
Electrons are arranged in shells around the nucleus. The outermost shell contains the valence electrons, which determine an element's chemical reactivity.
Valence Electrons: The number of valence electrons corresponds to the group number for main group elements.
Example: Aluminum (Group 3A) has 3 valence electrons; Oxygen (Group 6A) has 6 valence electrons.

The Octet Rule
The Octet Rule states that elements tend to react to achieve a stable configuration of eight electrons in their outer shell, similar to the nearest noble gas.
Elements on the right side of the table gain electrons to reach the next noble gas configuration.
Elements on the left side lose electrons to reach the previous noble gas configuration.
Atoms gain, lose, or share electrons in chemical reactions to attain this stable state.
Ions and Their Formation
Cations and Anions
Ions are electrically charged particles formed when atoms gain or lose electrons.
Cation: Positively charged ion formed by loss of electrons (e.g., Na → Na+ + 1e-).
Anion: Negatively charged ion formed by gain of electrons (e.g., F + 1e- → F-).
Example: Aluminum (Al) in Group 3A loses 3 electrons to form Al3+.
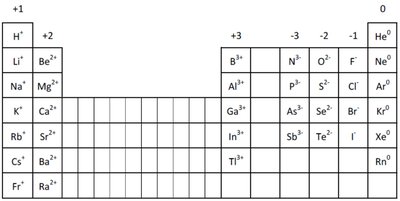
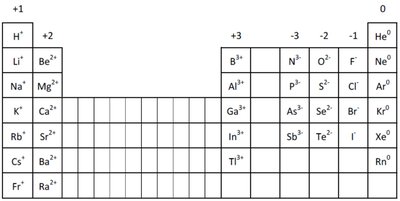
Ion Formation and the Octet Rule
Atoms of main group elements form ions to achieve the electron configuration of the nearest noble gas.
All atoms in a group lose or gain the same number of electrons.
Resulting ions have the same number of electrons as the nearest noble gas.
Example: Oxygen (O) gains 2 electrons to form O2-, achieving the configuration of neon (Ne).
Summary Table: Groups, Periods, and Element Properties
Group | Period | Element Example | Valence Electrons | Common Ion Charge |
|---|---|---|---|---|
1A (Alkali Metals) | 2 | Li | 1 | +1 |
2A (Alkaline Earth Metals) | 4 | Ca | 2 | +2 |
3A | 2 | B | 3 | +3 |
6A | 4 | Se | 6 | -2 |
8A (Noble Gases) | 1 | He | 2 | 0 |
Additional info:
The lecture and lab schedules confirm coverage of all major topics in general chemistry, including atomic structure, periodic table, chemical bonding, acids and bases, hydrocarbons, and carbohydrates.
Quiz and exam dates are aligned with the progression of these topics, supporting their relevance to the course.
