 Back
BackAtoms, Ions, and Chemical Reactions: Core Concepts and Solubility Guidelines
Study Guide - Smart Notes

Chapter 2: Atoms and the Periodic Table
Atomic Theory
Atomic theory forms the foundation of modern chemistry, describing the nature and behavior of matter at the atomic level.
All matter is composed of atoms, which are indivisible particles.
Atoms of one element cannot change into atoms of another element during chemical reactions.
Atoms of the same element have the same number of protons and electrons, determining their chemical behavior.
Compounds are formed by the chemical combination of two or more elements in a specific ratio.
Atomic Symbols and Numbers
Atomic symbol (X): The one- or two-letter abbreviation for an element.
Atomic number (Z): Number of protons in the nucleus; also equals the number of electrons in a neutral atom.
Atomic mass number (A): Total number of protons and neutrons in the nucleus.
Number of neutrons (N):
Isotopes and Ions
Isotopes: Atoms of the same element with the same number of protons but different numbers of neutrons.
Ions: Atoms or molecules with a net electric charge due to the loss or gain of electrons.
For ions: Number of electrons = Number of protons – charge
Atomic Weight
The atomic weight is the weighted average of the isotopic masses of an element.
Calculated as:
Electron Configuration and Periodic Table
Electron configuration: The arrangement of electrons in an atom or ion.
Element blocks: s, p, d, f blocks correspond to the type of atomic orbital being filled.
Major group elements: Groups 1A, 2A, 7A, and 8A are especially important for chemical reactivity.
Chapter 3: Ionic Compounds
Key Terms and Concepts
Molecule: Two or more atoms bonded together.
Diatomic molecule: Molecule consisting of two atoms (e.g., O2).
Polyatomic molecule: Molecule with more than two atoms.
Cation: Positively charged ion.
Anion: Negatively charged ion.
Monatomic ion: Ion formed from a single atom.
Polyatomic ion: Ion composed of multiple atoms.
Transition metal ion: Ion formed from a transition metal, often with variable charge.
Common Ions and Acids
Know the names and charges of common ions (e.g., Na+, Cl-, SO42-).
Know the names of common acids (e.g., HCl, H2SO4).
Constructing and Naming Ionic Compounds
Combine cations and anions to form neutral compounds.
Write formulas for ionic compounds (monatomic, polyatomic, transition metal).
Name ionic compounds according to standard nomenclature rules.
Periodic Table and Ion Formation
Major group elements (1A, 2A, 3A, 5A, 6A, 7A) form predictable ions based on their group number.
Chapter 4: Molecular Compounds and Bonding
Valence Electrons and Bonding
Number of valence electrons: Equals the group number for main group elements.
Valence electrons participate in chemical bonding.
Types of Bonds
Ionic bond: Electrostatic attraction between cations and anions.
Covalent bond: Two or more atoms share electrons.
Polar covalent bond: Covalent bond between atoms with different electronegativities.
Electronegativity and Bond Polarity
Electronegativity (EN): Ability of an atom to attract electrons in a bond.
EN increases from bottom to top and left to right on the periodic table.
Common EN values: F (4.0), O/Cl (3.5), N (3.0), C (2.5), H (2.1), Br (2.8), others below 2.5.
Bond type by ΔEN:
ΔEN > 2: Ionic
0.5 < ΔEN < 1.9: Polar covalent
ΔEN < 0.4: Nonpolar covalent
Lewis Structures
Count valence electrons.
Draw skeleton structure.
Deduct 2 electrons for each bond.
Add lone pairs to complete octets.
If octet is not achieved, create multiple bonds as needed.
Molecular Geometry and Polarity
Electron group = number of outer atoms + number of lone pairs.
Geometry and bond angles:
2 groups: Linear, 180°, nonpolar
3 groups: Trigonal planar, 120°, nonpolar; Bent, 120°, polar
4 groups: Tetrahedral, 109°, nonpolar; Trigonal pyramidal, 109°, polar; Bent, 109°, polar
Polarity depends on both bond polarity and molecular geometry.
Chapter 5: Classification & Balancing of Chemical Reactions
Balancing Chemical Equations
Balancing chemical equations ensures the law of conservation of mass is obeyed in chemical reactions.
List each element on both sides of the equation.
Balance polyatomic ions or compounds first.
Balance atoms that appear only once on each side.
Balance hydrogens, then oxygens.
Double-check atom counts on both sides.
Use only coefficients; do not change subscripts.
Classifying Reactions
Redox reactions: Involve transfer of electrons.
Acid/base reactions: Involve transfer of protons (H+).
Precipitation reactions: Formation of an insoluble solid from soluble reactants.
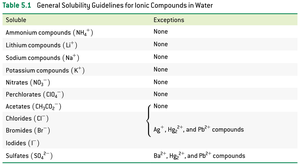
Solubility Guidelines for Ionic Compounds in Water
Solubility rules help predict whether a compound will dissolve in water or form a precipitate.
Soluble | Exceptions |
|---|---|
Ammonium compounds (NH4+) | None |
Lithium compounds (Li+) | None |
Sodium compounds (Na+) | None |
Potassium compounds (K+) | None |
Nitrates (NO3-) | None |
Perchlorates (ClO4-) | None |
Acetates (CH3COO-) | None |
Chlorides (Cl-) | Ag+, Hg22+, and Pb2+ compounds |
Bromides (Br-) | Ag+, Hg22+, and Pb2+ compounds |
Iodides (I-) | Ag+, Hg22+, and Pb2+ compounds |
Sulfates (SO42-) | Ba2+, Hg22+, and Pb2+ compounds |
Example: Predicting Precipitation
Mixing solutions of NaCl and AgNO3 forms a precipitate of AgCl, since AgCl is insoluble.
Additional info: The solubility table is essential for predicting the outcomes of double displacement reactions and for identifying the formation of precipitates in aqueous solutions.
