 Back
BackBiochemical Energy and the Generation of ATP
Study Guide - Smart Notes

Biochemical Energy and Cellular Metabolism
Energy and Life
All living organisms require a continuous flow of energy to sustain life. This energy is used to perform various types of work, including mechanical work (such as muscle movement and transport of substances in the blood) and chemical work (such as the synthesis of biomolecules and the transport of ions across membranes). Ultimately, all energy used by living organisms originates from the sun, with plants capturing solar energy through photosynthesis and animals obtaining energy by consuming plants.
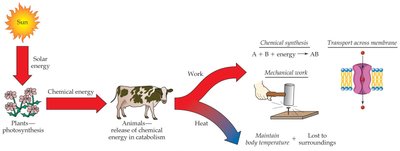
Energy Conservation: Energy cannot be created or destroyed, only converted from one form to another.
Energy Storage: Organisms must store energy for future use and release it gradually to maintain homeostasis.
Free Energy and Chemical Reactions
The energy available to do work in a system is called free energy (ΔG). Chemical reactions can either release or absorb free energy:
Exergonic Reactions: Release free energy (ΔG < 0); products have less energy than reactants; these reactions are favorable.
Endergonic Reactions: Absorb free energy (ΔG > 0); products have more energy than reactants; these reactions are unfavorable.
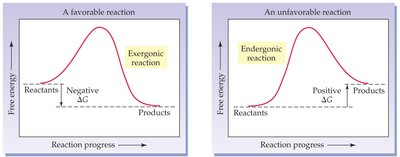
Many biochemical reactions are endergonic and require energy input, often provided by coupling with exergonic reactions such as ATP hydrolysis.
Photosynthesis and Oxidation
Photosynthesis in plants is an endergonic process that stores solar energy in glucose. Animals obtain this energy by oxidizing glucose, an exergonic process that releases energy for cellular work.
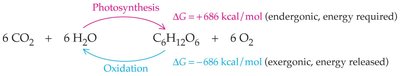
Metabolism and Metabolic Pathways
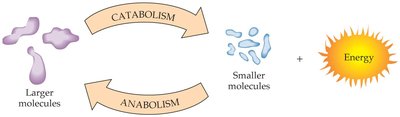
Catabolism and Anabolism
Metabolism is the sum of all chemical reactions in an organism. It is divided into two main types:
Catabolism: The breakdown of larger molecules into smaller ones, releasing energy.
Anabolism: The synthesis of larger molecules from smaller ones, requiring energy input.
Types of Metabolic Pathways
Metabolic pathways can be classified as:
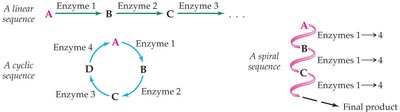
Linear: A sequence of reactions leading to a single product (e.g., glycolysis).
Cyclic: A series of reactions that regenerate the starting material (e.g., citric acid cycle).
Spiral: The same set of enzymes is used repeatedly to build up or break down molecules (e.g., beta-oxidation of fatty acids).
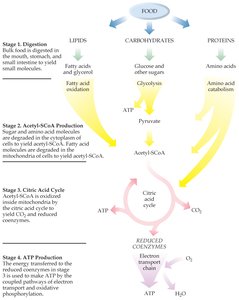
Stages of Energy Production
Energy production in cells occurs in four main stages:
Digestion: Breakdown of large food molecules into smaller units by enzymes.
Acetyl-CoA Production: Small molecules are converted to acetyl-CoA, a common intermediate.
Citric Acid Cycle: Acetyl-CoA is oxidized to CO2, and high-energy coenzymes (NADH, FADH2) are produced.
ATP Production: Electrons from NADH and FADH2 are transferred through the electron transport chain, generating ATP.
Acetyl-CoA: The Central Metabolite
Acetyl-CoA is formed by attaching an acetyl group to coenzyme A via a thiol group. It is the common intermediate in the breakdown of carbohydrates, fats, and proteins.
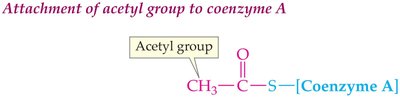
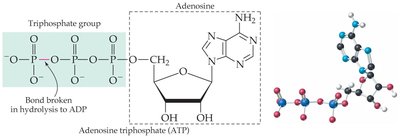
ATP: The Energy Currency of the Cell
Structure and Function of ATP
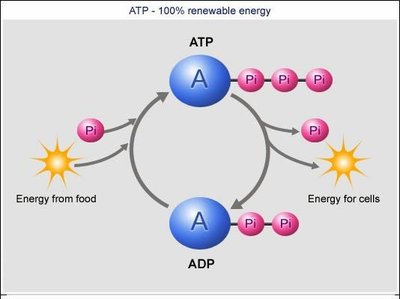
Adenosine triphosphate (ATP) is the primary energy carrier in cells. Its energy is stored in the high-energy phosphate bonds, particularly the bond between the second and third phosphate groups. Hydrolysis of ATP to ADP (adenosine diphosphate) or AMP (adenosine monophosphate) releases energy that can be used for cellular work.
ATP Hydrolysis: ATP + H2O → ADP + Pi + H+ (ΔG = -7.3 kcal/mol)
ATP Synthesis: ADP + Pi + H+ → ATP + H2O (ΔG = +7.3 kcal/mol)
ATP/ADP Cycle
ATP is continuously recycled in cells. Energy from food is used to regenerate ATP from ADP, and the hydrolysis of ATP provides energy for cellular processes.
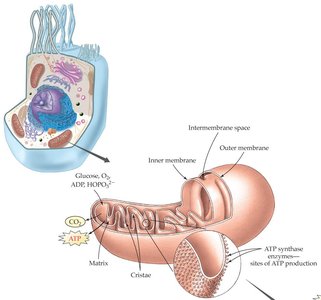
Cellular Sites of Energy Production
Mitochondria: The Powerhouse of the Cell
Mitochondria are the organelles where most ATP is produced. They contain their own DNA and are believed to have originated from ancient symbiotic bacteria. The inner mitochondrial membrane and matrix are the sites of the citric acid cycle and electron transport chain.
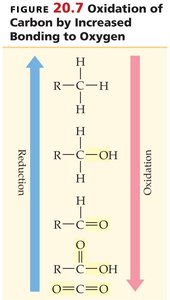
Oxidation, Reduction, and Coenzymes
Oxidation and Reduction in Metabolism
Energy is obtained from food by oxidation reactions, where electrons (often as hydrogen atoms) are transferred from one molecule to another. Oxidation is characterized by the loss of electrons or hydrogen atoms, or the gain of oxygen atoms. Reduction is the gain of electrons or hydrogen atoms.
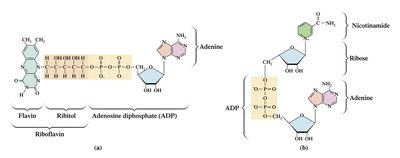
Coenzymes in Electron Transfer
Coenzymes such as NAD+ (nicotinamide adenine dinucleotide) and FAD (flavin adenine dinucleotide) act as electron carriers. They are reduced to NADH and FADH2 during catabolic reactions and transport electrons to the electron transport chain.
Coenzyme | Oxidized Form | Reduced Form |
|---|---|---|
NAD+ | NAD+ | NADH/H+ |
FAD | FAD | FADH2 |
The Citric Acid Cycle (Krebs Cycle)
Overview and Steps
The citric acid cycle is a cyclic metabolic pathway that oxidizes acetyl-CoA to CO2 and reduces coenzymes NAD+ and FAD to NADH and FADH2. It also produces GTP, which can be converted to ATP. The cycle is regulated by the energy needs of the cell.
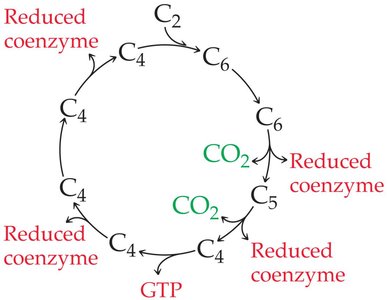
Key Products: 2 CO2, 3 NADH, 1 FADH2, 1 GTP (per acetyl-CoA)
Regulation: Isocitrate dehydrogenase is activated by ADP and inhibited by NADH.
Electron Transport Chain and ATP Synthesis
Electron Transport Chain (ETC)
The ETC is a series of protein complexes in the inner mitochondrial membrane that transfer electrons from NADH and FADH2 to oxygen, forming water. The energy released is used to pump H+ ions across the membrane, creating a proton gradient.
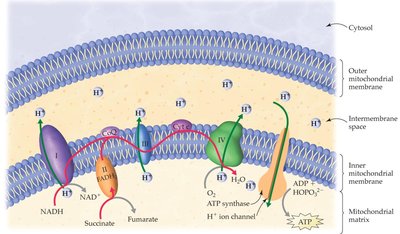
Complex I: Accepts electrons from NADH and pumps H+.
Complex II: Accepts electrons from FADH2 (no H+ pumping).
Complex III & IV: Continue electron transfer and pump H+.
ATP Synthase and Oxidative Phosphorylation
ATP synthase is an enzyme that uses the energy of the proton gradient to synthesize ATP from ADP and Pi as H+ ions flow back into the mitochondrial matrix. This process is called oxidative phosphorylation.
Each NADH yields approximately 3 ATP, and each FADH2 yields approximately 2 ATP.
Reactive Oxygen Species (ROS) and Antioxidants
Formation and Effects of ROS
Reactive oxygen species (ROS) are highly reactive oxygen-containing molecules produced as by-products of the electron transport chain. They can damage cell membranes and DNA if not controlled.
Examples of ROS: Superoxide ion (·O2–), hydroxyl radical (·OH), hydrogen peroxide (H2O2).
Defense Mechanisms
Cells use enzymes such as superoxide dismutase and catalase, as well as antioxidants like vitamin E, vitamin C, and glutathione, to neutralize ROS and protect against oxidative damage.
Summary Table: Key Steps in Biochemical Energy Production
Stage | Main Process | Key Products |
|---|---|---|
1. Digestion | Breakdown of food macromolecules | Small molecules (glucose, fatty acids, amino acids) |
2. Acetyl-CoA Production | Conversion to acetyl-CoA | Acetyl-CoA |
3. Citric Acid Cycle | Oxidation of acetyl-CoA | CO2, NADH, FADH2, GTP |
4. Electron Transport & ATP Synthesis | Electron transfer and ATP production | ATP, H2O |
