 Back
BackCarbohydrates: Structure, Classification, and Biological Importance
Study Guide - Smart Notes

Carbohydrates
Introduction to Carbohydrates
Carbohydrates are essential biomolecules that serve as a major source of energy in the human diet. They are composed of carbon, hydrogen, and oxygen, and are commonly referred to as saccharides or sugars. Carbohydrates are produced by plants through photosynthesis and are oxidized in living cells to release energy.
Major dietary sources: Bread, pasta, potatoes, and other plant-based foods.
Biological importance: Energy storage, structural components, and cellular recognition.
Types of Carbohydrates
Carbohydrates are classified based on the number of saccharide units:
Monosaccharides: The simplest carbohydrates, consisting of a single sugar unit (e.g., glucose, fructose).
Disaccharides: Composed of two monosaccharide units linked by a glycosidic bond (e.g., sucrose, lactose).
Polysaccharides: Large molecules formed by the linkage of many monosaccharide units (e.g., starch, cellulose, glycogen).
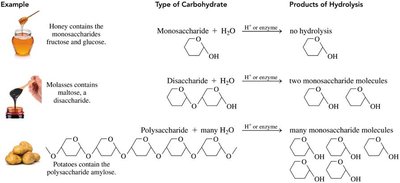
Monosaccharides
Structure and Classification
Monosaccharides contain several hydroxyl groups attached to a carbon chain of three to eight atoms. They are classified by:
Functional group: Aldoses contain an aldehyde group; ketoses contain a ketone group.
Number of carbon atoms: Trioses (3C), tetroses (4C), pentoses (5C), hexoses (6C).
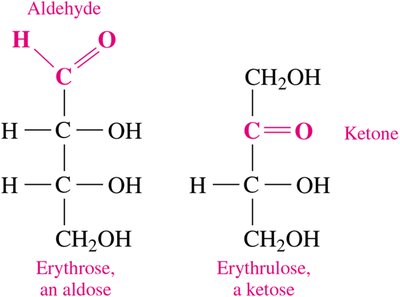
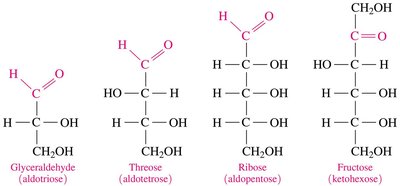
Stereochemistry of Carbohydrates
Chirality and Stereoisomers
Many monosaccharides are chiral molecules, meaning they have non-superimposable mirror images (enantiomers). Chirality is a key concept in carbohydrate chemistry, affecting biological activity and recognition.
Chiral carbon: A carbon atom bonded to four different groups.
Enantiomers: Stereoisomers that are mirror images but not superimposable.
Achiral: Molecules or objects that are superimposable on their mirror images.


Fischer Projections
Fischer projections are two-dimensional representations of three-dimensional molecules, commonly used for carbohydrates. The most oxidized group is placed at the top, and the configuration of chiral centers is shown by the arrangement of horizontal and vertical lines.
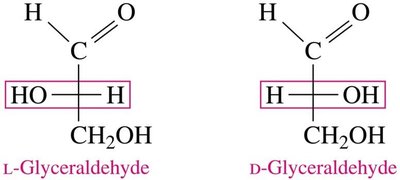
D- and L- notation: Determined by the position of the –OH group on the chiral carbon farthest from the carbonyl group (right = D, left = L).
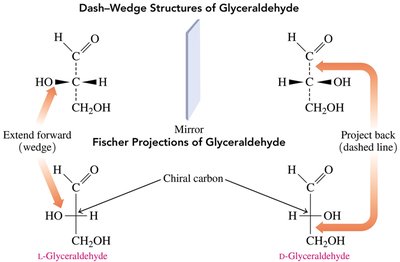
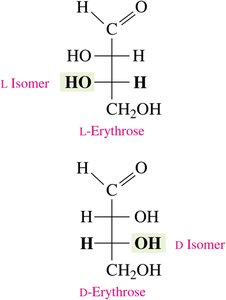
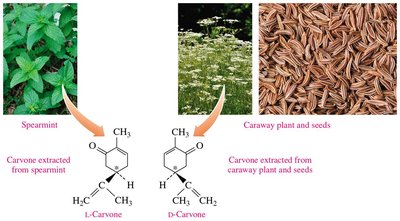
Biological Importance of Enantiomers
Enantiomers can have different biological effects. For example, one enantiomer of carvone smells like spearmint, while its mirror image smells like caraway.
Common Monosaccharides
D-Glucose
D-Glucose is the most common hexose and is found in fruits, vegetables, and as blood sugar. It is a building block for disaccharides and polysaccharides.
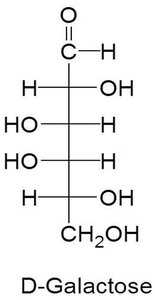
D-Galactose
D-Galactose is an aldohexose obtained from lactose and is important in brain and nervous system membranes.
D-Fructose
D-Fructose is a ketohexose and the sweetest carbohydrate, found in honey and high-fructose corn syrup.

Haworth Structures of Monosaccharides
Formation of Cyclic Structures
Pentoses and hexoses commonly form five- or six-membered rings (Haworth structures) through the reaction of a carbonyl group with a hydroxyl group in the same molecule. The orientation of the –OH group on the anomeric carbon determines the α or β isomer.
Chemical Properties of Monosaccharides
Oxidation and Reduction
Monosaccharides can undergo oxidation and reduction reactions:
Oxidation: Aldehyde groups can be oxidized to carboxylic acids (e.g., by Benedict’s solution). Monosaccharides that can reduce other substances are called reducing sugars.
Reduction: The carbonyl group can be reduced to an alcohol, forming sugar alcohols (alditols) such as sorbitol and xylitol, used as sweeteners in sugar-free products.
Disaccharides
Structure and Formation
Disaccharides are formed by the dehydration reaction between two monosaccharides, creating a glycosidic bond. Common examples include:
Maltose: Two glucose units, α(1→4) glycosidic bond.
Lactose: Galactose and glucose, β(1→4) glycosidic bond.
Sucrose: Glucose and fructose, α(1→2)β glycosidic bond.
Polysaccharides
Major Types and Functions
Polysaccharides are large polymers of monosaccharides with diverse biological roles:
Starch: Storage form of glucose in plants; consists of amylose (unbranched, α(1→4) bonds) and amylopectin (branched, α(1→4) and α(1→6) bonds).
Glycogen: Storage form of glucose in animals; highly branched, similar to amylopectin but more frequent branching.
Cellulose: Structural component in plants; unbranched chains of glucose with β(1→4) bonds, indigestible by humans.
Glycosaminoglycans
Glycosaminoglycans are unbranched polysaccharides consisting of repeating disaccharide units, important for cushioning and lubricating structures in the body (e.g., keratan sulfate in the cornea).
Summary Table: Types of Carbohydrates
Type | Example | Bond Type | Biological Role |
|---|---|---|---|
Monosaccharide | Glucose, Fructose | — | Energy source |
Disaccharide | Sucrose, Lactose | Glycosidic bond | Transport, energy |
Polysaccharide | Starch, Glycogen, Cellulose | α(1→4), α(1→6), β(1→4) | Storage, structure |
