 Back
BackCarbohydrates: Structure, Isomerism, and Biochemical Significance
Study Guide - Smart Notes

Biochemical Substances
Classification of Biochemical Substances
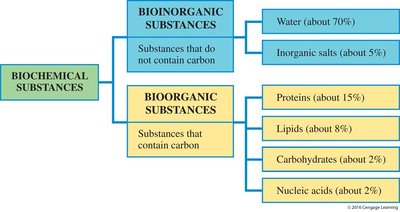
Biochemical substances are the chemical compounds that constitute living organisms. They are broadly classified into two categories: bioinorganic and bioorganic substances. This classification is based on the presence or absence of carbon atoms in their molecular structure.
Bioinorganic Substances: Do not contain carbon. Examples include water (about 70% of body mass) and inorganic salts (about 5%).
Bioorganic Substances: Contain carbon. Major classes include proteins (about 15%), lipids (about 8%), carbohydrates (about 2%), and nucleic acids (about 2%).
Carbohydrates: Structure and Classification
Monosaccharides: Aldoses and Ketoses
Monosaccharides are the simplest carbohydrates and are classified based on the functional group present:
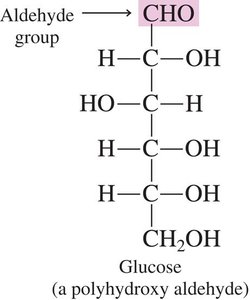
Aldoses: Contain an aldehyde group (–CHO) at one end of the molecule. Example: Glucose.
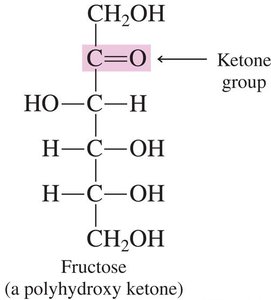
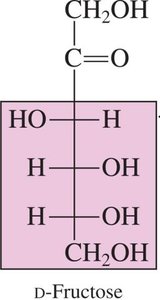
Ketoses: Contain a ketone group (C=O) typically at the second carbon atom. Example: Fructose.
Ring Structures of Monosaccharides
Monosaccharides with five or more carbon atoms commonly exist in cyclic (ring) forms in aqueous solutions. The ring formation involves a reaction between the carbonyl group and a hydroxyl group within the same molecule.
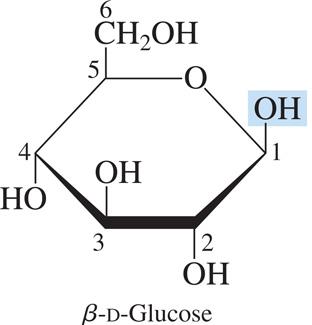
Pyranose: Six-membered ring structure (e.g., β-D-glucose).
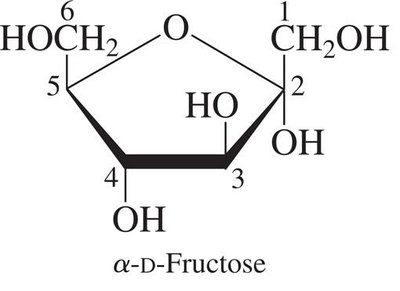
Furanose: Five-membered ring structure (e.g., α-D-fructose).
Stereochemistry of Carbohydrates
Chirality and Stereoisomerism
Many carbohydrates contain chiral centers, leading to the existence of stereoisomers. A chiral center is a carbon atom bonded to four different groups. Stereoisomers are molecules with the same molecular formula and sequence of bonded atoms, but different three-dimensional orientations.
Enantiomers: Stereoisomers that are nonsuperimposable mirror images of each other.
Diastereomers: Stereoisomers that are not mirror images.
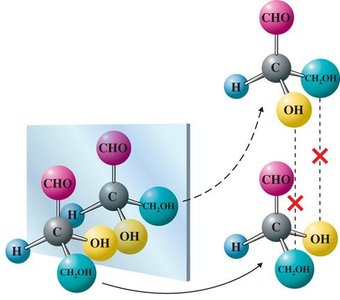
Chiral Carbons in Carbohydrates
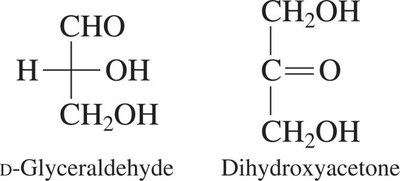
Each chiral carbon in a carbohydrate can give rise to different stereoisomers. For example, glyceraldehyde is the simplest carbohydrate with one chiral center.
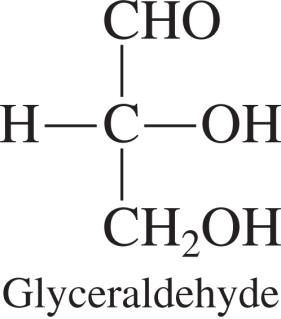
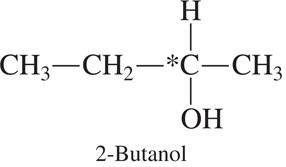
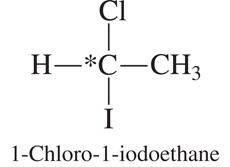
Examples of Chiral Centers
2-Butanol: Has one chiral center (marked with an asterisk).
1-Chloro-1-iodoethane: Another example of a molecule with a chiral center.
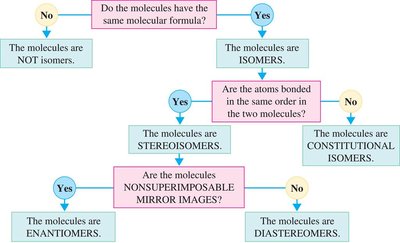
Isomerism in Carbohydrates
Carbohydrates can exist as different types of isomers:
Constitutional Isomers: Differ in the order of attachment of atoms.
Stereoisomers: Atoms are bonded in the same order but differ in spatial arrangement.
Enantiomers: Nonsuperimposable mirror images.
Diastereomers: Stereoisomers that are not mirror images.
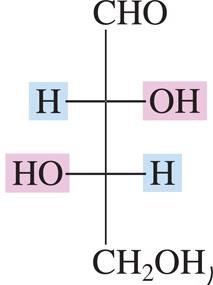
Fischer Projections and D/L Notation
Fischer Projections
Fischer projections are a two-dimensional representation of three-dimensional organic molecules. They are commonly used to depict carbohydrates and amino acids.
Horizontal lines represent bonds projecting out of the plane (toward the viewer).
Vertical lines represent bonds projecting behind the plane (away from the viewer).
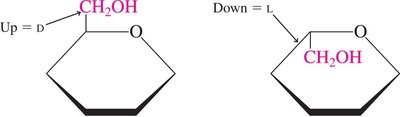
D- and L- Isomers
The D- and L- notation is based on the configuration of the chiral carbon farthest from the carbonyl group. In D-isomers, the hydroxyl group on this carbon is on the right; in L-isomers, it is on the left.
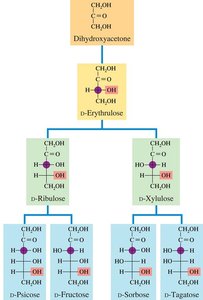
Classification of Monosaccharides by Carbon Number
Trioses, Tetroses, Pentoses, and Hexoses
Monosaccharides are classified by the number of carbon atoms:
Trioses: 3 carbons (e.g., glyceraldehyde, dihydroxyacetone)
Tetroses: 4 carbons
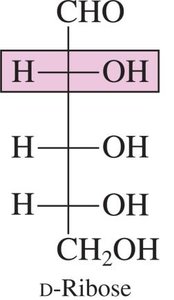
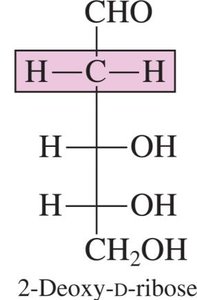
Pentoses: 5 carbons (e.g., ribose, deoxyribose)
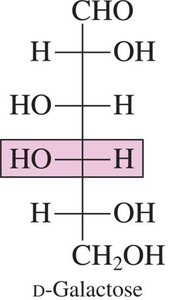
Hexoses: 6 carbons (e.g., glucose, galactose, fructose)
Common Monosaccharides and Their Structures
Glucose, Galactose, Fructose, Ribose, and Deoxyribose
These monosaccharides are essential for biological processes:
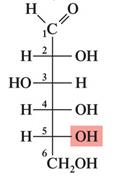
D-Glucose: The most common monosaccharide, also known as blood sugar.
D-Galactose: An epimer of glucose, differing at carbon 4.
D-Fructose: A ketohexose, found in fruit and honey.
D-Ribose: A component of RNA.
2-Deoxy-D-ribose: A component of DNA.
Cyclic Forms and Anomerism
Formation of Cyclic Structures
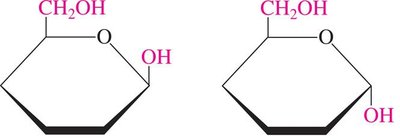
Monosaccharides with five or more carbons form cyclic hemiacetals or hemiketals. The carbonyl group reacts with a hydroxyl group to form a ring, creating a new chiral center called the anomeric carbon.
α-Anomer: The OH group on the anomeric carbon is trans to the CH2OH group.
β-Anomer: The OH group on the anomeric carbon is cis to the CH2OH group.
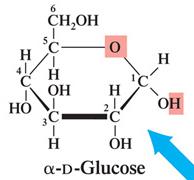
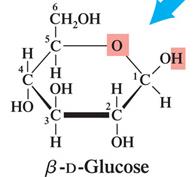
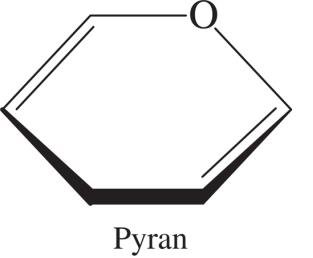
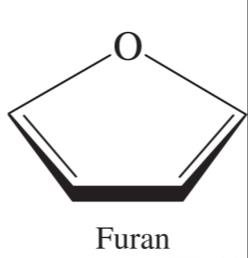
Pyranose and Furanose Rings
Pyranose rings are six-membered, while furanose rings are five-membered. These names are derived from the parent heterocyclic compounds pyran and furan.
Oxidation and Reduction of Monosaccharides
Redox Reactions of Carbohydrates
Monosaccharides can undergo oxidation and reduction reactions. Aldoses can be oxidized to form carboxylic acids, while reduction can convert carbonyl groups to alcohols.
Oxidation: Aldehyde group (–CHO) is oxidized to a carboxylic acid (–COOH).
Reduction: Carbonyl group is reduced to an alcohol.
Summary Table: Major Classes of Biochemical Substances
Class | Example(s) | Approximate % in Body |
|---|---|---|
Water | H2O | 70% |
Inorganic Salts | NaCl, KCl, etc. | 5% |
Proteins | Enzymes, structural proteins | 15% |
Lipids | Fats, oils | 8% |
Carbohydrates | Glucose, starch | 2% |
Nucleic Acids | DNA, RNA | 2% |
