Back
BackCarbohydrates: Structure, Properties, and Biological Significance
Study Guide - Smart Notes

Carbohydrates
Introduction
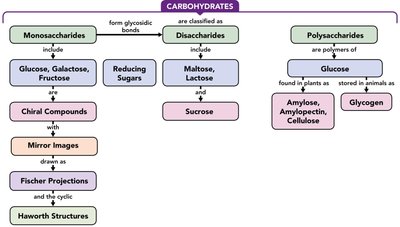
Carbohydrates are a fundamental class of biomolecules, essential for energy storage, structural integrity, and cellular communication in living organisms. They are classified into monosaccharides, disaccharides, and polysaccharides based on their complexity and structure. 
Chemical Properties of Monosaccharides
Oxidation of Monosaccharides
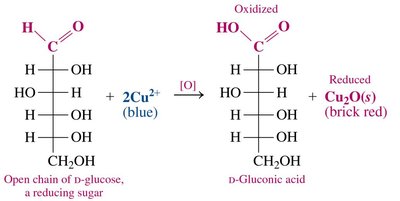
Monosaccharides in aqueous solution exist in equilibrium between their cyclic and open-chain forms. The open-chain form contains an aldehyde group (in aldoses) that can be oxidized to a carboxylic acid by oxidizing agents such as Benedict’s solution.
Oxidation Reaction: The aldehyde group is converted to a carboxylic acid, producing a sugar acid.
Reducing Sugars: Monosaccharides capable of reducing other substances (e.g., D-glucose) are termed reducing sugars.
Naming: The suffix -ose is replaced with -onic acid for the oxidized product.
Example: D-glucose oxidized by Cu2+ yields D-gluconic acid and Cu2O (brick red precipitate).

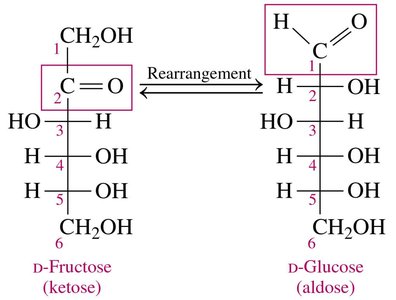
Oxidation: Fructose to Glucose
Fructose, a ketohexose, typically cannot be oxidized directly. However, in basic conditions (Benedict’s solution), it rearranges to form glucose, which can then be oxidized.
Rearrangement: The ketone group on carbon 2 and hydroxyl group on carbon 1 of fructose rearrange to form an aldehyde group.
Result: Fructose is converted to glucose, which is then oxidized.
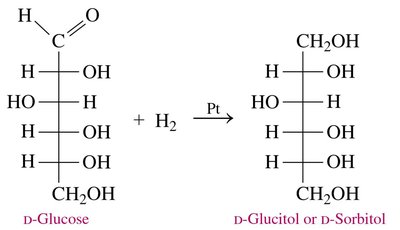
Reduction of Monosaccharides
Reduction of the carbonyl group in monosaccharides produces sugar alcohols (alditols).
Reaction: D-glucose is reduced to D-glucitol (D-sorbitol).
Naming: The suffix -ose is replaced with -itol for sugar alcohols.
Applications: Sugar alcohols are used as sweeteners in sugar-free products.
Examples of Sugar Alcohols
D-sorbitol: From D-glucose
D-xylitol: From D-xylose
D-mannitol: From D-mannose
Uses: Found in diet drinks, sugarless gum, and other sugar-free products.

Chemistry Link to Health: Testing for Glucose
Glucose in Urine (Glucosuria)
Normally, blood glucose is reabsorbed by the kidneys. When blood glucose exceeds ~160 mg/dL, excess glucose is excreted in urine, a condition called glucosuria.
Clinical Test: Glucose in urine is detected using a paper strip containing 2-methylaniline and enzymes (glucose oxidase, peroxidase).
Reaction: Glucose oxidase catalyzes the oxidation of glucose, producing hydrogen peroxide, which reacts with dye to produce a color change.
Interpretation: The intensity of the color correlates with glucose concentration.
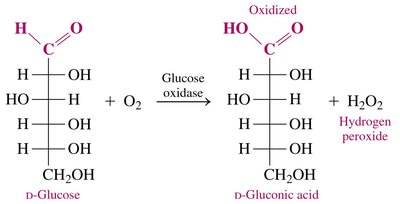
Table: Glucose Oxidase Test Results
Color | % (m/v) | mg/dL |
|---|---|---|
Blue | 0 | 0 |
Blue-green | 0.10 | 100 |
Green | 0.25 | 250 |
Green-brown | 0.50 | 500 |
Brown | 1.00 | 1000 |
Dark brown | 2.00 | 2000 |
Disaccharides
Formation and Structure
Disaccharides are formed by the linkage of two monosaccharides via a dehydration reaction, producing a glycosidic bond.
Common Disaccharides: Maltose, lactose, sucrose
Formation: Glucose + glucose → maltose + H2O; Glucose + galactose → lactose + H2O; Glucose + fructose → sucrose + H2O
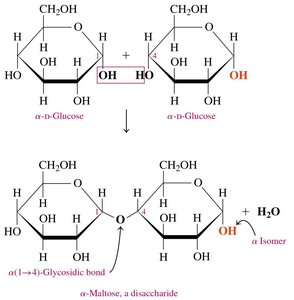
Maltose
Maltose (malt sugar) is composed of two α-D-glucose molecules linked by an α-1,4-glycosidic bond. It is obtained from starch hydrolysis and used in cereals, candies, and brewing. 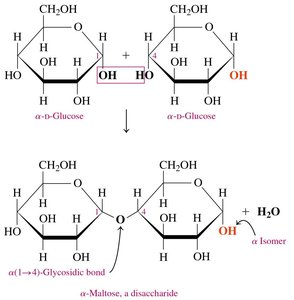
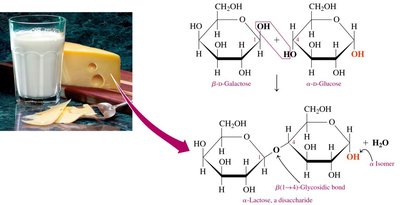
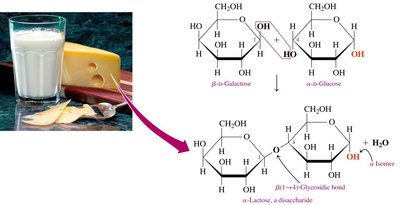
Lactose
Lactose (milk sugar) is a disaccharide found in milk, composed of β-D-galactose and α- or β-D-glucose, linked by a β-1,4-glycosidic bond.
Content: 6–8% in human milk, 4–5% in cow’s milk
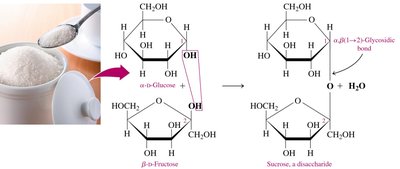
Sucrose
Sucrose (table sugar) is obtained from sugar cane and beets. It consists of α-D-glucose and β-D-fructose linked by an α,β-1,2-glycosidic bond. Sucrose cannot form an open chain and is not a reducing sugar. 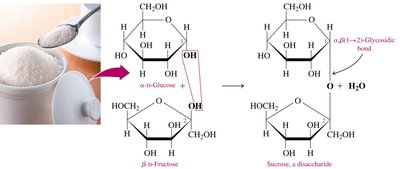
Sweetness of Sweeteners
Relative Sweetness
Sugars and artificial sweeteners differ in sweetness, measured relative to sucrose (assigned a value of 100).
Table: Relative Sweetness of Sugars and Artificial Sweeteners
Type | Sweetness Relative to Sucrose (=100) |
|---|---|
Galactose | 30 |
Glucose | 75 |
Fructose | 175 |
Lactose | 16 |
Maltose | 33 |
Sucrose | 100 |
Sorbitol | 60 |
Maltitol | 80 |
Xylitol | 100 |
Stevia | 15,000 |
Aspartame | 18,000 |
Saccharin | 45,000 |
Sucralose | 60,000 |
Neotame | 1,000,000 |
Advantame | 2,000,000 |
Artificial Sweeteners
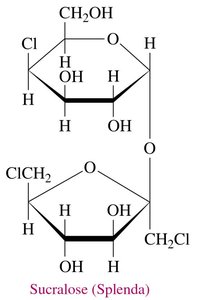
Sucralose: Marketed as Splenda, made from sucrose by replacing hydroxyl groups with chlorine atoms.
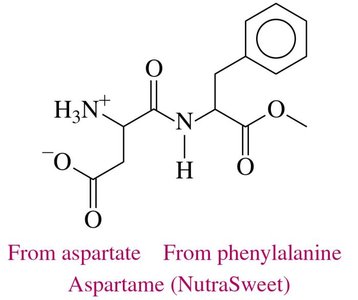
Aspartame: Marketed as NutraSweet and Equal, composed of aspartate and a methyl ester of phenylalanine.
Disaccharide Example: Melibiose
Melibiose is a disaccharide 30 times sweeter than sucrose, linked by an α-1,6-glycosidic bond. 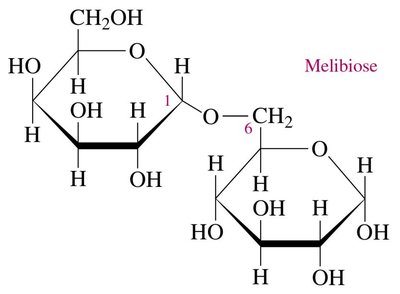

Polysaccharides
Structure and Types
Polysaccharides are polymers of monosaccharides, primarily glucose. Major types include amylose, amylopectin, glycogen, and cellulose.
Amylose: Straight-chain polymer of α-D-glucose, connected by α-1,4-glycosidic bonds.
Amylopectin: Branched-chain polymer, connected by α-1,4- and α-1,6-glycosidic bonds.
Glycogen: Highly branched polymer, similar to amylopectin but with more frequent α-1,6 branches.
Cellulose: Unbranched polymer of glucose, connected by β-1,4-glycosidic bonds; major structural unit in plants.
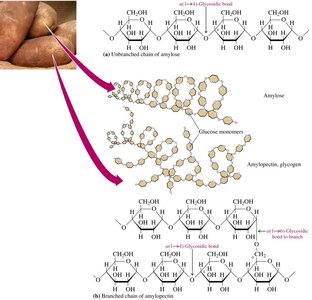
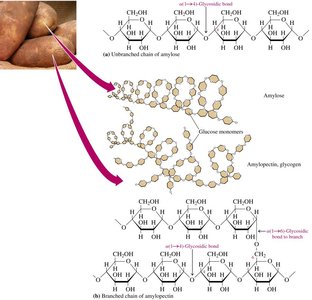
Amylose
Amylose makes up about 20% of starch and consists of 250–4000 α-D-glucose units in a continuous chain, coiled in a helical fashion.
Amylopectin
Amylopectin makes up 80% of starch, is branched, and contains both α-1,4 and α-1,6-glycosidic bonds. Starches hydrolyze easily to dextrins, maltose, and glucose.
Glycogen
Glycogen is stored in the liver and muscles of animals, hydrolyzed to maintain blood glucose levels. It is more highly branched than amylopectin, with α-1,6 branches every 10–15 glucose units.
Cellulose
Cellulose is the major structural unit of wood and plant fibers. It is insoluble in water, resistant to hydrolysis, and cannot be digested by humans due to the β-1,4-glycosidic bonds. 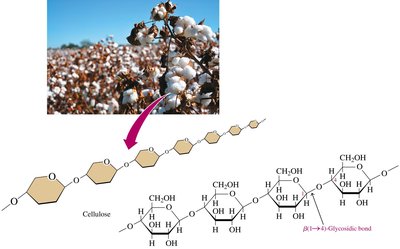
Learning Check: Polysaccharides and Glycosidic Bonds
Amylose: α-1,4-glycosidic bonds
Amylopectin: α-1,4 and α-1,6-glycosidic bonds
Cellulose: β-1,4-glycosidic bonds
Glycogen: α-1,4 and α-1,6-glycosidic bonds




Carbohydrates—Concept Map
A concept map summarizes the classification and relationships among monosaccharides, disaccharides, and polysaccharides, highlighting their structural features and biological roles.