Back
BackCarboxylic Acids and Esters: Structure, Properties, and Reactions
Study Guide - Smart Notes

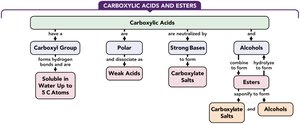
Carboxylic Acids and Esters
Introduction
Carboxylic acids and esters are important classes of organic compounds with significant roles in biological systems, industry, and daily life. This chapter explores their structures, nomenclature, properties, and reactions, providing foundational knowledge for further study in organic and biological chemistry.
Carboxylic Acids
Structure of Carboxylic Acids
Carboxylic acids contain a carboxyl group (–COOH), which consists of a carbonyl group (C=O) bonded to a hydroxyl group (–OH) on the same carbon atom.
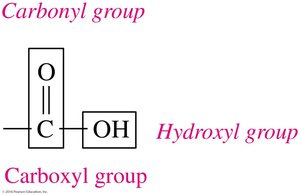
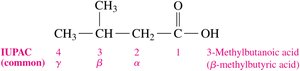
Naming Carboxylic Acids
Carboxylic acids are named using both IUPAC and common naming systems:
IUPAC Naming: Replace the -e ending of the parent alkane with -oic acid. Number the chain starting from the carboxyl carbon (carbon 1).
Common Naming: Use prefixes such as form-, acet-, propion-, and butyr-. Greek letters (α, β, γ) are used to indicate positions relative to the carboxyl group.
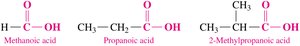
Examples of Common Carboxylic Acids
Methanoic acid (formic acid): Found in ant stings.
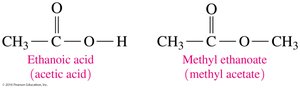
Ethanoic acid (acetic acid): Responsible for the sour taste of vinegar.


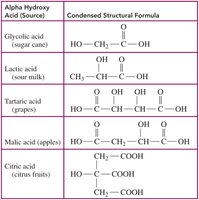
Alpha Hydroxy Acids (AHAs)
AHAs are naturally occurring carboxylic acids with a hydroxyl group on the carbon adjacent to the carboxyl group. They are used in skin care for exfoliation and treating pigmentation.
Preparation of Carboxylic Acids
Carboxylic acids can be synthesized by the oxidation of primary alcohols or aldehydes.
Example: Ethanol oxidizes to acetaldehyde, then to acetic acid (vinegar).
Properties of Carboxylic Acids
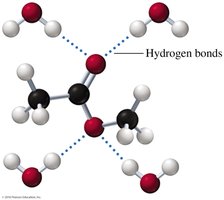
Polarity and Hydrogen Bonding
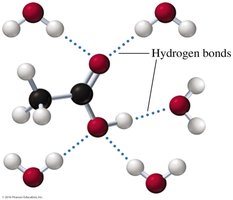
Carboxylic acids are highly polar due to the presence of both carbonyl and hydroxyl groups, allowing them to form strong hydrogen bonds.
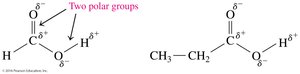
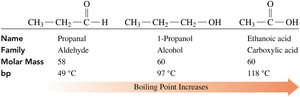
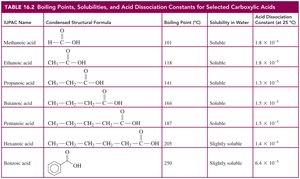
Boiling Points
Carboxylic acids have higher boiling points than alcohols, ketones, and aldehydes of similar molar mass due to extensive hydrogen bonding and dimer formation.

Solubility in Water
Carboxylic acids with one to five carbon atoms are very soluble in water due to hydrogen bonding. Solubility decreases as the carbon chain length increases.
Acidity and Dissociation
Carboxylic acids are weak acids that partially dissociate in water to produce carboxylate ions and hydronium ions. The negative charge is stabilized by resonance between the two oxygen atoms.
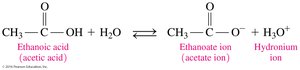
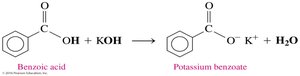
Neutralization Reactions
Carboxylic acids react with strong bases (e.g., NaOH, KOH) to form carboxylate salts and water. The carboxylate ion is named by replacing the -ic acid ending with -ate.
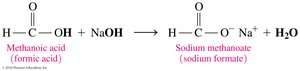
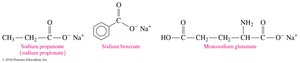
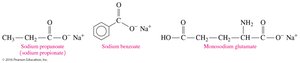
Carboxylic Acids as Preservatives and Flavor Enhancers
Carboxylate salts such as sodium propionate, sodium benzoate, and monosodium glutamate (MSG) are used as food preservatives and flavor enhancers.
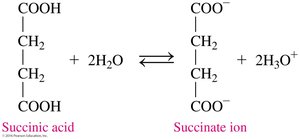
Carboxylic Acids in Metabolism
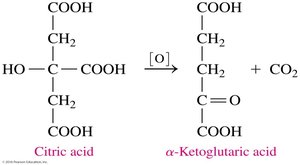
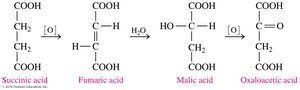
Carboxylate ions play key roles in metabolic pathways such as the citric acid cycle (Krebs cycle). For example, succinate, citrate, and α-ketoglutarate are intermediates in energy production.


Esters
Structure and Synthesis of Esters
Esters are derived from carboxylic acids and alcohols. They are commonly found in fats, oils, and as flavor and fragrance compounds in fruits and flowers.

Esterification Reaction
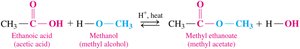
Esterification is the reaction of a carboxylic acid with an alcohol in the presence of an acid catalyst and heat, producing an ester and water. This is an equilibrium reaction.
Esters in Medicine and Industry
Esters such as acetylsalicylic acid (aspirin) and methyl salicylate (oil of wintergreen) are important in pharmaceuticals and topical treatments.
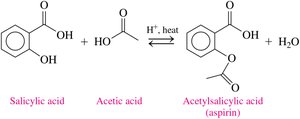
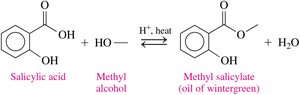
Esters in the Environment
Polyesters such as Dacron and PETE are produced from esterification reactions and are used in fabrics, plastic bottles, and medical devices.
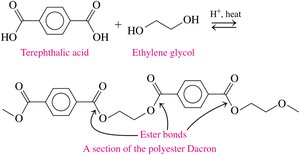
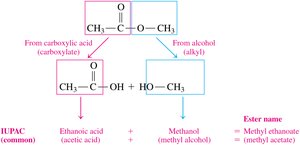
Naming Esters
The name of an ester consists of two parts:
The alkyl group from the alcohol (first word).
The carboxylate name from the acid (second word, replacing -ic acid with -ate).

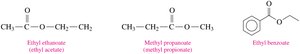
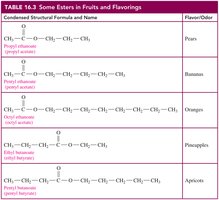
Esters in Fruits and Flavorings
Many esters are responsible for the characteristic flavors and odors of fruits and flowers. Small esters are volatile and soluble in water, making them detectable by taste and smell.

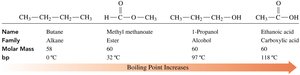
Properties of Esters
Boiling Points
Esters have boiling points higher than alkanes and ethers but lower than alcohols and carboxylic acids of similar molar mass. They cannot form hydrogen bonds with each other due to the absence of a hydroxyl group.

Solubility in Water
Esters with two to five carbon atoms are soluble in water due to hydrogen bonding between the carbonyl oxygen and water. Solubility decreases with increasing carbon chain length.
Reactions of Esters
Acid Hydrolysis of Esters
In the presence of water, acid catalyst, and heat, esters hydrolyze to form a carboxylic acid and an alcohol. This is the reverse of esterification.
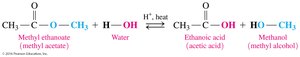


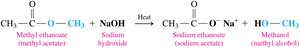
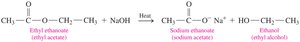
Base Hydrolysis (Saponification)
Base hydrolysis of esters (saponification) involves reaction with a strong base (e.g., NaOH) to produce a carboxylate salt and an alcohol.


Summary Concept Map