 Back
BackCarboxylic Acids
Study Guide - Smart Notes

Carboxylic Acids
Introduction to Carboxylic Acids
Carboxylic acids are a class of organic compounds characterized by the presence of a carboxyl group (-COOH). This functional group consists of a carbonyl group (C=O) directly bonded to a hydroxyl group (-OH). Carboxylic acids are widely found in nature and play essential roles in biological and chemical processes.
Nomenclature of Carboxylic Acids
IUPAC Naming Rules
The IUPAC system names carboxylic acids by replacing the "-e" ending of the corresponding alkane with "-oic acid." The carboxyl carbon is always assigned as carbon #1. Substituents are numbered from this carbon.
Methanoic acid (from methane, CH4)
Ethanoic acid (from ethane, CH3CH3)
Substituents are indicated by their position and name, e.g., 3-methylbutanoic acid, 2,3-dichlorobenzoic acid.
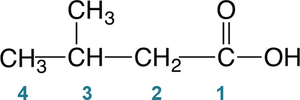
Common Names of Carboxylic Acids
Many carboxylic acids have traditional common names, such as formic acid (methanoic acid) and acetic acid (ethanoic acid). These names are often used in biological and industrial contexts.
Examples of Carboxylic Acids in Nature
Formic acid is found in ant stings.
Acetic acid gives vinegar its sour taste.
Example: Naming Carboxylic Acids
To name a carboxylic acid:
Identify the longest carbon chain containing the carboxyl group and replace the "-e" with "-oic acid" (e.g., pentanoic acid).
Number the chain starting from the carboxyl carbon and name/number any substituents (e.g., 2-methylpentanoic acid).
Properties of Carboxylic Acids
Polarity and Hydrogen Bonding
Carboxylic acids are strongly polar due to the presence of both a hydroxyl group and a carbonyl group. This allows them to form extensive hydrogen bonds with water and with each other.
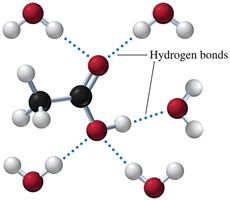
Solubility in Water
Carboxylic acids with 1-5 carbon atoms are very soluble in water due to hydrogen bonding.
As the carbon chain length increases, solubility decreases.
Acidity of Carboxylic Acids
Weak Acids and Ionization
Carboxylic acids are weak acids; they partially ionize in water to produce carboxylate ions and hydronium ions. The negative charge on the carboxylate ion is stabilized by resonance between the two oxygen atoms.
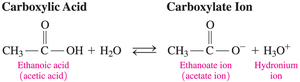
General dissociation equation:
Neutralization of Carboxylic Acids
Formation of Carboxylate Salts
Carboxylic acids react with strong bases (such as NaOH or KOH) to form carboxylate salts and water. These salts are ionic compounds, typically solid at room temperature, with high melting points and good water solubility. They are used as preservatives and flavor enhancers.
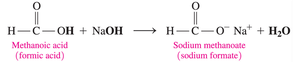
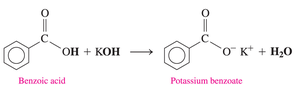
The carboxylate ion is named by replacing "-ic acid" with "-ate" (e.g., acetate, benzoate).
Carboxylic Acids in Metabolism
Role in Cellular Metabolism
Carboxylic acids are integral to metabolic pathways. For example, during glycolysis, glucose is broken down into pyruvate (the carboxylate salt of pyruvic acid). Under anaerobic conditions, pyruvic acid is reduced to lactic acid or the lactate ion.

The Citric Acid Cycle (Krebs Cycle)
The citric acid cycle is a series of reactions in which di- and tricarboxylic acids are oxidized and decarboxylated to produce energy for cells. Key steps include:
Oxidation of citric acid to α-ketoglutaric acid (5 carbons)
Decarboxylation to succinic acid (4 carbons)
Conversion of succinic acid to oxaloacetic acid
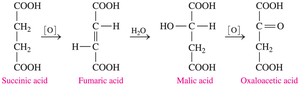

Additional info: Many functional groups studied in organic chemistry, such as carboxyl, are directly involved in these metabolic processes.
