 Back
BackChapter 1: Chemistry, Matter, and Measurement – Structured Study Notes
Study Guide - Smart Notes

Chapter 1: Chemistry, Matter, and Measurement
Chemistry: The Central Science
Chemistry is the study of the nature, properties, and transformations of matter. It is considered the central science because it connects and overlaps with many other scientific and health-related disciplines.
Matter: Anything that has mass and occupies space; includes all physical material in the universe.
Scientific Method: The process of observation, hypothesis, and experimentation used to expand scientific knowledge.
Properties: Characteristics useful for identifying substances, such as size, color, temperature, chemical composition, and chemical reactivity.
Physical and Chemical Changes
Changes in matter can be classified as physical or chemical. Understanding these changes is fundamental to chemistry.
Physical Change: A change that does not affect the chemical makeup of a substance (e.g., boiling water, dissolving salt, chopping vegetables).
Chemical Change: A change that alters the chemical composition of a substance (e.g., burning potassium in water, combustion reactions).
Examples of Physical Changes:
Boiling water
Making salt water
Chopping vegetables



Examples of Chemical Changes:
Combustion: Reacting fuel with oxygen to release energy.
Sugar heated to make caramel.



Classification of Matter
Matter can be classified based on its composition and uniformity.
Pure Substance: Has a uniform chemical composition throughout (e.g., sugar, water).
Mixture: A blend of two or more substances, each retaining its chemical identity.
Homogeneous Mixture: Uniform composition throughout (e.g., salt water).
Heterogeneous Mixture: Nonuniform composition with distinct regions (e.g., oil and water).



Element: A fundamental substance that cannot be broken down chemically into simpler substances.
Compound: A pure substance that can be broken down into simpler substances by chemical reactions.
Chemical Elements and Symbols
Elements are represented by unique symbols, and compounds are written using these symbols and subscripts to indicate the number of atoms.
Chemical Formula: Notation using element symbols and subscripts (e.g., H2O for water).
Subscripts indicate the number of atoms of each element in a compound.
Chemical Reactions
Chemical reactions involve the transformation of reactants into products, often accompanied by observable changes such as color change or gas production.
Chemical Reaction: A process in which the identity and composition of substances are changed.
Reactant: Starting substance in a chemical reaction.
Product: Substance formed as a result of a chemical reaction.
Physical Quantities: Units and Scientific Notation
Physical properties are measured using standardized units. Scientific notation is used to express very large or small numbers.
Physical Quantity: A property that can be measured (e.g., mass, volume, temperature).
Unit: Standard of measurement (e.g., kilogram, meter, liter).
SI Units: International System of Units (kg, m, s, K).
Prefixes: Used to modify unit size (e.g., kilo-, centi-, milli-, micro-).
Scientific Notation: Expresses numbers as a product of a number between 1 and 10 and a power of 10.
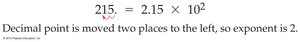
Measuring Mass, Length, and Volume
Mass, length, and volume are fundamental measurements in chemistry, each with specific units in the SI and metric systems.
Mass: Amount of matter in an object (measured in kilograms or grams).
Weight: Gravitational force exerted on an object.
Length: Measured in meters.
Volume: Amount of space occupied, measured in liters or cubic meters.
Measurement and Significant Figures
Significant figures indicate the precision of a measurement. Rules help determine which digits are significant.
Significant Figures: Meaningful digits in a measurement.
Rules for significant figures:
Zeroes in the middle are significant.
Leading zeroes are not significant.
Trailing zeroes after a decimal are significant.
Trailing zeroes before an implied decimal may or may not be significant.
Rounding Off Numbers
Rounding is used to delete nonsignificant figures, ensuring results reflect the precision of the original data.
Multiplication/division: Answer cannot have more significant figures than the original numbers.
Addition/subtraction: Answer cannot have more digits after the decimal point than the original numbers.
Rules for rounding:
If the first digit removed is four or less, drop it.
If the first digit removed is five or greater, round up.
Unit Conversions and Estimating Answers
Unit conversions are essential in laboratory and medical settings. The factor-label method is used to convert between units.
Factor-Label Method: Sets up equations so unwanted units cancel, leaving only desired units.
Conversion Factor: Expresses the numerical relationship between two units.
Steps for problem solving:
Identify given information and units.
Identify needed information and units.
Find relationships and plan steps.
Solve the problem.
Temperature, Heat, and Energy
All chemical reactions involve energy changes. Temperature is a measure of heat energy, and different scales are used in science.
Energy: Capacity to do work or supply heat.
Temperature: Measure of heat energy; reported in Fahrenheit, Celsius, or Kelvin.
Kelvin Scale: Absolute zero is 0 K.
Celsius Scale: Freezing point of water is 0°C.
Fahrenheit Scale: Freezing point of water is 32°F.
Conversion equations:
Density and Specific Gravity
Density relates mass to volume and is used to identify substances and predict their behavior in mixtures.
Density: Mass per unit volume; expressed in g/cm3 for solids and g/mL for liquids.
Less dense substances float on more dense fluids; more dense substances sink.
Formula for Density:
Summary Table: Classification of Matter
Type | Description | Example |
|---|---|---|
Element | Cannot be broken down chemically | Oxygen (O2) |
Compound | Can be broken down chemically | Water (H2O) |
Homogeneous Mixture | Uniform composition | Salt water |
Heterogeneous Mixture | Nonuniform composition | Oil and water |
Additional info: Academic context and examples have been expanded for clarity and completeness. Images included only where directly relevant to the explanation.
