 Back
BackChapter 11: Introduction to Organic Chemistry – Hydrocarbons
Study Guide - Smart Notes

Organic Compounds
Definition and Characteristics
Organic compounds are molecules primarily composed of carbon atoms, often bonded with hydrogen, and may include oxygen, sulfur, nitrogen, and halogens (Cl, Br, I). The formulas are written with carbon first, followed by hydrogen, then other elements. Organic compounds are prevalent in everyday products such as gasoline, medicines, shampoos, plastics, and perfumes. In contrast, inorganic compounds are typically composed of metals and nonmetals, such as sodium chloride (NaCl).
Key Point 1: Organic compounds always contain at least one carbon atom.
Key Point 2: They are often covalently bonded and nonpolar, unless a strongly electronegative atom is present.
Example: Gasoline (hydrocarbon), NaCl (inorganic compound).
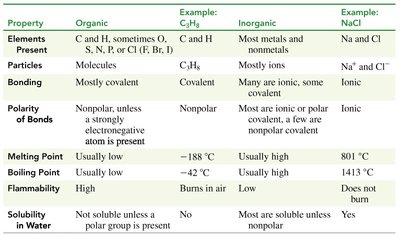
Additional info: Organic compounds are usually low melting and boiling, flammable, and not water soluble unless a polar group is present.
Hydrocarbons
Structure and Properties
Hydrocarbons are organic compounds consisting solely of carbon and hydrogen atoms. Carbon atoms form chains or rings, each making four covalent bonds. Hydrocarbons are classified as nonpolar molecules, generally less dense than water, and have low boiling points. As the molecular weight increases, melting and boiling points, as well as density, increase.
Key Point 1: Hydrocarbons are nonpolar and not water soluble; they dissolve in nonpolar organic solvents.
Key Point 2: Their physical properties change with molecular size.
Example: Gasoline, composed of hydrocarbons, is less dense than water and not water soluble.
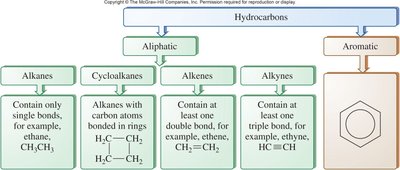
Classification of Hydrocarbons
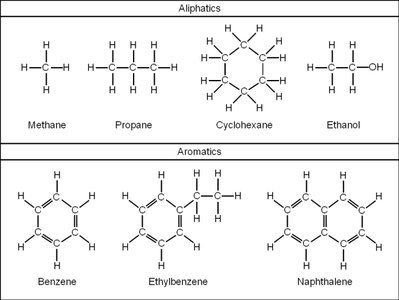
Aliphatic and Aromatic Hydrocarbons
Hydrocarbons are divided into two main categories: aliphatic and aromatic. Aliphatic hydrocarbons include alkanes, cycloalkanes, alkenes, and alkynes. Aromatic hydrocarbons contain benzene rings.
Key Point 1: Alkanes contain only single bonds (saturated hydrocarbons).
Key Point 2: Alkenes contain at least one double bond; Alkynes contain at least one triple bond.
Key Point 3: Aromatic compounds have a benzene ring structure.
Example: Ethane (alkane), ethene (alkene), ethyne (alkyne), benzene (aromatic).
Alkanes
General Formula and Structure
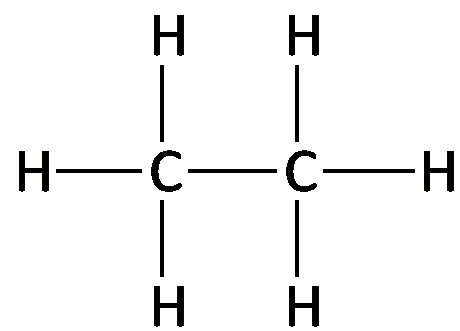
Alkanes are saturated hydrocarbons with only carbon and hydrogen atoms, connected by single bonds. The general formula for a straight-chain alkane is , where n is the number of carbon atoms. Alkanes can be represented by molecular, structural, condensed, and line-angle formulas.
Key Point 1: Alkanes are nonpolar, saturated hydrocarbons.
Key Point 2: Molecular formula shows the number and type of atoms; structural formula shows bonding.
Example: Ethane (), Propane ().
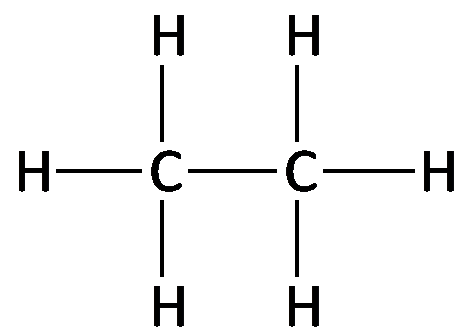
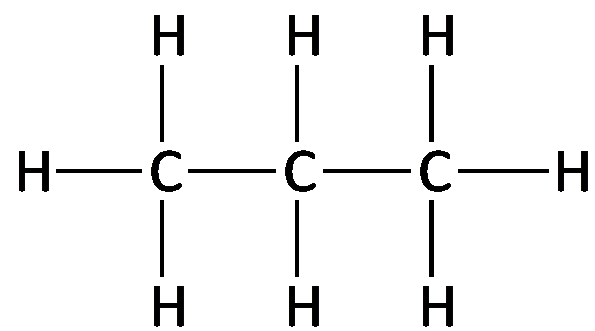
Alkanes: Naming and Formulas
IUPAC Naming System
The names of alkanes are determined by the IUPAC system and end in "-ane." Alkanes with 1-4 carbons use prefixes: methane, ethane, propane, butane. Alkanes with 5-10 carbons use Greek prefixes: pentane, hexane, heptane, octane, nonane, decane.
Key Point 1: Prefixes indicate the number of carbon atoms.
Key Point 2: The molecular formula follows .
Example: Butane (), Octane ().
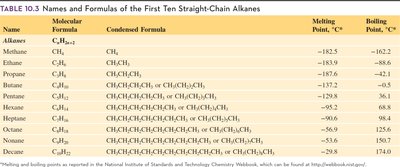
Additional info: Melting and boiling points increase with chain length.
Isomers and Alkyl Groups
Structural Isomers and Branching
Isomers are compounds with the same molecular formula but different structural arrangements. When an alkane has four or more carbon atoms, branching can occur, forming structural (constitutional) isomers. Alkyl groups are formed by removing one hydrogen atom from an alkane, and are named by replacing "-ane" with "-yl." These groups act as substituents on the main carbon chain.
Key Point 1: Isomers have identical molecular formulas but different connectivity.
Key Point 2: Alkyl groups form branches on the main chain.
Example: Butane and methylpropane are isomers of .
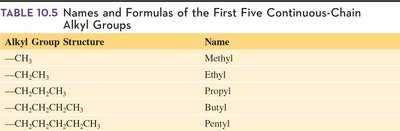
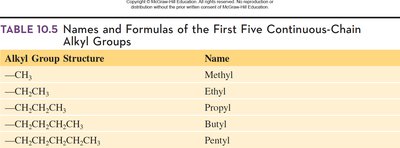
Additional info: The first five alkyl groups are methyl, ethyl, propyl, butyl, and pentyl.
