 Back
BackChapter 11: Introduction to Organic Chemistry – Hydrocarbons
Study Guide - Smart Notes

Introduction to Organic Chemistry
Overview of Organic Compounds
Organic chemistry is the study of compounds primarily composed of carbon and hydrogen, often with oxygen, nitrogen, sulfur, or halogens. These compounds are foundational to life and are found in fuels, medicines, plastics, and more. In contrast, inorganic compounds typically consist of metals and nonmetals and do not always contain carbon.
Organic compounds contain at least one carbon atom, usually bonded to hydrogen, and may include O, S, N, or halogens.
Inorganic compounds are composed of metals and nonmetals, such as sodium chloride (NaCl).
Formulas for organic compounds are written with carbon first, then hydrogen, followed by other elements.
Examples: Propane (C3H8) is an organic fuel; sodium chloride (NaCl) is an inorganic salt.
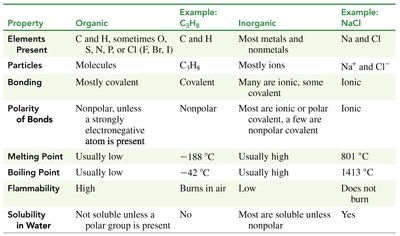
Table Purpose: This table compares the properties of organic and inorganic compounds, including elements present, bonding, melting/boiling points, and solubility.
Hydrocarbons
Definition and Structure
Hydrocarbons are the simplest organic compounds, consisting only of carbon and hydrogen. They can form chains or rings, with each carbon atom forming four covalent bonds. Hydrocarbons are classified as either aliphatic (chains or non-aromatic rings) or aromatic (containing benzene-like rings).
Aliphatic hydrocarbons include alkanes, alkenes, alkynes, and cycloalkanes.
Aromatic hydrocarbons contain benzene rings and related structures.
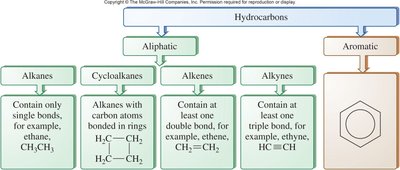
Table Purpose: This diagram classifies hydrocarbons into aliphatic and aromatic types, with examples of each subclass.
Physical Properties of Hydrocarbons
Hydrocarbons are non-polar molecules, making them insoluble in water but soluble in nonpolar solvents.
They are generally less dense than water.
Hydrocarbons have low boiling points and low melting points compared to inorganic compounds.
As the molecular weight (chain length) increases, melting and boiling points, as well as density, increase.
Alkanes
Structure and General Formula
Alkanes are saturated hydrocarbons, meaning they contain only single bonds between carbon atoms. The general formula for a straight-chain alkane is:
n = number of carbon atoms
Each carbon forms four single covalent bonds (either with hydrogen or other carbons).
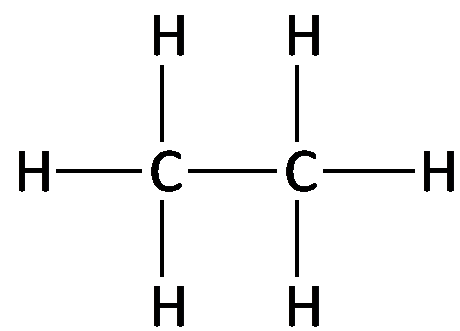
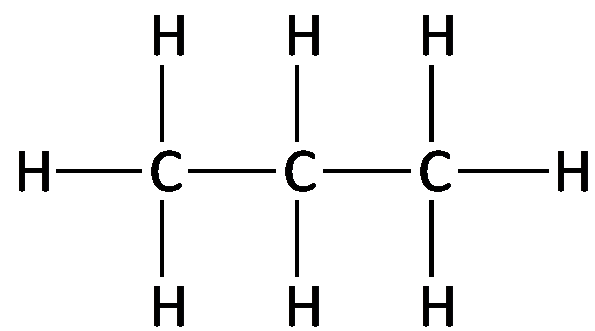
Examples: Methane (CH4), Ethane (C2H6), Propane (C3H8), Butane (C4H10).
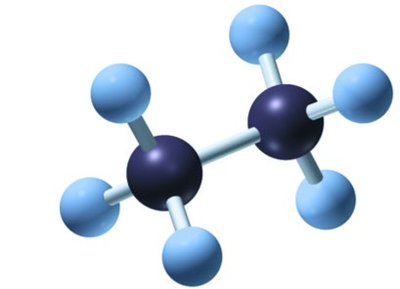
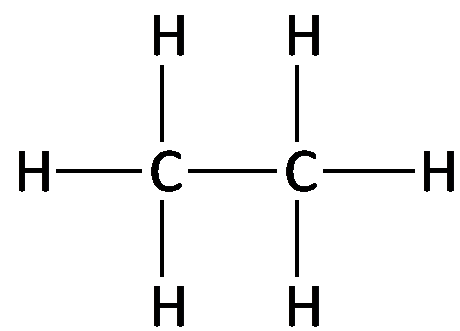
Formulas Used in Organic Chemistry
Molecular formula: Shows the number and type of atoms (e.g., C2H6).
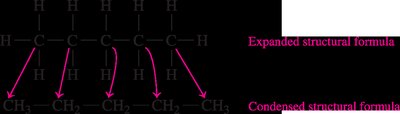
Structural formula: Shows each atom and bond explicitly.
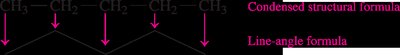
Condensed formula: Groups atoms to show connectivity (e.g., CH3CH2CH3).
Line-angle formula: Represents the carbon skeleton with lines; each vertex or end represents a carbon atom.
Naming Alkanes (IUPAC System)
The names of alkanes are determined by the International Union of Pure and Applied Chemistry (IUPAC) system. All alkane names end in "-ane." The prefixes indicate the number of carbon atoms in the chain.
Name | # Carbons | Molecular Formula |
|---|---|---|
Methane | 1 | CH4 |
Ethane | 2 | CH3CH3 |
Propane | 3 | CH3CH2CH3 |
Butane | 4 | CH3CH2CH2CH3 |
Pentane | 5 | CH3CH2CH2CH2CH3 |
Hexane | 6 | CH3CH2CH2CH2CH2CH3 |
Heptane | 7 | CH3(CH2)5CH3 |
Octane | 8 | CH3(CH2)6CH3 |
Nonane | 9 | CH3(CH2)7CH3 |
Decane | 10 | CH3(CH2)8CH3 |
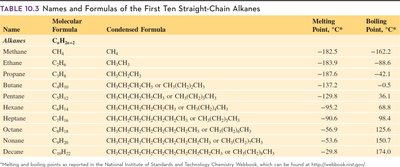
Table Purpose: This table lists the names, formulas, and melting/boiling points of the first ten straight-chain alkanes.
Physical Properties and Trends in Alkanes
Alkanes are nonpolar and insoluble in water.
They have low melting and boiling points, which increase with molecular weight (chain length).
Alkanes are less dense than water and are flammable.
Example: Methane (CH4) boils at -162°C, while decane (C10H22) boils at 174°C.
Examples and Applications
Propane (C3H8): Used as a fuel for heating and cooking.
Octane (C8H18): A major component of gasoline.
Butane (C4H10): Used in lighters and portable stoves.
Summary Table: Properties of Organic vs. Inorganic Compounds
Property | Organic | Example: C3H8 | Inorganic | Example: NaCl |
|---|---|---|---|---|
Elements Present | C and H, sometimes O, S, N, P, or Cl | C3H8 | Most metals and nonmetals | Na and Cl |
Particles | Molecules | C3H8 | Mostly ions | Na+ and Cl- |
Bonding | Mostly covalent | Covalent | Many are ionic, some covalent | Ionic |
Polarity of Bonds | Nonpolar, unless a strongly electronegative atom is present | Nonpolar | Most are ionic or polar covalent | Ionic |
Melting Point | Usually low | -188°C | Usually high | 801°C |
Boiling Point | Usually low | -42°C | Usually high | 1413°C |
Flammability | High | Burns in air | Low | Does not burn |
Solubility in Water | Not soluble unless a polar group is present | No | Most are soluble unless nonpolar | Yes |
Visualizing Hydrocarbons
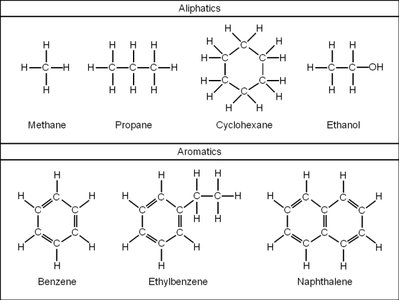
Figure Purpose: This figure shows the structures of common aliphatic (methane, propane, cyclohexane, ethanol) and aromatic (benzene, ethylbenzene, naphthalene) hydrocarbons, illustrating the diversity of organic molecules.
Practice Problem Example
Question: An alkane used as a motor lubricant contains 10 carbon atoms. How many hydrogen atoms are present?
Solution: Use the general formula with n = 10:
Answer: 22 hydrogen atoms
Key Takeaways
Organic compounds are carbon-based and have distinct properties compared to inorganic compounds.
Hydrocarbons are the simplest organic compounds and are classified as alkanes, alkenes, alkynes, cycloalkanes, or aromatics.
Alkanes are saturated hydrocarbons with the general formula .
Physical properties such as melting and boiling points increase with molecular size in alkanes.
