 Back
BackChapter 11: Introduction to Organic Chemistry – Alkanes, Alkenes, and Alkynes
Study Guide - Smart Notes

Introduction to Organic Chemistry
Overview of Organic Compounds
Organic chemistry is the study of compounds primarily composed of carbon and hydrogen, often with oxygen, nitrogen, sulfur, phosphorus, and halogens. Organic compounds are distinguished from inorganic compounds by their bonding, structure, and properties.
Organic compounds contain mostly covalent bonds and are often nonpolar.
Inorganic compounds typically contain ionic bonds and are often polar.
Examples: Methane (CH4) is organic; sodium chloride (NaCl) is inorganic.
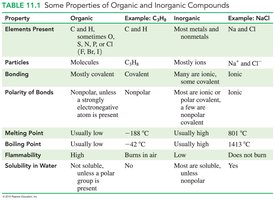
Table Purpose: Comparison of physical and chemical properties between organic and inorganic compounds.
Hydrocarbons
Types and Structure
Hydrocarbons are organic compounds containing only carbon and hydrogen. They are classified based on the types of bonds between carbon atoms:
Saturated hydrocarbons (alkanes): Only single bonds between carbon atoms.
Unsaturated hydrocarbons: Contain double (alkenes) or triple (alkynes) bonds.
Methane and Ethane Structures
Methane (CH4) and ethane (C2H6) are the simplest alkanes. Their three-dimensional structures are tetrahedral, with bond angles of 109°.
Methane: Four covalent bonds to hydrogen.
Ethane: Each carbon forms three bonds to hydrogen and one to another carbon.
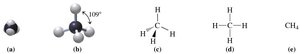
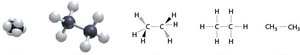
Alkanes: Structure and Naming
IUPAC Naming of Alkanes
The International Union of Pure and Applied Chemistry (IUPAC) system names alkanes based on the number of carbon atoms in the longest continuous chain. The first ten alkanes are:
Number of Carbon Atoms | IUPAC Name | Molecular Formula | Condensed Structural Formula | Line-Angle Formula |
|---|---|---|---|---|
1 | Methane | CH4 | CH4 | --- |
2 | Ethane | C2H6 | CH3-CH3 | --- |
3 | Propane | C3H8 | CH3-CH2-CH3 | --- |
4 | Butane | C4H10 | CH3-CH2-CH2-CH3 | --- |
5 | Pentane | C5H12 | CH3-CH2-CH2-CH2-CH3 | --- |
6 | Hexane | C6H14 | CH3-CH2-CH2-CH2-CH2-CH3 | --- |
7 | Heptane | C7H16 | CH3-CH2-CH2-CH2-CH2-CH2-CH3 | --- |
8 | Octane | C8H18 | CH3-CH2-CH2-CH2-CH2-CH2-CH2-CH3 | --- |
9 | Nonane | C9H20 | CH3-CH2-CH2-CH2-CH2-CH2-CH2-CH2-CH3 | --- |
10 | Decane | C10H22 | CH3-CH2-CH2-CH2-CH2-CH2-CH2-CH2-CH2-CH3 | --- |

Structural and Condensed Formulas
Alkanes can be represented by expanded structural formulas, condensed structural formulas, and line-angle formulas. Condensed formulas group each carbon atom with its attached hydrogens.
Expanded formula: Shows all bonds.
Condensed formula: Groups atoms for simplicity.
Line-angle formula: Each vertex or line end represents a carbon atom.
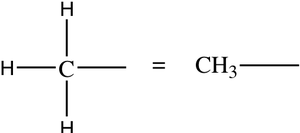
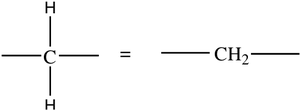
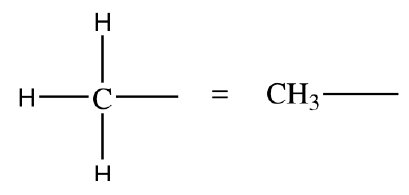
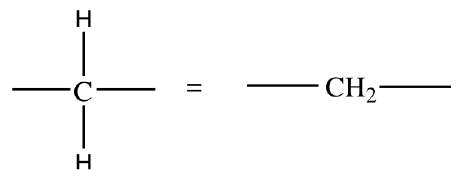
Cycloalkanes
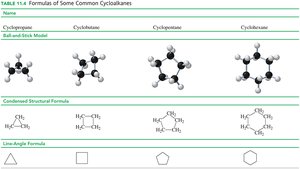
Cycloalkanes are alkanes in which carbon atoms form a ring. They are named by adding the prefix 'cyclo-' to the alkane name.
Examples: Cyclopropane, cyclobutane, cyclopentane, cyclohexane.
Alkanes with Substituents
Structural Isomers
Structural isomers have the same molecular formula but different arrangements of atoms. For example, butane (C4H10) exists as a straight chain and a branched chain.
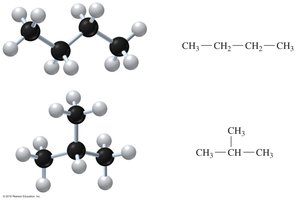
Alkyl and Halo Substituents
Substituents are groups attached to the main carbon chain. Alkyl groups are carbon branches, named with an 'yl' ending. Halo groups are halogen atoms (fluoro, chloro, bromo, iodo).
IUPAC Naming Rules for Substituted Alkanes
The IUPAC system provides rules for naming alkanes with substituents:
Find the longest chain or cycle; this serves as the root.
If the chain and cycle have the same number of carbons, the chain is the root.
Substituents are listed before the root, identified by number position, in alphabetical order.
Number the chain so that substituents have the lowest possible numbers.
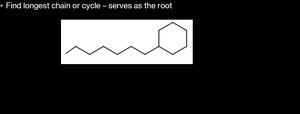
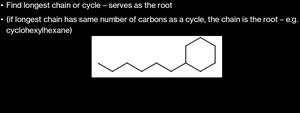
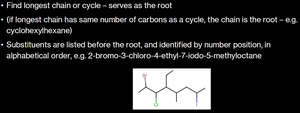


Example: Drawing and Naming Substituted Alkanes
To draw and name a substituted alkane, follow these steps:
Draw the main chain of carbon atoms.
Number the chain and place substituents on indicated carbons.
Add the correct number of hydrogen atoms to each carbon.

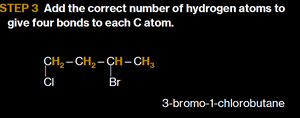
Example: Naming Alkanes
Write the alkane name for the longest chain, number the carbons from the end nearer a substituent, and give the location and name of each substituent in alphabetical order.

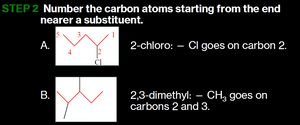
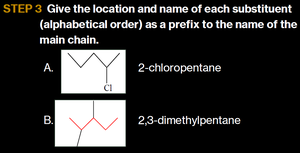
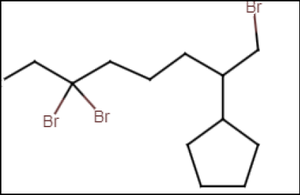
Example: Drawing Formulas for Alkanes
Draw the structure for 1,6,6-tribromo-2-cyclopentyloctane by identifying the main chain and placing substituents accordingly.

Properties of Alkanes
Solubility and Density
Alkanes are nonpolar, insoluble in water, less dense than water, and flammable. They are commonly found in crude oil and float on water during oil spills.

Combustion of Alkanes
Alkanes react with oxygen in combustion reactions to produce carbon dioxide, water, and energy. Methane is a common fuel used in homes.
General equation:

Alkenes and Alkynes
Structure and Identification
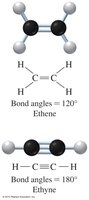
Alkenes contain one or more carbon–carbon double bonds, while alkynes contain one or more carbon–carbon triple bonds. These bonds affect the geometry and reactivity of the molecules.
Alkene: Trigonal planar arrangement, bond angles of 120°.
Alkyne: Linear arrangement, bond angles of 180°.

Naming Alkenes and Alkynes
The IUPAC names for alkenes and alkynes are based on the alkane name, replacing the 'ane' ending with 'ene' for alkenes and 'yne' for alkynes. Cyclic alkenes are named as cycloalkenes.

Guide to Naming Alkenes and Alkynes
To name alkenes and alkynes:
Name the longest carbon chain containing the double or triple bond.
Number the chain from the end nearer the bond.
Give the location and name of each substituent in alphabetical order.
Example: Naming Alkenes and Alkynes
2-pentene: Five carbon atoms, double bond at position 2.
2-hexyne: Six carbon atoms, triple bond at position 2.
4-methyl-2-pentene: Methyl group at position 4, double bond at position 2.
Naming Cycloalkenes
Cycloalkenes have a double bond within a ring. The double bond is always between carbon 1 and 2, and substituents are numbered to give the lowest possible numbers.
Example: 3-methylcyclopentene
Cis–Trans Isomers
Geometric Isomerism in Alkenes
Alkenes can exhibit cis–trans isomerism due to the rigidity of the double bond, which prevents rotation. Cis isomers have substituents on the same side; trans isomers have them on opposite sides.
Naming Cis–Trans Isomers
The prefix 'cis' or 'trans' is placed before the alkene name to indicate the isomer type.
Example: cis-1,2-dibromoethene, trans-1,2-dibromoethene
Addition Reactions of Alkenes and Alkynes
Hydrogenation
Hydrogenation is the addition of hydrogen to the carbon atoms of a double or triple bond, converting unsaturated compounds to saturated ones. A catalyst such as Pt, Ni, or Pd is used.
Example equation:
Hydration
Hydration is the addition of water to an alkene, forming an alcohol. The hydrogen atom from water bonds to the carbon with more hydrogen atoms, and the hydroxyl group bonds to the other carbon. This reaction is catalyzed by a strong acid such as H2SO4.
Summary Table: Comparison of Alkanes, Alkenes, and Alkynes
Type | General Formula | Bond Type | Example |
|---|---|---|---|
Alkane | CnH2n+2 | Single bonds | CH4, C2H6 |
Alkene | CnH2n | Double bond | C2H4, C3H6 |
Alkyne | CnH2n-2 | Triple bond | C2H2, C3H4 |
*Additional info: Academic context and examples were added to clarify concepts and ensure completeness.*
