 Back
BackChapter 3: Matter and Energy – Study Notes
Study Guide - Smart Notes

Chapter 3: Matter and Energy
3.1 Classification of Matter
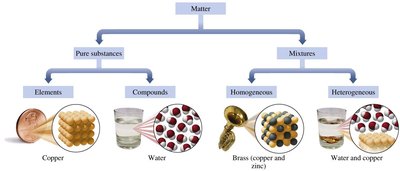
Matter is anything that has mass and occupies space. It can be classified based on its composition into pure substances and mixtures.
Pure Substances: Have a fixed or definite composition. They are further classified as:
Elements: Composed of only one type of atom (e.g., copper, aluminum, helium).
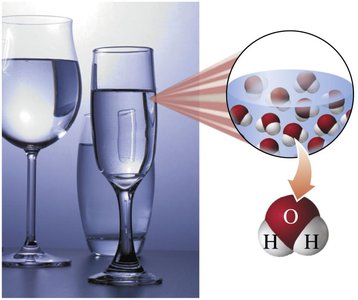
Compounds: Consist of two or more elements chemically combined in a fixed ratio (e.g., water, hydrogen peroxide, table salt).
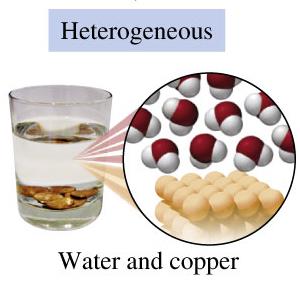
Mixtures: Contain two or more substances physically mixed, not chemically combined. The components can be present in varying proportions and separated by physical methods.


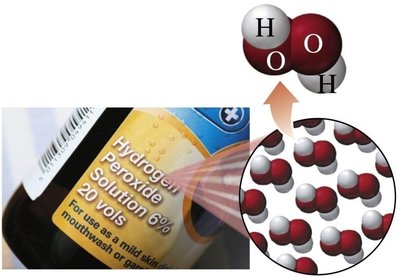
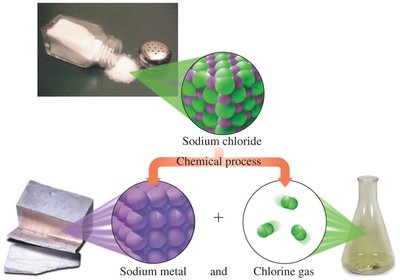

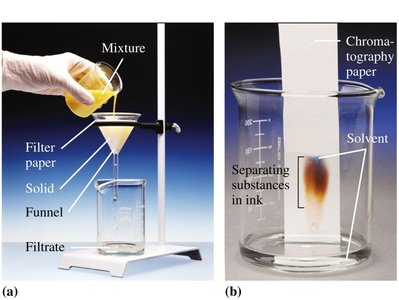
Types of Mixtures
Homogeneous Mixtures (Solutions): Uniform composition throughout; components are not visibly distinguishable (e.g., brass, air, salt water).
Heterogeneous Mixtures: Composition varies; different parts are visible (e.g., salad, sand in water).
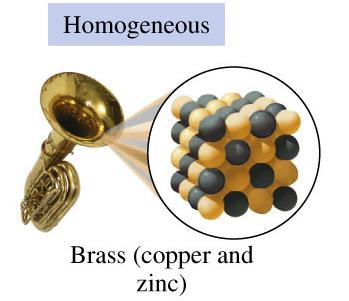

3.2 States and Properties of Matter
Matter exists in three physical states: solid, liquid, and gas. Each state has distinct properties based on particle arrangement and movement.
Solids: Definite shape and volume; particles are closely packed in a fixed arrangement and vibrate in place.
Liquids: Definite volume but no definite shape; particles are close but can move past each other, allowing liquids to flow.
Gases: No definite shape or volume; particles are far apart and move rapidly, filling the container.

Characteristic | Solid | Liquid | Gas |
|---|---|---|---|
Shape | Definite | Takes shape of container | Takes shape of container |
Volume | Definite | Definite | Fills container |
Arrangement of Particles | Fixed, very close | Random, close | Random, far apart |
Interaction between Particles | Very strong | Strong | Essentially none |
Movement of Particles | Very slow | Moderate | Very fast |
Examples | Ice, salt, iron | Water, oil, vinegar | Water vapor, helium, air |

Physical Properties
Observed or measured without changing the substance's identity (e.g., color, melting point, density).
Examples: Copper is reddish-orange, shiny, and a good conductor of heat and electricity.

Physical and Chemical Changes
Physical Change: Alters physical form, not composition (e.g., melting, boiling, dissolving, cutting).
Chemical Change: Produces new substances with different properties (e.g., rusting, burning, caramelizing sugar).



Physical Changes | Chemical Changes |
|---|---|
Water boils to form vapor | Silver reacts in air to form a black coating |
Copper drawn into wires | Wood burns to form ashes, CO2, and water vapor |
Sugar dissolves in water | Heating sugar forms caramel |
Paper cut into confetti | Iron rusts to form Fe2O3 |

Physical | Chemical | |
|---|---|---|
Property | Color, shape, odor, melting point, density | Ability to form new substances (e.g., burning, rusting) |
Change | Change in state or shape, identity retained | Original substance converted to new substances |

3.3 Temperature
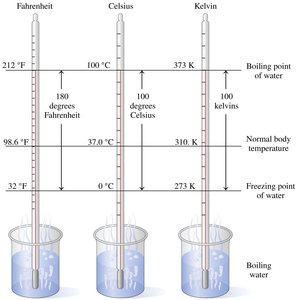
Temperature measures how hot or cold an object is compared to another. It is measured in Celsius (°C), Fahrenheit (°F), or Kelvin (K).
Celsius (°C): Used in science; water freezes at 0°C and boils at 100°C.
Fahrenheit (°F): Common in the US; water freezes at 32°F and boils at 212°F.
Kelvin (K): Used in scientific measurements; absolute zero is 0 K (−273°C).



Temperature Conversions
To convert °C to °F:
To convert °F to °C:
To convert °C to K:


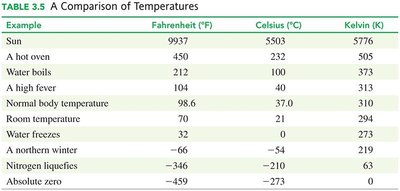
Example | Fahrenheit (°F) | Celsius (°C) | Kelvin (K) |
|---|---|---|---|
Sun | 9937 | 5503 | 5776 |
Water boils | 212 | 100 | 373 |
Normal body temperature | 98.6 | 37.0 | 310 |
Water freezes | 32 | 0 | 273 |
Absolute zero | -459 | -273 | 0 |
3.4 Energy
Energy is the ability to do work. It exists as kinetic energy (energy of motion) and potential energy (stored energy due to position or composition).
Kinetic Energy: Associated with moving objects (e.g., rollerblading, mowing the lawn).
Potential Energy: Stored energy (e.g., water behind a dam, gasoline in a tank).
Heat and Units of Energy
Heat: Energy associated with the motion of particles; measured in joules (J) or calories (cal).
1 kilojoule (kJ) = 1000 J
1 kilocalorie (kcal) = 1000 cal
1 cal = 4.184 J
Example Calculation: To convert 150 J to calories:
