 Back
BackChapter 3: Matter and Energy – Study Notes
Study Guide - Smart Notes

Matter and Energy
Introduction
Matter and energy are foundational concepts in chemistry, essential for understanding the composition, properties, and transformations of substances. This chapter explores the classification of matter, its physical states, properties, temperature, energy, and the changes matter undergoes.
Classification of Matter
Pure Substances and Mixtures
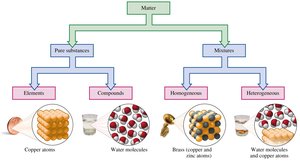
Matter is anything that has mass and occupies space. It can be classified based on its composition:
Pure Substances: Have a fixed or definite composition. They are further divided into:
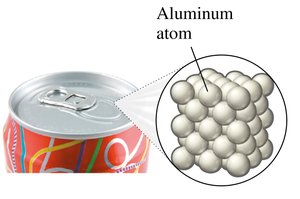
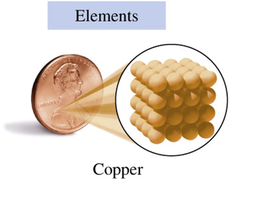
Elements: Composed of only one type of atom (e.g., copper, aluminum).
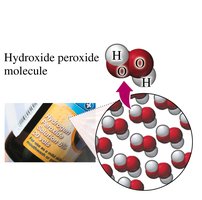
Compounds: Composed of two or more elements chemically combined in a fixed ratio (e.g., water, table salt).
Mixtures: Contain two or more substances physically mixed, not chemically combined. The composition can vary, and components can be separated by physical methods.
Examples of Pure Substances
Elements: Copper (Cu), Lead (Pb), Aluminum (Al)
Compounds: Water (H2O), Table salt (NaCl), Sugar, Hydrogen peroxide (H2O2)
Examples of Mixtures
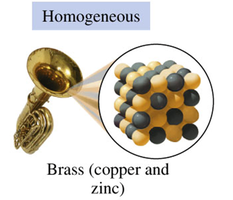
Homogeneous Mixtures (Solutions): Uniform composition throughout; components are not visibly distinct (e.g., brass, air, salt water).
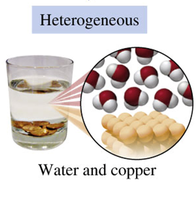
Heterogeneous Mixtures: Composition varies; different parts are visible (e.g., salad, sand and water).
Separation of Mixtures
Physical methods such as filtration and chromatography are used to separate mixtures based on physical properties.

States and Properties of Matter
Physical States
Matter exists in three physical states: solid, liquid, and gas.
Solids: Definite shape and volume; particles are closely packed in a fixed arrangement and vibrate in place.
Liquids: Definite volume but no definite shape; particles are close but can move past each other.
Gases: Neither definite shape nor volume; particles are far apart and move rapidly.

Physical and Chemical Properties
Physical Properties: Observed or measured without changing the substance's identity (e.g., color, melting point, density).
Chemical Properties: Describe the ability of a substance to undergo a chemical change, forming new substances (e.g., flammability, reactivity).

Physical and Chemical Changes
Physical Change: Alters the form or state of a substance without changing its composition (e.g., melting, boiling, dissolving).
Chemical Change: Produces one or more new substances with different properties (e.g., rusting, burning, caramelizing sugar).


Temperature
Temperature Scales
Temperature measures how hot or cold an object is. The main scales are Celsius (°C), Fahrenheit (°F), and Kelvin (K).
Celsius: Used in science; water freezes at 0°C and boils at 100°C.
Fahrenheit: Common in the United States; water freezes at 32°F and boils at 212°F.
Kelvin: The SI unit; absolute zero (0 K) is the lowest possible temperature.

Temperature Conversions
To convert between Celsius and Fahrenheit:
To convert between Celsius and Kelvin:
Energy
Kinetic and Potential Energy
Kinetic Energy: Energy of motion (e.g., moving water, running).
Potential Energy: Stored energy due to position or composition (e.g., water behind a dam, chemical bonds in food).

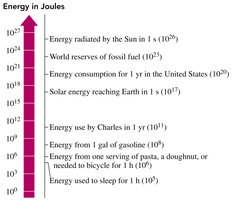
Units of Energy
The SI unit of energy is the joule (J).
1 kilojoule (kJ) = 1000 J
The calorie (cal) is another unit: 1 cal = energy to raise 1 g of water by 1°C.
1 kilocalorie (kcal) = 1000 cal
1 cal = 4.184 J
Specific Heat
Definition and Calculation
Specific Heat (SH): The amount of heat required to raise the temperature of 1 g of a substance by 1°C.
Units: J/g·°C or cal/g·°C
The heat (q) absorbed or released is calculated by:
where m is mass (g), SH is specific heat, and ΔT is the temperature change (°C).
Energy and Nutrition
Energy Values in Food
Carbohydrates, fats, and proteins provide energy measured in kilocalories (kcal) or kilojoules (kJ).
1 Calorie (nutritional Calorie) = 1 kcal = 1000 cal
Energy values are determined using a calorimeter.
Changes of State
Melting, Freezing, Sublimation, Deposition, Evaporation, Boiling, and Condensation
Melting: Solid to liquid at the melting point.
Freezing: Liquid to solid at the freezing point.
Sublimation: Solid to gas without becoming liquid (e.g., dry ice).
Deposition: Gas to solid without becoming liquid.
Evaporation: Liquid to gas at the surface below boiling point.
Boiling: Liquid to gas throughout the liquid at the boiling point.
Condensation: Gas to liquid at the boiling point (reverse of vaporization).
Heats of Fusion and Vaporization
Heat of Fusion: Energy required to melt 1 g of a solid at its melting point (for water: 334 J/g or 80 cal/g).
Heat of Vaporization: Energy required to vaporize 1 g of a liquid at its boiling point (for water: 2260 J/g or 540 cal/g).
To calculate heat for a phase change:
Heating and Cooling Curves
Heating curves show temperature changes and phase changes as heat is added.
Plateaus (horizontal lines) represent phase changes at constant temperature.
Sloped lines represent temperature changes within a single phase.
Summary Table: Classification of Matter
Type | Description | Examples |
|---|---|---|
Element | One type of atom | Copper, Aluminum |
Compound | Two or more elements chemically combined | Water, Salt |
Homogeneous Mixture | Uniform composition | Brass, Air |
Heterogeneous Mixture | Non-uniform composition | Salad, Sand and water |
Summary Table: Physical States of Matter
State | Shape | Volume | Particle Arrangement |
|---|---|---|---|
Solid | Definite | Definite | Fixed, close together |
Liquid | Indefinite | Definite | Close, can move past each other |
Gas | Indefinite | Indefinite | Far apart, move freely |
Key Equations
Temperature Conversions:
Heat Equation:
Phase Change:
Additional info: These notes are based on Chapter 3 of "Chemistry: An Introduction to General, Organic, and Biological Chemistry" and are suitable for introductory college-level chemistry courses.
