 Back
BackChapter 4: Molecular Compounds – Covalent Bonding, Structure, and Properties
Study Guide - Smart Notes

Chapter 4: Molecular Compounds
4.1 Covalent Bonds
Covalent bonds are fundamental to molecular compounds, involving the sharing of electrons between atoms, typically nonmetals. This contrasts with ionic bonds, which involve the complete transfer of electrons. The resulting group of atoms held together by covalent bonds is called a molecule.
Covalent Bond: A chemical bond formed by the sharing of electrons between two atoms.
Ionic Bond: A bond formed by the complete transfer of electrons from one atom to another.
Octet Rule: Atoms share electrons to achieve a noble gas configuration, usually eight valence electrons.
Single, Double, Triple Bonds: Two, four, or six shared electrons between atoms, respectively.
Molecule: A group of atoms held together by covalent bonds.
Example: The hydrogen molecule (H2) forms when two hydrogen atoms share their electrons, resulting in a stable bond.
Bond formation depends on the balance between attractive and repulsive forces. The optimal bond length for H2 is 74 pm, where the energy is minimized.
4.2 Covalent Bonds and the Periodic Table
Molecules can form from atoms of the same element (homonuclear) or different elements (heteronuclear). The number of covalent bonds formed by main group elements generally follows the octet rule, but there are exceptions.
Diatomic Elements: H2, N2, O2, F2, Cl2, Br2, I2 are naturally occurring as pairs.
Exceptions to Octet Rule: Boron (forms only 3 bonds), elements in the third row and below can have expanded octets due to vacant d orbitals.

4.3 Multiple Covalent Bonds
Atoms can share more than one pair of electrons, forming double or triple bonds. Carbon, nitrogen, and oxygen frequently participate in multiple bonds.
Double Bond: Four electrons shared (e.g., CO2).
Triple Bond: Six electrons shared (e.g., N2).
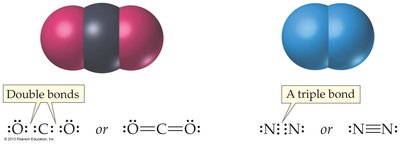
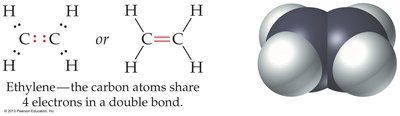
Multiple bonds are common in organic compounds such as ethylene (C2H4) and acetylene (C2H2).
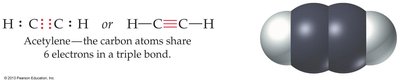
4.4 Coordinate Covalent Bonds
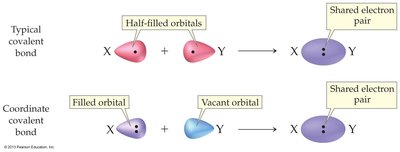
A coordinate covalent bond forms when both electrons in the shared pair are donated by the same atom, unlike typical covalent bonds where each atom donates one electron.
Typical Covalent Bond: Each atom donates one electron.
Coordinate Covalent Bond: One atom donates both electrons.
4.5 Characteristics of Molecular Compounds
Molecular compounds differ from ionic compounds in their physical properties, such as melting point, boiling point, and electrical conductivity.
Lower melting and boiling points than ionic compounds.
Do not conduct electricity when melted or dissolved.
Many are water-insoluble, but soluble in organic liquids.
Ionic Compounds | Molecular Compounds |
|---|---|
Smallest components are ions (e.g., Na+, Cl-) | Smallest components are molecules (e.g., CO2, H2O) |
Usually composed of metals with nonmetals | Usually composed of nonmetals |
Crystalline solids | Gases, liquids, or low-melting solids |
High melting/boiling points | Low melting/boiling points |
Conduct electricity when molten/dissolved | Do not conduct electricity |
Many are water-soluble | Relatively few are water-soluble |
Not soluble in organic liquids | Many are soluble in organic liquids |

4.6 Molecular Formulas and Lewis Structures
Molecular compounds can be represented in several ways, including molecular formulas, condensed structures, structural formulas, space-filling models, ball-and-stick models, and Lewis structures.
Molecular Formula: Shows the numbers and kinds of atoms.
Condensed Structure: Minimal structural information, bonds inferred.
Structural Formula: Bonds shown as lines.
Space-Filling Model: Shows bonding and relative size of atoms.
Ball-and-Stick Model: Shows bonding and 3-D structure.
Lewis Structure: Shows connections among atoms and locations of lone-pair valence electrons.
4.7 Drawing Lewis Structures
Lewis structures are drawn using two main approaches: common bonding patterns (useful for organic molecules) and a general stepwise procedure (works for all molecules).
Common Bonding Patterns: C forms 4 bonds, N forms 3 bonds and 1 lone pair, O forms 2 bonds and 2 lone pairs, halogens form 1 bond and 3 lone pairs, H forms 1 bond.
Stepwise Procedure:
Count total valence electrons.
Draw skeleton structure with single bonds.
Distribute remaining electrons to peripheral atoms to achieve octet.
Place remaining lone pairs on central atom.
If central atom lacks octet, form multiple bonds by sharing lone pairs.
4.8 The Shapes of Molecules (VSEPR Theory)
The shape of a molecule is determined by the number and location of valence electrons around the atoms. The Valence Shell Electron Pair Repulsion (VSEPR) model predicts molecular geometry by minimizing repulsion between electron pairs.
Linear: 2 electron clouds, bond angle 180°
Planar Triangular: 3 electron clouds, bond angle 120°
Tetrahedral: 4 electron clouds, bond angle 109.5°
Bent: Bond angle less than 120° or 109.5° due to lone pairs
Steps to apply VSEPR:
Draw Lewis structure and identify central atom.
Count electron charge clouds (single, double, triple bonds, and lone pairs each count as one cloud).
Predict shape by orienting charge clouds as far apart as possible.
4.9 Polar Covalent Bonds and Electronegativity
Bond polarity describes the sharing of electrons between atoms. If electrons are shared equally, the bond is nonpolar; if unequally, the bond is polar. Electronegativity is the ability of an atom to attract electrons in a bond.
Nonpolar Covalent Bond: Electrons shared equally (e.g., Cl2, H2).
Polar Covalent Bond: Electrons shared unequally (e.g., H2O, HCl).
Electronegativity: Increases across a period, decreases down a group. Highest for fluorine (F = 4.0).
Bond polarity increases with electronegativity difference:
E.N. difference < 0.5: Slightly polar covalent
0.5 < E.N. difference < 1.9: Increasingly polar covalent
2.0 ≤ E.N. difference: Ionic
4.10 Polar Molecules
Molecular polarity depends on both the polarity of individual bonds and the shape of the molecule. Symmetrical molecules may be nonpolar even if they contain polar bonds.
Electrostatic Potential Map: Visualizes electron distribution and molecular polarity.
Physical Properties: Polarity affects solubility, melting point, boiling point, and other properties.
4.11 Naming Binary Molecular Compounds
Binary molecular compounds consist of two different types of atoms. Naming rules include:
Least electronegative element written first.
Prefixes indicate the number of atoms (mono-, di-, tri-, etc.).
Second element ends with '-ide'.
Prefix 'mono-' is omitted for the first element.
Examples:
H2O2: dihydrogen dioxide (hydrogen peroxide)
CS2: carbon disulfide
CF4: carbon tetrafluoride
NO: nitrogen monoxide
Chapter Summary
Covalent bonds involve sharing electrons; ionic bonds involve transfer.
Single, double, and triple bonds correspond to two, four, and six shared electrons.
Molecules are groups of atoms held by covalent bonds.
Coordinate covalent bonds involve both electrons donated by one atom.
Atoms share electrons to achieve noble gas configuration.
Molecular formulas show atom types and numbers; Lewis structures show connections and lone pairs.
VSEPR model predicts molecular shapes based on electron repulsion.
Bond polarity and molecular polarity depend on electronegativity and shape.
Molecular compounds have lower melting/boiling points, are often water-insoluble, and do not conduct electricity.
