 Back
BackChapter 5: Chemical Reactions – Thermodynamics and Kinetics
Study Guide - Smart Notes

Chemical Reactions
Thermodynamics in Chemical Reactions
Thermodynamics is the study of energy changes that occur during chemical reactions. It helps us understand whether a reaction will occur spontaneously and how energy is transferred or transformed.
Energy: Defined as the capacity to do work. Chemical reactions involve exchanges of energy, often transforming potential energy (stored in chemical bonds) into kinetic energy.
Reactants and Products: In chemical equations, reactants are on the left, products on the right, and the arrow indicates the direction of the reaction.
Chemical Energy: Stored in chemical bonds as potential energy. Breaking bonds releases energy, and forming new bonds may require or release energy.
Heat of Reactions
Heat is a form of energy released or absorbed during chemical reactions. The nature of heat exchange classifies reactions as exothermic or endothermic.
Exothermic Reaction: Releases energy as heat. Example: Combustion of methane.
Endothermic Reaction: Absorbs energy as heat.
Chemical Energy: Potential energy stored in the structure of substances.
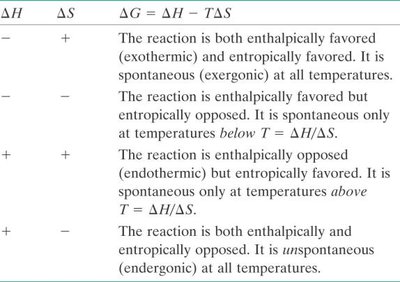
Enthalpy (ΔH)
Enthalpy is the heat content of a system. The change in enthalpy (ΔH) during a reaction indicates whether heat is released or absorbed.
Exothermic: ΔH is negative; products have lower enthalpy than reactants.
Endothermic: ΔH is positive; products have higher enthalpy than reactants.
Thermodynamically favorable: Decreased enthalpy (-ΔH).
Entropy (ΔS)
Entropy measures the disorder or randomness in a system. The change in entropy (ΔS) is the difference between the entropy of products and reactants.
Increased disorder: ΔS is positive; thermodynamically favorable.
Decreased disorder: ΔS is negative; thermodynamically unfavorable.
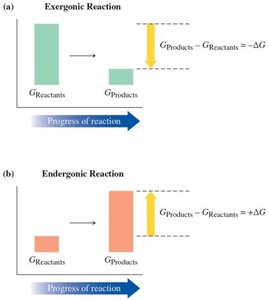
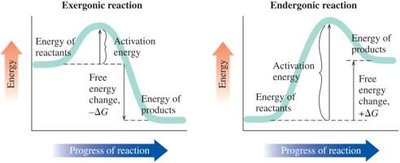
Gibbs Free Energy (ΔG)
Gibbs Free Energy combines enthalpy and entropy to determine if a reaction is spontaneous. The equation is:
ΔG < 0: Reaction is exergonic, spontaneous.
ΔG > 0: Reaction is endergonic, not spontaneous.
ΔG = 0: Reaction is at equilibrium.
Determining Spontaneity
Spontaneity depends on the sign and magnitude of ΔG. Exergonic reactions occur naturally, while endergonic reactions require energy input.
Exergonic: ΔG < 0, spontaneous.
Endergonic: ΔG > 0, not spontaneous; reverse reaction is spontaneous.
Equilibrium: ΔG = 0, no net reaction occurs.
Chemical Reactions in Biology
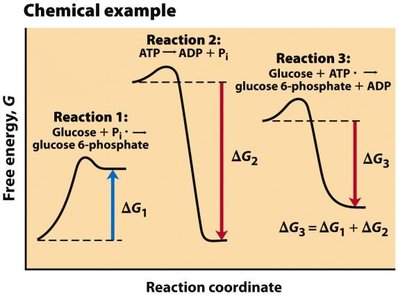
Biological systems use free energy to perform work under physiological conditions. Energy released from one reaction is often used to drive another, especially through ATP.
ATP (Adenosine Triphosphate): Main energy carrier in cells, produced from food metabolism.
Coupled Reactions: Endergonic reactions are powered by exergonic reactions, making the overall process exergonic.
Energy in Food
Food contains chemical energy, measured in Calories. The energy content depends on the composition of fats, carbohydrates, and proteins.
Combustion: Reaction of food molecules with oxygen produces CO2, H2O, and energy.
Energy Calculation: Fats provide 9 Cal/g, carbohydrates and proteins provide 4 Cal/g.

Thermodynamics vs Kinetics
Thermodynamics tells us if a reaction will occur, but kinetics tells us how fast it will happen. Activation energy is the initial energy required to start a reaction.
Activation Energy: The energy needed to initiate a reaction.
Thermodynamics: Relationship between reactants and products (ΔG).
Kinetics: Rate of reaction, determined by how quickly reactants are converted to products.
Chemical Reaction Kinetics
Measuring Reaction Rate
The rate of a chemical reaction is measured by the amount of product formed or reactant consumed over time. For a reaction to occur, reactant molecules must collide with sufficient energy and proper orientation.
Collision Theory: Reactants must collide with enough energy and correct orientation.
Factors Affecting Rate: Temperature, concentration, and catalysts.
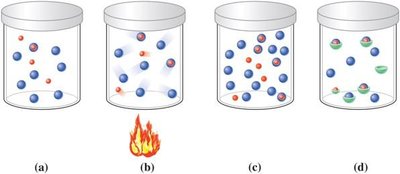
Changing the Rate of a Reaction
Several factors can increase the rate of a chemical reaction:
Temperature: Higher temperature increases kinetic energy, leading to more frequent and energetic collisions.
Amount of Reactant: Higher concentration increases collision frequency.
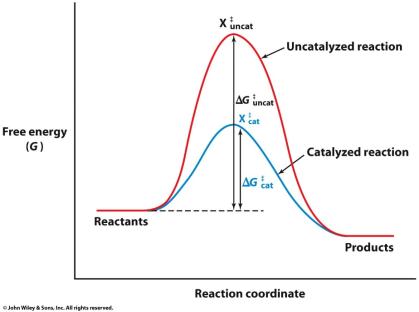
Catalysts: Substances that speed up reactions by lowering activation energy, without being consumed.
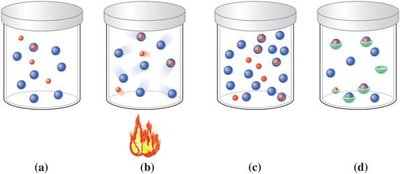
Catalysts and Activation Energy
Catalysts lower the activation energy required for a reaction, increasing the reaction rate. They are not consumed in the reaction and can be reused.
Acids, bases, and metal ions often act as catalysts.
Immobilization: Catalysts often bring reactants together, increasing collision likelihood.
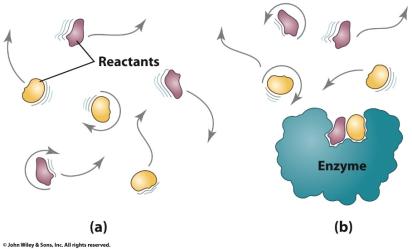
Enzymes as Biological Catalysts
Enzymes are proteins that act as highly specific catalysts in biological systems. They increase reaction rates by immobilizing reactants at the active site and orienting them for reaction.
Enzyme Specificity: Enzymes are specific to particular reactions.
Activation Energy Reduction: Enzymes lower activation energy, increasing reaction rate.
Enzyme Deficiency: Defective enzymes can lead to disease (e.g., lactose intolerance).
Enzymes and Disease
Many diseases are linked to improper enzyme function. The table below summarizes some common disorders, their associated enzymes, and clinical symptoms.
Disease | Enzyme | Clinical Symptoms |
|---|---|---|
Albinism | Tyrosinase | Absence of pigment in skin, hair, and eyes |
Gout | Phosphoribosyl pyrophosphate Synthetase | Renal problems, joint problems, high levels of uric acid in the bloodstream |
Lactose intolerance | Lactase | Diarrhea, bloating, flatulence, nausea, abdominal cramping |
Glycogen storage deficiency | Glycogen synthase | Enlarged fatty liver, low sugar levels during fasting |
Phenylketonuria (PKU) | Phenylalanine hydroxylase | Neurologic symptoms, mental retardation |
Tay–Sachs disease | Hexosaminidase A | Mental retardation, blindness, muscular weakness, death |
