 Back
BackChapter 5: Nuclear Chemistry and Radioactivity Study Guide
Study Guide - Smart Notes

Radioactivity and Nuclear Chemistry
Stable vs Unstable Isotopes
Atoms of the same element can have different numbers of neutrons, resulting in isotopes. The stability of an isotope depends on the ratio of protons to neutrons in its nucleus. Stable isotopes have a balanced ratio, while unstable isotopes undergo radioactive decay to achieve stability.
Isotope: Atoms of the same element with different numbers of neutrons.
Stable Isotopes: Do not undergo radioactive decay.
Unstable Isotopes: Undergo radioactive decay, emitting radiation.
Example: Magnesium has three common isotopes: 24Mg, 25Mg, and 26Mg.
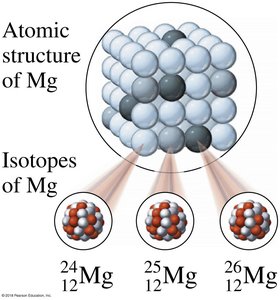
Classification of Isotopes
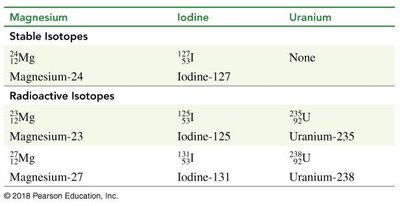
Isotopes can be classified as stable or radioactive. Radioactive isotopes emit radiation as they decay.
Element | Stable Isotopes | Radioactive Isotopes |
|---|---|---|
Magnesium | 24Mg | 23Mg, 27Mg |
Iodine | 127I | 125I, 131I |
Uranium | None | 235U, 238U |
Types of Radioactive Decay
Alpha Decay
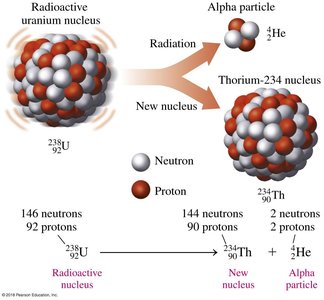
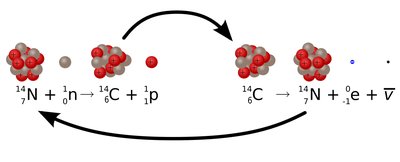
Alpha decay occurs when an unstable nucleus emits an alpha particle, which consists of two protons and two neutrons (a helium nucleus). This process decreases the atomic number by 2 and the mass number by 4.
Alpha Particle: or
Example:
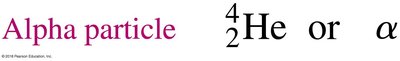
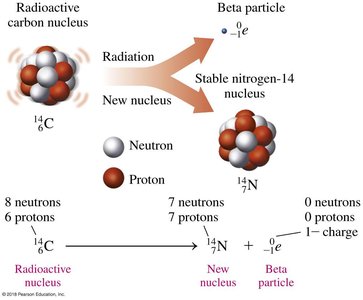
Beta Decay
Beta decay occurs when a neutron in the nucleus transforms into a proton and emits a beta particle (an electron). This increases the atomic number by 1, while the mass number remains unchanged.
Beta Particle: or
Example:
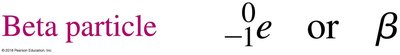
Positron Emission
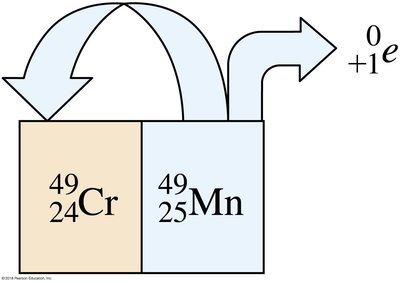
Positron emission occurs when a proton in the nucleus transforms into a neutron and emits a positron (the antimatter equivalent of an electron). This decreases the atomic number by 1, while the mass number remains unchanged.
Positron: or
Example:
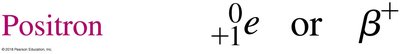
Gamma Emission
Gamma emission involves the release of high-energy electromagnetic waves (gamma rays) from the nucleus. Gamma rays do not change the atomic number or mass number, but they release excess energy.
Gamma Ray: or
Summary of Radiation Types
Different types of radioactive decay produce different particles or waves, each affecting the nucleus in specific ways.
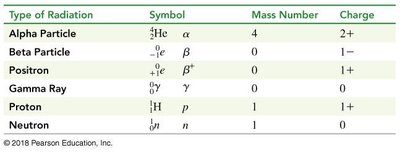
Type | Symbol | Mass Number | Charge |
|---|---|---|---|
Alpha Particle | 4 | 2+ | |
Beta Particle | 0 | 1− | |
Positron | 0 | 1+ | |
Gamma Ray | 0 | 0 | |
Proton | 1 | 1+ | |
Neutron | 1 | 0 |
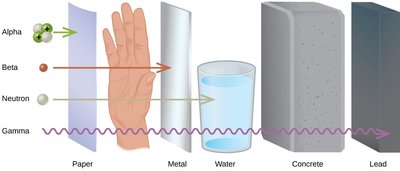
Penetration Ability of Radiation
Comparing Radiation Types
The ability of radiation to penetrate materials varies by type. Alpha particles are stopped by paper, beta particles by metal, neutrons by water, and gamma rays require dense materials like lead for shielding.
Alpha: Least penetrating, stopped by paper.
Beta: Moderate penetration, stopped by metal.
Gamma: Most penetrating, requires lead or concrete.
Measurement of Radiation
Units of Measurement
Radiation is measured in terms of activity, absorbed dose, and biological damage. Common units include curie (Ci), rad, and rem, while SI units are becquerel (Bq), gray (Gy), and sievert (Sv).
Measurement | Common Unit | SI Unit | Relationship |
|---|---|---|---|
Activity | curie (Ci) | becquerel (Bq) | 1 Ci = 3.7 × 1010 Bq |
Absorbed Dose | rad | gray (Gy) | 1 Gy = 100 rad |
Biological Damage | rem | sievert (Sv) | 1 Sv = 100 rem |

Sources and Effects of Radiation
Common Radiation Sources
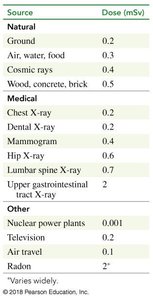
Radiation exposure comes from both natural and artificial sources. The average annual dose for Americans is about 3.6 mSv.
Source | Dose (mSv) |
|---|---|
Ground | 0.2 |
Air, water, food | 0.3 |
Cosmic rays | 0.4 |
Wood, concrete, brick | 0.5 |
Chest X-ray | 0.2 |
Dental X-ray | 0.2 |
Mammogram | 0.4 |
Hip X-ray | 0.6 |
Lumbar spine X-ray | 0.7 |
Upper GI X-ray | 0.6 |
Nuclear power plants | 0.001 |
Television | 0.2 |
Air travel | 0.1 |
Radon | 2* |
Toxic Radiation Exposure
High doses of radiation can cause severe biological effects, including radiation sickness and death. The LD50 (lethal dose for 50% of population) varies by organism.
Life Form | LD50 (Sv) |
|---|---|
Insect | 1000 |
Bacterium | 500 |
Rat | 8 |
Human | 5 |
Dog | 3 |
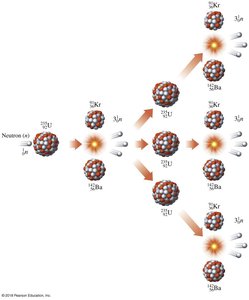
Nuclear Fission and Fusion
Chain Reactions (Fission)
Nuclear fission is the splitting of a heavy nucleus into lighter nuclei, releasing energy and neutrons. These neutrons can cause further fission reactions, resulting in a chain reaction.
Example:
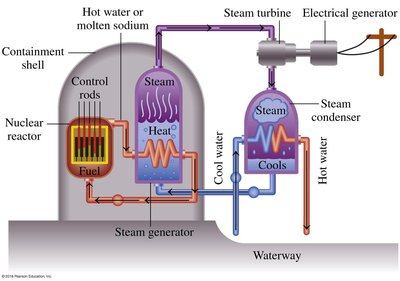
Nuclear Power Reactors (Fission)
Nuclear reactors use controlled fission reactions to generate heat, which is used to produce steam and drive turbines for electricity generation.
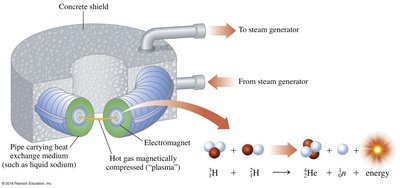
Fusion
Nuclear fusion is the process where two light nuclei combine to form a heavier nucleus, releasing large amounts of energy. Fusion is the energy source of stars, including the Sun.
Example:
Half-Life and Radioactive Decay
Half-Life
The half-life of a radioactive isotope is the time required for half of the sample to decay. This property is used to estimate the age of materials and to understand the persistence of radioactivity.
Formula:
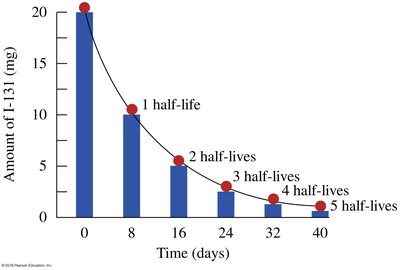
Example: Iodine-131 has a half-life of 8 days.
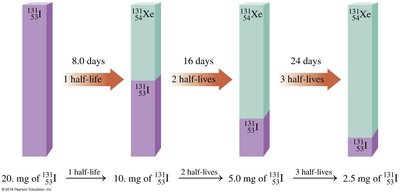
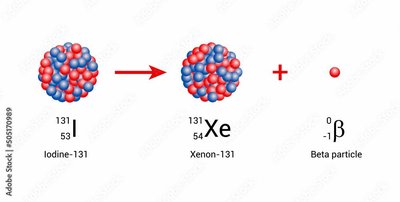
Half-Lives of Isotopes
Different isotopes have different half-lives, ranging from seconds to millions of years. This affects their use in medicine, dating, and environmental studies.
Element | Radioisotope | Half-Life | Type of Radiation |
|---|---|---|---|
Carbon | 5730 yr | Beta | |
Potassium | 1.3 × 109 yr | Beta, gamma | |
Radium | 1600 yr | Alpha | |
Strontium | 28.8 yr | Beta | |
Uranium | 4.5 × 109 yr | Alpha |
Carbon Dating (Beta Decay)
Carbon dating uses the decay of carbon-14 to estimate the age of organic materials. Living organisms constantly exchange carbon with the environment, but after death, the carbon-14 decays at a known rate.
Formula:
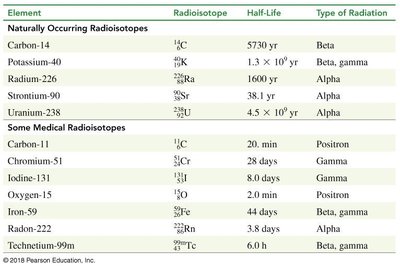
Medical Applications of Radioactivity
Radiotracers and PET Scans
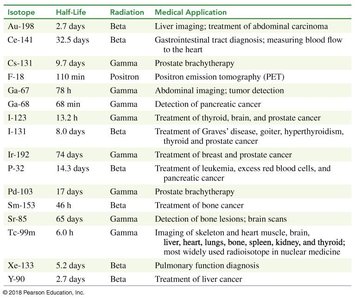
Radioactive isotopes are used in medicine for diagnosis and treatment. Radiotracers help visualize biological processes, and positron emission tomography (PET) scans use positron-emitting isotopes to image organs and tissues.
Isotope | Half-Life | Radiation | Medical Application |
|---|---|---|---|
Au-198 | 2.7 days | Beta | Liver imaging; treatment of abdominal carcinoma |
Ce-141 | 32.5 days | Beta | Gastrointestinal tract diagnosis |
C-11 | 20 min | Positron | PET imaging |
I-131 | 8.0 days | Beta | Treatment of thyroid, brain, and prostate cancer |
Xe-133 | 5.2 days | Beta | Pulmonary function diagnosis |
Radiation Medical Applications
Radioisotopes are used for imaging, cancer treatment, and monitoring organ function. The choice of isotope depends on its half-life and type of radiation emitted.
 Additional info: The notes above expand on the original content by providing definitions, formulas, and context for each topic, ensuring completeness and academic quality for exam preparation.
Additional info: The notes above expand on the original content by providing definitions, formulas, and context for each topic, ensuring completeness and academic quality for exam preparation.
