 Back
BackChapter 8: Gases – Properties, Laws, and Applications
Study Guide - Smart Notes

Gases: Properties and Measurement
Properties That Describe a Gas
Gases are characterized by four main properties: pressure, volume, temperature, and amount. Understanding these properties is essential for describing and predicting the behavior of gases in various conditions.
Pressure (P): The force exerted by a gas against the walls of its container. Measured in atmosphere (atm), millimeters of mercury (mmHg), torr, pascal (Pa).
Volume (V): The space occupied by a gas. Measured in liters (L) or milliliters (mL).
Temperature (T): Determines kinetic energy and rate of motion of gas particles. Measured in degrees Celsius (°C) or Kelvin (K).
Amount (n): The quantity of gas present, measured in grams or moles.

Units for Measuring Pressure
Pressure can be measured in several units, each with a specific equivalence to 1 atmosphere. Conversion between units is often required in gas law calculations.
Unit | Abbreviation | Equivalent to 1 atm |
|---|---|---|
Atmosphere | atm | 1 atm |
Millimeters of Hg | mmHg | 760 mmHg |
Torr | torr | 760 torr |
Inches of Hg | in. Hg | 29.9 in. Hg |
Pounds per square inch | psi | 14.7 lb/in2 |
Pascal | Pa | 101,325 Pa |
Kilopascal | kPa | 101.325 kPa |
Kinetic Molecular Theory of Gases
Fundamental Assumptions
The kinetic molecular theory explains the behavior of gases based on the motion and interactions of their particles:
Gases consist of small particles (atoms or molecules) moving randomly with high velocities.
Attractive forces between gas particles are very small.
The actual volume occupied by gas molecules is extremely small compared to the total volume.
Gas particles move rapidly in straight paths and are in constant motion.
The average kinetic energy of gas molecules is proportional to the Kelvin temperature.
Gas Pressure and Measurement
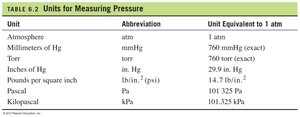
Barometer and Atmospheric Pressure
Gas pressure is the result of collisions of gas particles with the container walls. It can be measured using a barometer, which compares atmospheric pressure to the height of a mercury column.
At 1 atmosphere, the mercury column is exactly 760 mm high.
Pressure can be calculated as:
Gas Laws
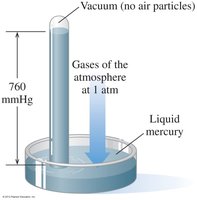
Boyle’s Law: Pressure-Volume Relationship
Boyle’s Law states that the pressure of a gas is inversely related to its volume when temperature and amount are constant.
Mathematical relationship:
If volume decreases, pressure increases.
Example: Compressing a gas in a piston increases its pressure.
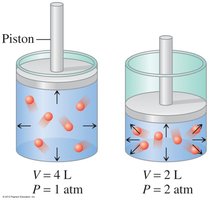
Charles’s Law: Temperature-Volume Relationship
Charles’s Law states that the volume of a gas is directly proportional to its Kelvin temperature at constant pressure and amount.
Mathematical relationship:
When temperature increases, volume increases.
Example: Heating a gas causes it to expand.
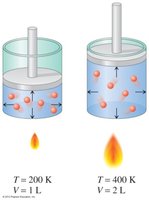
Gay-Lussac’s Law: Temperature-Pressure Relationship
Gay-Lussac’s Law states that the pressure of a gas is directly proportional to its Kelvin temperature at constant volume and amount.
Mathematical relationship:
When temperature increases, pressure increases.
Summary of Gas Laws
The main gas laws can be summarized as follows:
Law | Properties Held Constant | Relationship |
|---|---|---|
Boyle’s Law | T, n | |
Charles’s Law | P, n | |
Gay-Lussac’s Law | V, n |
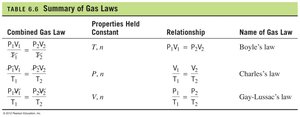
Combined Gas Law
The combined gas law incorporates Boyle’s, Charles’s, and Gay-Lussac’s laws to relate pressure, volume, and temperature when the amount of gas is constant:
Mathematical relationship:
Gas Behavior in Biological Systems
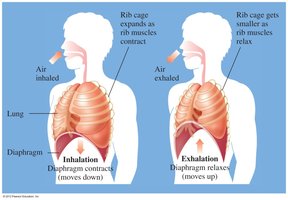
Inhalation and Exhalation
Gas laws explain the mechanics of breathing. During inhalation, lung volume increases and pressure decreases, causing air to flow in. During exhalation, lung volume decreases and pressure increases, causing air to flow out.
Avogadro’s Law: Volume and Moles
Relationship Between Volume and Amount
Avogadro’s Law states that the volume of a gas is directly proportional to the number of moles at constant temperature and pressure.
Mathematical relationship:
If the number of moles doubles, the volume doubles.
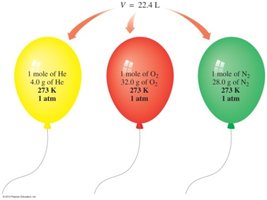
Standard Temperature and Pressure (STP) and Molar Volume
At STP (0°C, 1 atm), one mole of any gas occupies 22.4 L. This is known as the molar volume and is used for conversions between moles and volume.
Equality: 1 mole of gas at STP = 22.4 L
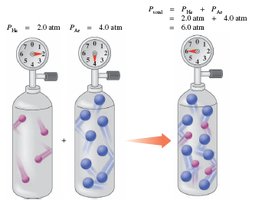
Dalton’s Law of Partial Pressures
Partial Pressures in Gas Mixtures
Dalton’s Law states that the total pressure exerted by a mixture of gases is the sum of the partial pressures of each individual gas.
Mathematical relationship:
Pressure depends on the total number of gas particles, not their type.
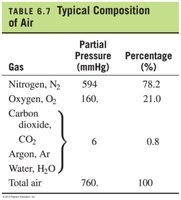
Composition of Air
Air is a mixture of gases, and its total pressure is the sum of the partial pressures of its components. The typical composition of air is as follows:
Gas | Partial Pressure (mmHg) | Percentage (%) |
|---|---|---|
Nitrogen, N2 | 594 | 78.2 |
Oxygen, O2 | 160 | 21.0 |
Carbon dioxide, CO2 | 6 | 0.8 |
Argon, Ar | ||
Water, H2O | ||
Total air | 760 | 100 |
Practice and Applications
Sample Problems
Convert pressure units (e.g., mmHg to atm, atm to torr).
Apply Boyle’s, Charles’s, and Gay-Lussac’s laws to calculate changes in gas properties.
Use Avogadro’s Law to relate volume and moles.
Calculate total pressure in gas mixtures using Dalton’s Law.
Example: A scuba tank contains O2 at 0.450 atm and He at 855 mmHg. What is the total pressure in mmHg?
Example: What is the volume of 64.0 g of O2 at STP?
Example: How many grams of N2 are in 5.6 L of N2 at STP?
Additional info: Practice problems reinforce understanding of gas laws and their applications in real-world and laboratory settings.
